The development of near-infrared II (NIR-II, 1000–1700 nm) technologies has opened new opportunities for biomedical applications, particularly in cancer diagnosis and therapy. Compared with conventional NIR-I systems (700–900 nm), NIR-II nanoplatforms offer deeper tissue penetration, reduced photon scattering, and minimal autofluorescence, enabling high-resolution, high-contrast imaging and improved therapeutic performance. A wide range of NIR-II-responsive nanostructures has been developed, including metal-based, carbon-based, semiconductor-based, hybrid/composite nanostructures, and organic fluorophores. These nanomaterials exhibit unique optical and physicochemical properties that support advanced applications such as fluorescence imaging (FLI), photothermal therapy (PTT), and photodynamic therapy (PDT), particularly in cancer theranostics. This review systematically summarizes recent advances and integration strategies of NIR-II functional nanostructures, with a particular focus on their roles in cancer imaging and image-guided therapy. However, key challenges associated with clinical translation including long-term toxicity, biodistribution, clearance, scalability, cost, and stability remain significant and are critically discussed. Finally, future perspectives are outlined, emphasizing the development of biocompatible, efficient, and clinically translatable NIR-II nanoplatforms.
- Open Access
- Review
Integration of NIR-II Functional Nanostructures for Cancer Theranostics
Author Information
Received: 16 Jan 2026 | Revised: 08 Apr 2026 | Accepted: 13 Apr 2026 | Published: 14 Apr 2026
Abstract
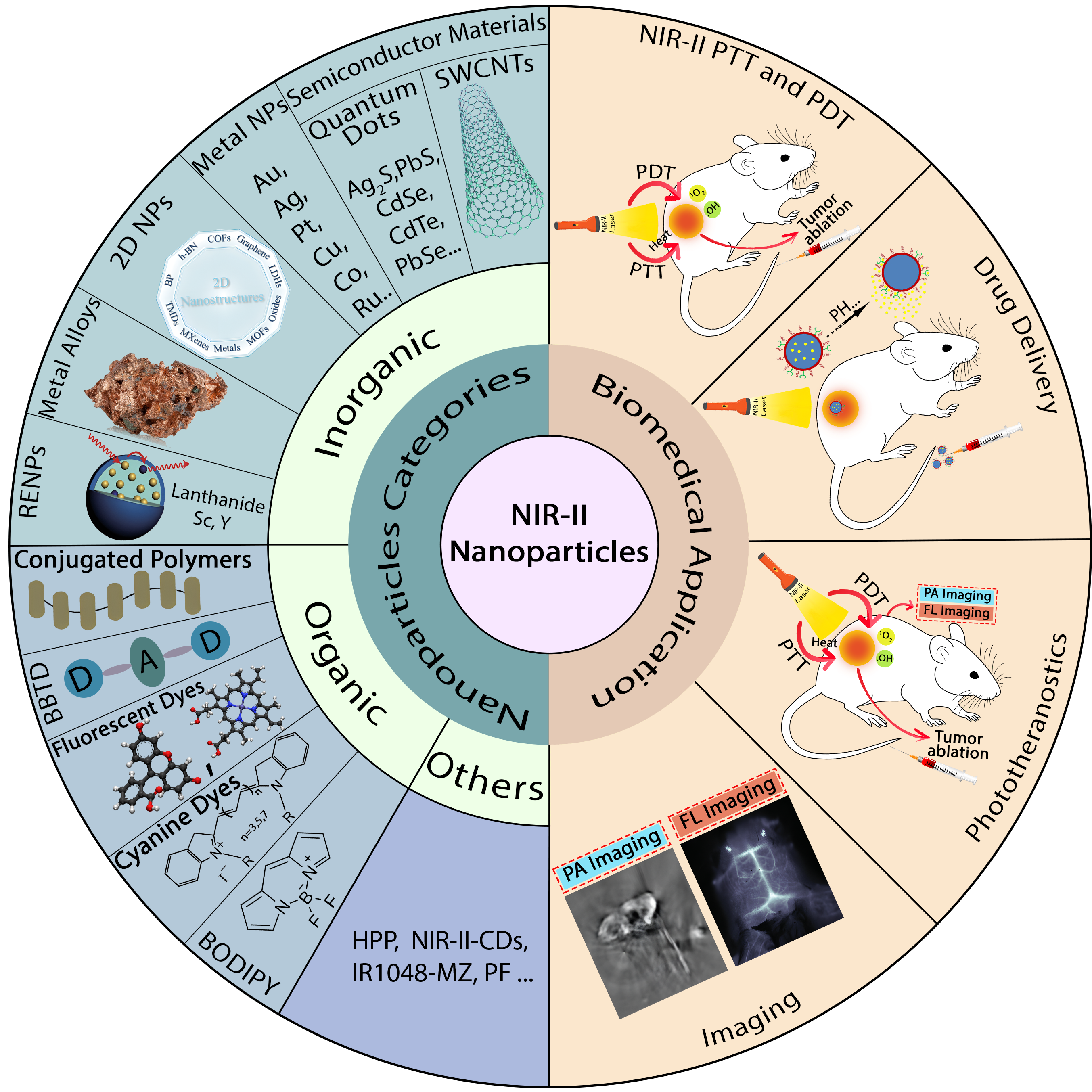
Graphical Abstract
References
- 1.
