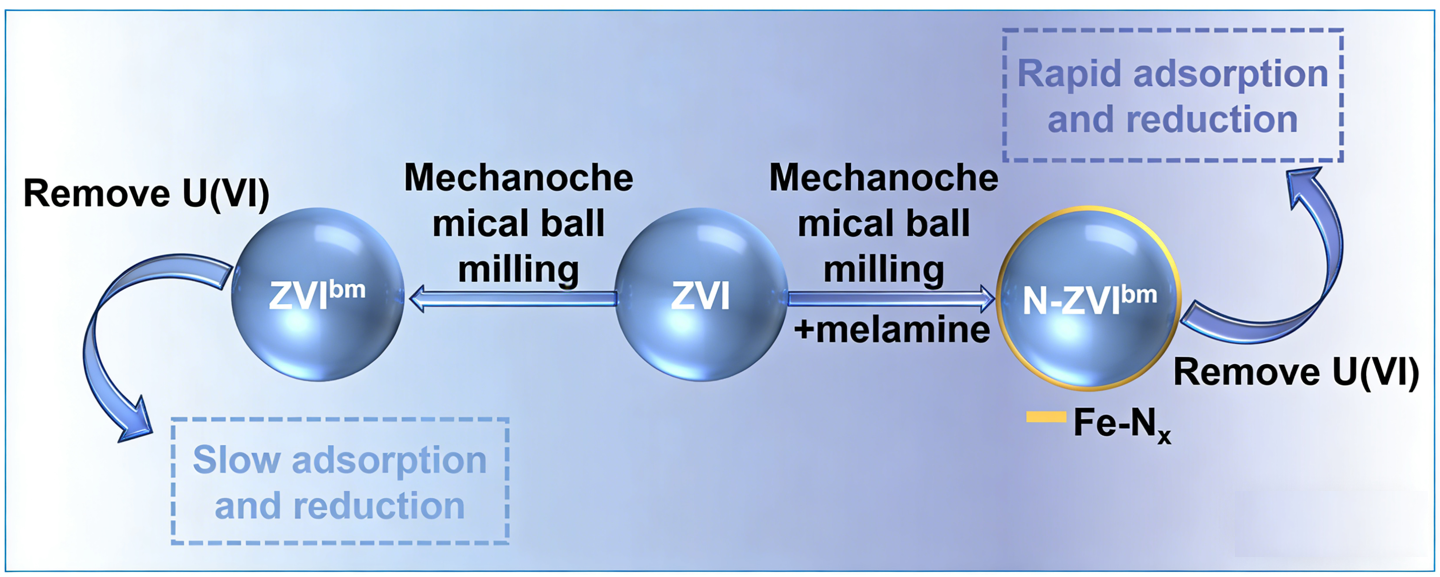
The remediation of uranium (U(VI)) contamination in water is of critical importance due to its chemical toxicity, long-term radiotoxicity, and environmental mobility. This study employed a green and efficient mechanical ball-milling method, using melamine as a nitrogen source, to successfully synthesize a nitrogen-modified zerovalent iron material (N-ZVIbm). Systematic characterization, revealed that the ball-milling process successfully constructed Fe–Nx coordination structures on the ZVI surface. This structure optimized the electronic distribution of zerovalent iron and enhanced its surface reactivity. Batch experiments demonstrated that the U(VI) removal by N-ZVIbm follows pseudo-first-order kinetics, exhibiting a rate constant as high as 0.288 h−1, which is 3.7 times that of unmodified ball-milled zerovalent iron (ZVIbm). Mechanistic studies revealed that the Fe–Nx sites not only promoted the corrosion of ZVI, accelerating the formation of adsorptive iron(III) (oxyhydr) oxides, but also functioned as highly efficient adsorption and electron transfer sites, synergistically enhancing both the adsorption and reduction of U(VI). Post-reaction solid analysis indicated a U(IV) content of up to 68%, markedly exceeding that in the control system (29%), confirming the efficacy of nitrogen modification in enhancing U(VI) reduction and immobilization. This work not only establishes a novel green pathway for synthesizing high-performance zerovalent iron materials but also deepens the understanding of the mechanisms of Fe–Nx structures in radionuclide remediation.