Torre, L.A.; Trabert, B.; DeSantis, C.E.; et al. Ovarian cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 284–296.
- 2.
Yang, Q.; Ma, Z.; Wang, H.; et al. Rational design of molecular fluorophores for biological imaging in the NIR-II window. Adv. Mater. 2017, 29, 1605497.
- 3.
Chen Y, Sun B, Jiang X; et al. Double-acceptor conjugated polymers for NIR-II fluorescence imaging and NIR-II photothermal therapy applications. J. Mater. Chem. B 2021, 9, 1002–1008.
- 4.
Suo Y, Wu F, Xu P; et al. NIR-II fluorescence endoscopy for targeted imaging of colorectal cancer. Adv. Healthc. Mater. 2019, 8, 1900974.
- 5.
Liu, Y.; Bhattarai, P.; Dai, Z.; et al. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem. Soc. Rev. 2019, 48, 2053–2108.
- 6.
Yuan, S.; Zhao, Y.; Liu, S.; et al. Optical imaging probes for mitochondrial metabolism: Mechanism, design and frontier applications. Coord. Chem. Rev. 2026, 553, 217570.
- 7.
Hilderbrand, S.A.; Weissleder, R. Near-infrared fluorescence: Application to in vivo molecular imaging. Curr. Opin. Chem. Biol. 2010, 14, 71–79.
- 8.
Chen, G.; Qiu, H.; Prasad, P.N.; et al. Upconversion nanoparticles: Design, nanochemistry, and applications in theranostics. Chem. Rev. 2014, 114, 5161–5214.
- 9.
Sevick-Muraca, E.M.; Houston, J.P.; Gurfinkel, M. Fluorescence-enhanced, near infrared diagnostic imaging with contrast agents. Curr. Opin. Chem. Biol. 2002, 6, 642–650.
- 10.
Cao, J.; Zhu, B.; Zheng, K.; et al. Recent progress in NIR-II contrast agent for biological imaging. Front. Bioeng. Biotechnol. 2020, 7, 487.
- 11.
Hu, Z.; Fang, C.; Li, B.; et al. First-in-human liver-tumour surgery guided by multispectral fluorescence imaging in the visible and near-infrared-I/II windows. Nat. Biomed. Eng. 2020, 4, 259–271.
- 12.
Zhen, X.; Pu, K.; Jiang, X. Photoacoustic imaging and photothermal therapy of semiconducting polymer nanoparticles: Signal amplification and second near-infrared construction. Small 2021, 17, 2004723.
- 13.
Wen, G.; Li, X.; Zhang, Y.; et al. Effective phototheranostics of brain tumor assisted by near-infrared-II light-responsive semiconducting polymer nanoparticles. ACS Appl. Mater. Interfaces 2020, 12, 33492–33499.
- 14.
Zhu, P.; Gao, S.; Lin, H.; et al. Inorganic nanoshell-stabilized liquid metal for targeted photonanomedicine in NIR-II biowindow. Nano Lett. 2019, 19, 2128–2137.
- 15.
Sun, T.; Han, J.; Liu, S.; et al. Tailor-made semiconducting polymers for second near-infrared photothermal therapy of orthotopic liver cancer. ACS Nano 2019, 13, 7345–7354.
- 16.
Hu, K.; Xie, L.; Zhang, Y.; et al. Marriage of black phosphorus and Cu2+ as effective photothermal agents for PET-guided combination cancer therapy. Nat. Commun. 2020, 11, 2778.
- 17.
Zha, Z.; Yue, X.; Ren, Q.; et al. Uniform polypyrrole nanoparticles with high photothermal conversion efficiency for photothermal ablation of cancer cells. Adv. Mater. 2012, 25, 777–782.
- 18.
Hu, X.; Lu, Y.; Dong, C.; et al. A RuII Polypyridyl Alkyne Complex Based Metal–Organic Frameworks for Combined Photodynamic/Photothermal/Chemotherapy. Chem. Eur. J. 2020, 26, 1668–1675.
- 19.
Li, Z.; Hu, Y.; Jiang, T.; et al. Human-serum-albumin-coated prussian blue nanoparticles as pH-/thermotriggered drug-delivery vehicles for cancer thermochemotherapy. Part. Part. Syst. Charact. 2016, 33, 53–62.
- 20.
Li, X.; Lovell, J.F.; Yoon, J.; et al. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat. Rev. Clin. Oncol. 2020, 17, 657–674.
- 21.
Tang, B.; Li, W.L.; Chang, Y.; et al. A supramolecular radical dimer: High-efficiency NIR-II photothermal conversion and therapy. Angew. Chem. Int. Ed. 2019, 58, 15526–15531.
- 22.
Huang, J.; Huang, Y.; Xue, Z.; et al. Tumor microenvironment responsive hollow mesoporous Co9S8@ MnO2-ICG/DOX intelligent nanoplatform for synergistically enhanced tumor multimodal therapy. Biomaterials 2020, 262, 120346.
- 23.
Zhang, Y.; Zhang, S.; Zhang, Z.; et al. Recent progress on NIR-II photothermal therapy. Front. Chem. 2021, 9, 728066.
- 24.
Jeevanandam P, Mulukutla R, Phillips M; et al. Near infrared reflectance properties of metal oxide nanoparticles. J. Phys. Chem. C 2007, 111, 1912–1918.
- 25.
Yang, N.; Gong, F.; Zhou, Y.; et al. A general in-situ reduction method to prepare core-shell liquid-metal/metal nanoparticles for photothermally enhanced catalytic cancer therapy. Biomaterials 2021, 277, 121125.
- 26.
Paramasivam, G.; Kayambu, N.; Rabel, A.M.; et al. Anisotropic noble metal nanoparticles: Synthesis, surface functionalization and applications in biosensing, bioimaging, drug delivery and theranostics. Acta Biomater. 2017, 49, 45–65.
- 27.
Kuchur, O.; Tsymbal, S.; Shestovskaya, M.; et al. Metal-derived nanoparticles in tumor theranostics: Potential and limitations. J. Inorg. Biochem. 2020, 209, 111117.
- 28.
Liu, Y.; Zhang, X.; Luo, L.; et al. Self-assembly of stimuli-responsive Au–Pd bimetallic nanoflowers based on betulinic acid liposomes for synergistic chemo-photothermal cancer therapy. ACS Biomater. Sci. Eng. 2018, 4, 2911–2921.
- 29.
Li, L.; Chen, C.; Liu, H.; et al. Multifunctional carbon–silica nanocapsules with gold core for synergistic photothermal and chemo-cancer therapy under the guidance of bimodal imaging. Adv. Funct. Mater. 2016, 26, 4252–4261.
- 30.
Luo, L.; Sun, W.; Feng, Y.; et al. Conjugation of a scintillator complex and gold nanorods for dual-modal image-guided photothermal and X-ray-induced photodynamic therapy of tumors. ACS Appl. Mater. Interfaces 2020, 12, 12591–12599.
- 31.
Sangnier, A.P.; Aufaure, R.; Cheong, S.; et al. Raspberry-like small multicore gold nanostructures for efficient photothermal conversion in the first and second near-infrared windows. Chem. Commun. 2019, 55, 4055–4058.
- 32.
Wang, Z.; Ju, Y.; Tong, S.; et al. Au 3 Cu tetrapod nanocrystals: Highly efficient and metabolizable multimodality imaging-guided NIR-II photothermal agents. Nanoscale Horiz. 2018, 3, 624–631.
- 33.
Gao, F.; He, G.; Yin, H.; et al. Titania-coated 2D gold nanoplates as nanoagents for synergistic photothermal/sonodynamic therapy in the second near-infrared window. Nanoscale 2019, 11, 2374–2384.
- 34.
Song, C.; Li, F.; Guo, X.; et al. Gold nanostars for cancer cell-targeted SERS-imaging and NIR light-triggered plasmonic photothermal therapy (PPTT) in the first and second biological windows. J. Mater. Chem. B 2019, 7, 2001–2008.
- 35.
Yang, H.; He, H.; Tong, Z.; et al. The impact of size and surface ligand of gold nanorods on liver cancer accumulation and photothermal therapy in the second near-infrared window. J. Colloid Interface Sci. 2020, 565, 186–196.
- 36.
Wang, J.; Sun, J.; Wang, Y.; et al. Gold nanoframeworks with mesopores for Raman–photoacoustic imaging and photo-chemo tumor therapy in the second near-infrared biowindow. Adv. Funct. Mater. 2020, 30, 1908825.
- 37.
Liu, H.; Chen, D.; Li, L.; et al. Multifunctional gold nanoshells on silica nanorattles: A platform for the combination of photothermal therapy and chemotherapy with low systemic toxicity. Angew. Chem. 2011, 123, 921–925.
- 38.
Wu, Z.; Yao, Q.; Chai, O.J.H.; et al. Unraveling the impact of gold (I)–thiolate motifs on the aggregation-induced emission of gold nanoclusters. Angew. Chem. 2020, 132, 10020–10025.
- 39.
Yang, Y.; Yu, Y.; Chen, H.; et al. Illuminating platinum transportation while maximizing therapeutic efficacy by gold nanoclusters via simultaneous near-infrared-I/II imaging and glutathione scavenging. ACS Nano 2020, 14, 13536–13547.
- 40.
Ding, X.; Liow, C.H.; Zhang, M.; et al. Surface plasmon resonance enhanced light absorption and photothermal therapy in the second near-infrared window. J. Am. Chem. Soc. 2014, 136, 15684–15693.
- 41.