- Open Access
- Article
Enhanced Removal of Uranium(VI) by Nitrogen-Modified Zerovalent Iron Synthesized via Mechanochemically Ball Milling
- Lianjie Ma 1,2,
- Peng Fan 2,*,
- Kunming Hou 2,
- Zhengwu Liu 3,
- Meng Du 2,
- Chen Tian 2,
- Zhenye Liang 3,
- Bing Nan 2,
- Luozhen Jiang 2,
- Lingling Guo 2,
- Yulin Qin 2,
- Zhuofan Zhang 2,
- Jisong Mi 2,
- Yingpeng Xie 1,*,
- Lina Li 2,3,*
Author Information
Received: 15 Jan 2026 | Revised: 17 Mar 2026 | Accepted: 18 Mar 2026 | Published: 30 Mar 2026
Abstract
Graphical Abstract
Keywords
zerovalent iron | ball milling | uranium removal | X-ray absorption fine structure | wastewater remediation
References
- 1.
An, R.; Li, X.; Xie, Y. Post-Fukushima Innovation: Establishing a Regional Marine Environmental Cooperation Mechanism in Northeast Asia with Global Implications. Front. Mar. Sci. 2025, 12, 1578912.
- 2.
Deng, S.; Yang, Y.; Han, X.; et al. Unlocking the Potential of Surface Modification with Phosphate on Ball Milled Zero-Valent Iron Reactivity: Implications for Radioactive Metal Ions Removal. Water Res. 2024, 260, 121912.
- 3.
Lin, T.; Chen, T.; Jiao, C.; et al. Ion Pair Sites for Efficient Electrochemical Extraction of Uranium in Real Nuclear Wastewater. Nat. Commun. 2024, 15, 4149.
- 4.
Nayak, T.; Mukherjee, S.; Kini, A.R.; et al. Cellulose-Derived Nanomaterials for Affordable and Rapid Remediation of Uranium in Water. ACS Sustain. Chem. Eng. 2025, 13, 1838–1850.
- 5.
Liu, X.; Xie, Y.; Hao, M.; et al. Secondary Metal Ion-Induced Electrochemical Reduction of U(VI) to U(IV) Solids. Nat. Commun. 2024, 15, 7736.
- 6.
Zhou, L.; Li, Y.; Shao, Y.; et al. Interface Coupling Induced Built-In Electric Fields Accelerate Electro-Assisted Uranium Extraction over Co3O4@FeOx Nanosheet Arrays. Appl. Catal. B 2024, 353, 124052.
- 7.
Fan, P.; Li, L.; Sun, Y.; et al. Selenate Removal by Fe0 Coupled with Ferrous Iron, Hydrogen Peroxide, Sulfidation, and Weak Magnetic Field: A Comparative Study. Water Res. 2019, 159, 375–384.
- 8.
Chen, J.; Feng, Y.; Liu, Y.; et al. MoS2 Modification to Enhance the Removal of Various Chlorinated Hydrocarbons by Zerovalent Iron. Environ. Sci. Technol. 2025, 59, 11330–11340.
- 9.
Korpayev, S.; Kavaklı, C.; Kavaklı, P.A. Nanoscale Zerovalent Iron Immobilized on Functionalized Nonwoven Cotton Fabric for As(V) Adsorption. Water Air Soil Pollut. 2021, 232, 131.
- 10.
Fan, P.; Guan, X.; Wei, G.; et al. Simply Closing the Reactor Improves the Electron Efficiency of Zerovalent Iron toward Various Metal(loid)s Removal. Water Environ. Res. 2021, 93, 1829–1836.
- 11.
Li, J.; Zhang, X.; Liu, M.; et al. Enhanced Reactivity and Electron Selectivity of Sulfidated Zerovalent Iron toward Chromate under Aerobic Conditions. Environ. Sci. Technol. 2018, 52, 2988–2997.
- 12.
Zhang, H.; Liu, X.; Zhou, B.; et al. Phosphorylated Zerovalent Iron Boosts Active Hydrogen Species Generation from Water Dissociation for Superior Hg(II) Reduction. Water Res. 2025, 283, 123787.
- 13.