Ferrando, R.; Jellinek, J.; Johnston, R.L. Nanoalloys: From theory to applications of alloy clusters and nanoparticles. Chem. Rev. 2008, 108, 845–910.
- 42.
Andolina, C.M.; Dewar, A.C.; Smith, A.M.; et al. Photoluminescent gold–copper nanoparticle alloys with composition-tunable near-infrared emission. J. Am. Chem. Soc. 2013, 135, 5266–5269.
- 43.
Marbella, L.E.; Andolina, C.M.; Smith, A.M.; et al. Gold-cobalt nanoparticle alloys exhibiting tunable compositions, near-infrared emission, and high T2 relaxivity. Adv. Funct. Mater. 2014, 24, 6532–6539.
- 44.
Wang, G.; Guo, R.; Kalyuzhny, G.; et al. NIR luminescence intensities increase linearly with proportion of polar thiolate ligands in protecting monolayers of Au38 and Au140 quantum dots. J. Phys. Chem. B 2006, 110, 20282–20289.
- 45.
Zheng, J.; Zhang, C.; Dickson, R.M. Highly fluorescent, water-soluble, size-tunable gold quantum dots. Phys. Rev. Lett. 2004, 93, 077402.
- 46.
Zheng, J.; Zhou, C.; Yu, M.; et al. Different sized luminescent gold nanoparticles. Nanoscale 2012, 4, 4073–4083.
- 47.
Christensen, A.; Ruban, A.; Stoltze, P.; et al. Phase diagrams for surface alloys. Phys. Rev. B 1997, 56, 5822.
- 48.
Crespo, O.; Gimeno, M.C.; Laguna, A.; et al. Highly luminescent gold (I)–silver (I) and gold (I)–copper (I) chalcogenide clusters. Chem. Eur. J. 2007, 13, 235–246.
- 49.
Xie, H.; Li, Z.; Sun, Z.; et al. Metabolizable ultrathin Bi2Se3 nanosheets in imaging-guided photothermal therapy. Small 2016, 12, 4136–4145.
- 50.
Xing, C.; Jing, G.; Liang, X.; et al. Graphene oxide/black phosphorus nanoflake aerogels with robust thermo-stability and significantly enhanced photothermal properties in air. Nanoscale 2017, 9, 8096–8101.
- 51.
Geng, B.; Shen, W.; Li, P.; et al. Carbon dot-passivated black phosphorus nanosheet hybrids for synergistic cancer therapy in the NIR-II window. ACS Appl. Mater. Interfaces 2019, 11, 44949–44960.
- 52.
Lin, H.; Gao, S.; Dai, C.; et al. A two-dimensional biodegradable niobium carbide (MXene) for photothermal tumor eradication in NIR-I and NIR-II biowindows. J. Am. Chem. Soc. 2017, 139, 16235–16247.
- 53.
Lin, S.; Lin, H.; Yang, M.; et al. A two-dimensional MXene potentiates a therapeutic microneedle patch for photonic implantable medicine in the second NIR biowindow. Nanoscale 2020, 12, 10265–10276.
- 54.
Zhou, Z.; Li, B.; Shen, C.; et al. Metallic 1T phase enabling MoS2 nanodots as an efficient agent for photoacoustic imaging guided photothermal therapy in the near-infrared-ii window. Small 2020, 16, 2004173.
- 55.
Cheng, Y.; Yang, F.; Xiang, G.; et al. Ultrathin tellurium oxide/ammonium tungsten bronze nanoribbon for multimodality imaging and second near-infrared region photothermal therapy. Nano Lett. 2019, 19, 1179–1189.
- 56.
Yang, C.; Chan, K.K.; Xu, G.; et al. Biodegradable polymer-coated multifunctional graphene quantum dots for light-triggered synergetic therapy of pancreatic cancer. ACS Appl. Mater. Interfaces 2018, 11, 2768–2781.
- 57.
Cui, X.; Li, M.; Wei, F.; et al. Biomimetic light-activatable graphene-based nanoarchitecture for synergistic chemophotothermal therapy. Chem. Eng. J. 2021, 420, 127710.
- 58.
Xu, Z.; Zhang, Y.; Zhou, W.; et al. NIR-II-activated biocompatible hollow nanocarbons for cancer photothermal therapy. J. Nanobiotechnol. 2021, 19, 137.
- 59.
O’Shea, D.; Hodgkinson, T.; Dixon, J.; et al. Development of an ‘Off-the-Shelf’Gene therapeutic nanoparticle formulation for incorporation into biomaterials for regenerative medicine applications. Eur. Cell Mater. 2024, 47, 152–169.
- 60.
O’connell, M.J.; Bachilo, S.M.; Huffman, C.B.; et al. Band gap fluorescence from individual single-walled carbon nanotubes. Science 2002, 297, 593–596.
- 61.
Robinson, J.T.; Welsher, K.; Tabakman, S.M.; et al. High performance in vivo near-IR (>1 μm) imaging and photothermal cancer therapy with carbon nanotubes. Nano Res. 2010, 3, 779–793.
- 62.
Takeuchi, T.; Iizumi, Y.; Yudasaka, M.; et al. Characterization and biodistribution analysis of oxygen-doped single-walled carbon nanotubes used as in vivo fluorescence imaging probes. Bioconjug. Chem. 2019, 30, 1323–1330.
- 63.
Yi, H.; Ghosh, D.; Ham, M.-H.; et al. M13 phage-functionalized single-walled carbon nanotubes as nanoprobes for second near-infrared window fluorescence imaging of targeted tumors. Nano Lett. 2012, 12, 1176–1183.
- 64.
Murakami, T.; Nakatsuji, H.; Inada, M.; et al. Photodynamic and photothermal effects of semiconducting and metallic-enriched single-walled carbon nanotubes. J. Am. Chem. Soc. 2012, 134, 17862–17865.
- 65.
Hong, G.; Diao, S.; Chang, J.; et al. Through-skull fluorescence imaging of the brain in a new near-infrared window. Nat. Photonics 2014, 8, 723–730.
- 66.
Hong, G.; Lee, J.C.; Robinson, J.T.; et al. Multifunctional in vivo vascular imaging using near-infrared II fluorescence. Nat. Med. 2012, 18, 1841–1846.
- 67.
Welsher, K.; Sherlock, S.P.; Dai, H. Deep-tissue anatomical imaging of mice using carbon nanotube fluorophores in the second near-infrared window. Proc. Natl. Acad. Sci. USA 2011, 108, 8943–8948.
- 68.
Zheng, M.; Jagota, A.; Semke, E.D.; et al. DNA-assisted dispersion and separation of carbon nanotubes. Nat. Mater. 2003, 2, 338–342.
- 69.
Ghosh, D.; Bagley, A.F.; Na, Y.J.; et al. Deep, noninvasive imaging and surgical guidance of submillimeter tumors using targeted M13-stabilized single-walled carbon nanotubes. Proc. Natl. Acad. Sci. USA 2014, 111, 13948–13953.
- 70.
Chen, J.; Wang, M.; Xi, B.; et al. SPARC is a key regulator of proliferation, apoptosis and invasion in human ovarian cancer. PLoS ONE 2012, 7, e42413.
- 71.
Iizumi, Y.; Yudasaka, M.; Kim, J.; et al. Oxygen-doped carbon nanotubes for near-infrared fluorescent labels and imaging probes. Sci. Rep. 2018, 8, 6272.
- 72.
Liu, Z.; Davis, C.; Cai, W.; et al. Circulation and long-term fate of functionalized, biocompatible single-walled carbon nanotubes in mice probed by Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2008, 105, 1410–1415.
- 73.
Welsher, K.; Liu, Z.; Sherlock, S.P.; et al. A route to brightly fluorescent carbon nanotubes for near-infrared imaging in mice. Nat. Nanotechnol. 2009, 4, 773–780.
- 74.
Geng, B.; Shen, W.; Fang, F.; et al. Enriched graphitic N dopants of carbon dots as F cores mediate photothermal conversion in the NIR-II window with high efficiency. Carbon 2020, 162, 220–233.
- 75.
Wang, H.; Pan, X.; Wang, X.; et al. Degradable carbon–silica nanocomposite with immunoadjuvant property for dual-modality photothermal/photodynamic therapy. ACS Nano 2020, 14, 2847–2859.
- 76.
Sun, W.; Zhang, X.; Jia, H.R.; et al. Water-dispersible candle soot–derived carbon nano-onion clusters for imaging-guided photothermal cancer therapy. Small 2019, 15, 1804575.
- 77.
Lu, G.-H.; Shang, W.-T.; Deng, H.; et al. Targeting carbon nanotubes based on IGF-1R for photothermal therapy of orthotopic pancreatic cancer guided by optical imaging. Biomaterials 2019, 195, 13–22.
- 78.
Li, Y.; Bai, G.; Zeng, S.; et al. Theranostic carbon dots with innovative NIR-II emission for in vivo renal-excreted optical imaging and photothermal therapy. ACS Appl. Mater. Interfaces 2019, 11, 4737–4744.
- 79.
Permatasari, F.A.; Fukazawa, H.; Ogi, T.; et al. Design of pyrrolic-N-rich carbon dots with absorption in the first near-infrared window for photothermal therapy. ACS Appl. Nano Mater. 2018, 1, 2368–2375.
- 80.
Gong, P.; Guo, L.; Pang, M.; et al. Nano-sized paramagnetic and fluorescent fluorinated carbon fiber with high NIR absorbance for cancer chemo-photothermal therapy. J. Mater. Chem. B 2018, 6, 3068–3077.
- 81.
Geng, B.; Yang, D.; Pan, D.; et al. NIR-responsive carbon dots for efficient photothermal cancer therapy at low power densities. Carbon 2018, 134, 153–162.
- 82.
Thakur, M.; Kumawat, M.K.; Srivastava, R. Multifunctional graphene quantum dots for combined photothermal and photodynamic therapy coupled with cancer cell tracking applications. RSC Adv. 2017, 7, 5251–5261.
- 83.
Cao, Y.; Li, S.; Chen, C.; et al. Rattle-type Au@Cu2−xS hollow mesoporous nanocrystals with enhanced photothermal efficiency for intracellular oncogenic microRNA detection and chemo-photothermal therapy. Biomaterials 2018, 158, 23–33.
- 84.
Zhao, Y.; Song, Z. Phase transfer-based synthesis of highly stable, biocompatible and the second near-infrared-emitting silver sulfide quantum dots. Mater. Lett. 2014, 126, 78–80.
- 85.
Yang, H.-Y.; Zhao, Y.-W.; Zhang, Z.-Y.; et al. One-pot synthesis of water-dispersible Ag2S quantum dots with bright fluorescent emission in the second near-infrared window. Nanotechnol. 2013, 24, 055706.
- 86.
Matea, C.T.; Mocan, T.; Tabaran, F.; et al. Quantum dots in imaging, drug delivery and sensor applications. Int. J. Nanomed. 2017, 12, 5421–5431.
- 87.
Hong, G.; Robinson, J.T.; Zhang, Y.; et al. In vivo fluorescence imaging with Ag2S quantum dots in the second near-infrared region. Angew. Chem. Int. Ed. 2012, 51, 9818–9821.
- 88.
Zhang, Y.; Hong, G.; Zhang, Y.; et al. Ag2S quantum dot: A bright and biocompatible fluorescent nanoprobe in the second near-infrared window. ACS Nano 2012, 6, 3695–3702.
- 89.
Zhang, Y.; Zhang, Y.; Hong, G.; et al. Biodistribution, pharmacokinetics and toxicology of Ag2S near-infrared quantum dots in mice. Biomaterials 2013, 34, 3639–3646.
- 90.
Shen, S.; Zhang, Y.; Peng, L.; et al. Matchstick-Shaped Ag2S–ZnS Heteronanostructures Preserving both UV/Blue and Near-Infrared Photoluminescence. Angew. Chem. Int. Ed. 2011, 50, 7115–7118.
- 91.
Du, Y.; Xu, B.; Fu, T.; et al. Near-infrared photoluminescent Ag2S quantum dots from a single source precursor. J. Am. Chem. Soc. 2010, 132, 1470–1471.
- 92.
Gao, X.; Cui, Y.; Levenson, R.M.; et al. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol. 2004, 22, 969–976.
- 93.
Mahesh, M. The essential physics of medical imaging. Med. Phys. 2013, 40, 077301.
- 94.
Guo, W.; Qiu, Z.; Guo, C.; et al. Multifunctional theranostic agent of Cu2(OH)PO4 quantum dots for photoacoustic image-guided photothermal/photodynamic combination cancer therapy. ACS Appl. Mater. Interfaces 2017, 9, 9348–9358.
- 95.
Liu, H.; Li, C.; Qian, Y.; et al. Magnetic-induced graphene quantum dots for imaging-guided photothermal therapy in the second near-infrared window. Biomaterials 2020, 232, 119700.
- 96.
Li, S.; Deng, Q.; Zhang, Y.; et al. Rational design of conjugated small molecules for superior photothermal theranostics in the NIR-II biowindow. Adv. Mater. 2020, 32, 2001146.
- 97.
Liu, Z.; Qiu, K.; Liao, X.; et al. Nucleus-targeting ultrasmall ruthenium (iv) oxide nanoparticles for photoacoustic imaging and low-temperature photothermal therapy in the NIR-II window. Chem. Commun. 2020, 56, 3019–3022.
- 98.
Guo, B.; Sheng, Z.; Hu, D.; et al. Through scalp and skull NIR-II photothermal therapy of deep orthotopic brain tumors with precise photoacoustic imaging guidance. Adv. Mater. 2018, 30, 1802591.
- 99.
Cao, Z.; Feng, L.; Zhang, G.; et al. Semiconducting polymer-based nanoparticles with strong absorbance in NIR-II window for in vivo photothermal therapy and photoacoustic imaging. Biomaterials 2018, 155, 103–111.
- 100.
Jiang, Y.; Li, J.; Zhen, X.; et al. Dual-peak absorbing semiconducting copolymer nanoparticles for first and second near-infrared window photothermal therapy: A comparative study. Adv. Mater. 2018, 30, 1705980.
- 101.
Cao, Y.; Dou, J.-H.; Zhao, N.-J.; et al. Highly efficient NIR-II photothermal conversion based on an organic conjugated polymer. Chem. Mater. 2017, 29, 718–725.
- 102.
Zhou, B.; Hu, Z.; Jiang, Y.; et al. Theoretical exploitation of acceptors based on benzobis (thiadiazole) and derivatives for organic NIR-II fluorophores. Phys. Chem. Chem. Phys. 2018, 20, 19759–19767.
- 103.
Antaris, A.L.; Chen, H.; Cheng, K.; et al. A small-molecule dye for NIR-II imaging. Nat. Mater. 2016, 15, 235–242.
- 104.
Fang, Y.; Shang, J.; Liu, D.; et al. Design, synthesis, and application of a small molecular NIR-II fluorophore with maximal emission beyond 1200 nm. J. Am. Chem. Soc. 2020, 142, 15271–15275.
- 105.
Schnermann, M.J. Organic dyes for deep bioimaging. Nat. 2017, 551, 176–177.
- 106.
Sun, C.; Li, B.; Zhao, M.; et al. J-aggregates of cyanine dye for NIR-II in vivo dynamic vascular imaging beyond 1500 nm. J. Am. Chem. Soc. 2019, 141, 19221–19225.
- 107.
Ren, T.B.; Wang, Z.Y.; Xiang, Z.; et al. A general strategy for development of activatable NIR-II fluorescent probes for in vivo high-contrast bioimaging. Angew. Chem. 2021, 133, 813–818.
- 108.
Yao, D.; Wang, Y.; Zou, R.; et al. Molecular engineered squaraine nanoprobe for NIR-II/photoacoustic imaging and photothermal therapy of metastatic breast cancer. ACS Appl. Mater. Interfaces 2020, 12, 4276–4284.
- 109.
Zeng, X.; Xiao, Y.; Lin, J.; et al. Near-infrared II dye-protein complex for biomedical imaging and imaging-guided photothermal therapy. Adv. Healthc. Mater. 2018, 7, 1800589.
- 110.
Zhou, H.; Zeng, X.; Li, A.; et al. Upconversion NIR-II fluorophores for mitochondria-targeted cancer imaging and photothermal therapy. Nat. Commun. 2020, 11, 6183.
- 111.
Wang, X.; Cheng, L. Multifunctional two-dimensional nanocomposites for photothermal-based combined cancer therapy. Nanoscale 2019, 11, 15685–15708.
- 112.
Sun, J.; Cai, W.; Sun, Y.; et al. Facile synthesis of melanin-dye nanoagent for NIR-II fluorescence/photoacoustic imaging-guided photothermal therapy. Int. J. Nanomed. 2020, 15, 10199–10213.
- 113.
Chen, S.; Sun, B.; Miao, H.; et al. NIR-II dye-based multifunctional telechelic glycopolymers for NIR-IIa fluorescence imaging-guided stimuli-responsive chemo-photothermal combination therapy. ACS Mater. Lett. 2020, 2, 174–183.
- 114.
Xu, Y.; Zhang, Y.; Li, J.; et al. NIR-II emissive multifunctional AIEgen with single laser-activated synergistic photodynamic/photothermal therapy of cancers and pathogens. Biomaterials 2020, 259, 120315.
- 115.
Zhang, J.; Wang, N.; Ji, X.; et al. BODIPY-based fluorescent probes for biothiols. Chem. Eur. J. 2020, 26, 4172–4192.
- 116.
Bai, L.; Sun, P.; Liu, Y.; et al. Novel aza-BODIPY based small molecular NIR-II fluorophores for in vivo imaging. Chem. Commun. 2019, 55, 10920–10923.
- 117.
Godard, A.; Kalot, G.; Pliquett, J.; et al. Water-soluble Aza-BODIPYs: Biocompatible organic dyes for high contrast in vivo NIR-II imaging. Bioconjug. Chem. 2020, 31, 1088–1092.
- 118.
Sinkeldam, R.W.; Greco, N.J.; Tor, Y. Fluorescent analogs of biomolecular building blocks: Design, properties, and applications. Chem. Rev. 2010, 110, 2579–2619.
- 119.
Kobayashi, H.; Ogawa, M.; Alford, R.; et al. New strategies for fluorescent probe design in medical diagnostic imaging. Chem. Rev. 2010, 110, 2620–2640.
- 120.
Willmann, J.K.; Van Bruggen, N.; Dinkelborg, L.M.; et al. Molecular imaging in drug development. Nat. Rev. Drug Discov. 2008, 7, 591–607.
- 121.
Thekkek, N.; Richards-Kortum, R. Optical imaging for cervical cancer detection: Solutions for a continuing global problem. Nat. Rev. Cancer 2008, 8, 725–731.
- 122.
Ma, Q.; Su, X. Near-infrared quantum dots: Synthesis, functionalization and analytical applications. Analyst 2010, 135, 1867–1877.
- 123.
Lavis, L.D.; Raines, R.T. Bright ideas for chemical biology. ACS Chem. Biol. 2008, 3, 142–155.
- 124.
Mishra, A.; Behera, R.K.; Behera, P.K.; et al. Cyanines during the 1990s: A review. Chem. Rev. 2000, 100, 1973–2012.
- 125.
Li, B.; Lu, L.; Zhao, M.; et al. An efficient 1064 nm NIR-II excitation fluorescent molecular dye for deep-tissue high-resolution dynamic bioimaging. Angew. Chem. Int. Ed. 2018, 57, 7483–7487.
- 126.
Li, T.; Li, C.; Ruan, Z.; et al. Polypeptide-conjugated second near-infrared organic fluorophore for image-guided photothermal therapy. Acs Nano 2019, 13, 3691–3702.
- 127.
Starosolski, Z.; Bhavane, R.; Ghaghada, K.B.; et al. Indocyanine green fluorescence in second near-infrared (NIR-II) window. PLoS ONE 2017, 12, e0187563.
- 128.
Meng, X.; Zhang, J.; Sun, Z.; et al. Hypoxia-triggered single molecule probe for high-contrast NIR II/PA tumor imaging and robust photothermal therapy. Theranostics 2018, 8, 6025.
- 129.
Buzea, C.; Pacheco, I.I.; Robbie, K. Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases 2007, 2, MR17–MR71.
- 130.
Oliveira, M.I.; Santos, S.G.; Oliveira, M.J.; et al. Chitosan drives anti-inflammatory macrophage polarisation and pro-inflammatory dendritic cell stimulation. Eur. Cell Mater. 2012, 24, 133–136.
- 131.
Tomić, S.; Rudolf, R.; Brunčko, M.; et al. Response of monocyte-derived dendritic cells to rapidly solidified nickel-titanium ribbons with shape memory properties. Eur. Cell Mater. 2012, 23, 58–80.
- 132.
Lazareva, P.I.; Stupin, V.A.; Lazarev, K.A.; et al. Biodistribution and toxicological impact assessment of cerium dioxide nanoparticles in murine models: A systematic review of in vivo and ex vivo studies. Pharmaceutics 2025, 17, 1475.
- 133.
Zhang, X.-D.; Wu, D.; Shen, X.; et al. In vivo renal clearance, biodistribution, toxicity of gold nanoclusters. Biomaterials 2012, 33, 4628–4638.
- 134.
Luo, R.; Zhang, C.; Zhang, Z.; et al. NIR-II upconversion nanomaterials for biomedical applications. Nanoscale 2025, 17, 2985–3002.
- 135.
Yang, S.; Li, N.; Xiao, H.; et al. Clearance pathways of near-infrared-II contrast agents. Theranostics 2022, 12, 7853.
- 136.
Thangudu, S.; Su, C.-H. Review of light activated antibacterial nanomaterials in the second biological window. J. Nanobiotechnology 2025, 23, 293.
- 137.
Zhang, X.D.; Chen, J.; Min, Y.; et al. Metabolizable Bi2Se3 nanoplates: Biodistribution, toxicity, and uses for cancer radiation therapy and imaging. Adv. Funct. Mater. 2014, 24, 1718–1729.
- 138.
Choi, H.S.; Liu, W.; Misra, P.; et al. Renal clearance of nanoparticles. Nat. Biotechnol. 2007, 25, 1165.
- 139.
Li, X.; Wang, B.; Zhou, S.; et al. Surface chemistry governs the sub-organ transfer, clearance and toxicity of functional gold nanoparticles in the liver and kidney. J. Nanobiotechnol. 2020, 18, 45.
- 140.
Yu, M.; Zheng, J. Clearance pathways and tumor targeting of imaging nanoparticles. ACS Nano 2015, 9, 6655–6674.
- 141.
Yu, S.; Xia, G.; Yang, N.; et al. Noble metal nanoparticle-based photothermal therapy: Development and application in effective cancer therapy. Int. J. Mol. Sci. 2024, 25, 5632.
- 142.
Zhang, L.; Niu, G.; Lu, N.; et al. Continuous and scalable production of well-controlled noble-metal nanocrystals in milliliter-sized droplet reactors. Nano Lett. 2014, 14, 6626–6631.
- 143.
Hua, S.; De Matos, M.B.; Metselaar, J.M.; et al. Current trends and challenges in the clinical translation of nanoparticulate nanomedicines: Pathways for translational development and commercialization. Front. Pharmacol. 2018, 9, 790.
- 144.
Zhang, L.; Yang, F.; Liu, J.; et al. Exosomes in tissue engineering and cell-free therapy: A comprehensive review. Eur. Cell Mater. 2026, 55, 80–105.
- 145.
Yang, C.; Lou, Y.; Bai, M.; et al. Advances in metal-based nanomedicine: From basic science to clinical implications. Chin. Chem. Lett. 2025, in press.
- 146.
Si, S.; Wu, K.; Zhang, X.; et al. Research progress of nanoparticles in the diagnosis and treatment of renal ischemia-reperfusion injury. J. Nanobiotechnol. 2026, 24, 201.
- 147.
Hulsart-Billström, G.; Dawson, J.; Hofmann, S.; et al. A surprisingly poor correlation between in vitro and in vivo testing of biomaterials for bone regeneration: Results of a multicentre analysis. Eur. Cell Mater. 2016, 31, 312–322.
- 148.
Shahalaei, M.; Azad, A.K.; Sulaiman, W.M.A.W.; et al. A review of metallic nanoparticles: Present issues and prospects focused on the preparation methods, characterization techniques, and their theranostic applications. Front. Chem. 2024, 12, 1398979.

This work is licensed under a Creative Commons Attribution 4.0 International License.