Yin, Y.; Shen, C.; Liu, Y.; et al. Use of Hollow Mesoporous Silica Spheres as Vehicles for Delivery of Nanoscale Zerovalent Iron in Porous Media. Microporous Mesoporous Mater. 2023, 362, 112755.
- 14.
Vasarevičius, S.; Danila, V.; Januševičius, T. Immobilisation of Cadmium, Copper, Lead, and Nickel in Soil Using Nano Zerovalent Iron Particles: Ageing Effect on Heavy Metal Retention. Water Air Soil Pollut. 2020, 231, 496.
- 15.
He, C.-S.; Ding, R.-R.; Zhou, G.-N.; et al. Coexistence of Humic Acid Enhances the Reductive Removal of Diatrizoate via Depassivating Zero-Valent Iron under Aerobic Conditions. J. Mater. Chem. A 2020, 8, 14634–14643.
- 16.
Gao, P.; Fan, X.; Sun, D.; et al. Recent Advances in Ball-Milled Materials and Their Applications for Adsorptive Removal of Aqueous Pollutants. Water 2024, 16, 1639.
- 17.
Wei, K.; Li, H.; Gu, H.; et al. Strained Zero-Valent Iron for Highly Efficient Heavy Metal Removal. Adv. Funct. Mater. 2022, 32, 2200498.
- 18.
Matheson, L.J.; Tratnyek, P.G. Reductive Dehalogenation of Chlorinated Methanes by Iron Metal. Environ. Sci. Technol. 1994, 28, 2045–2053.
- 19.
Bharti; Khurana, I.; Shaw, A.K.; et al. Removal of Trinitrotoluene with Nano Zerovalent Iron Impregnated Graphene Oxide. Water Air Soil Pollut. 2017, 229, 17.
- 20.
O’Carroll, D.; Sleep, B.; Krol, M.; et al. Nanoscale Zero Valent Iron and Bimetallic Particles for Contaminated Site Remediation. Adv. Water Resour. 2013, 51, 104–122.
- 21.
Li, M.; Mu, Y.; Shang, H.; et al. Phosphate Modification Enables High Efficiency and Electron Selectivity of nZVI toward Cr(VI) Removal. Appl. Catal. B 2020, 263, 118364.
- 22.
Zhu, X.; Han, B.; Feng, Q. Common Anions Affected Removal of Carbon Tetrachloride in Groundwater Using Granular Sponge Zerovalent Iron. Water Air Soil Pollut. 2020, 231, 138.
- 23.
Li, J.; Zhang, X.; Sun, Y.; et al. Advances in Sulfidation of Zerovalent Iron for Water Decontamination. Environ. Sci. Technol. 2017, 51, 13533–13544.
- 24.
Fan, P.; Sun, Y.; Lei, H.; et al. Mechanochemically Ball-Milled Zerovalent Iron and Ferrous Composite for Effective Removal of Various Metal(loid)s from Water. Chem. Eng. J. 2023, 452, 139380.
- 25.
Jones, A.C.; Leitch, J.A.; Raby-Buck, S.E.; et al. Mechanochemical Techniques for the Activation and Use of Zero-Valent Metals in Synthesis. Nat. Synth. 2022, 1, 763–775.
- 26.
Shi, Y.; Wang, D.; Gao, F.; et al. Mechanochemical Nitridation of Micron Zero-Valent Iron for Enhanced Dechlorination of Trichloroethylene: Mechanistic Insights into Nitrogen Sources and Ball Milling Conditions. Sep. Purif. Technol. 2024, 337, 126381.
- 27.
Liu, G.-h.; Zhu, S.-N.; Ye, Z. Reduction in the Acute Toxicity of Explosive Wastewater Containing Toxic Nitroaromatic Compounds by a Nanoscale Zerovalent Iron Pretreatment Process. Water Air Soil Pollut. 2012, 223, 5049–5055.
- 28.
He, M.-F.; Li, W.-Q.; Xie, Z.-H.; et al. Peracetic Acid Activation by Mechanochemically Sulfidated Zero Valent Iron for Micropollutants Degradation: Enhancement Mechanism and Strategy for Extending Applicability. Water Res. 2022, 222, 118887.
- 29.
Tan, X.; Li, H.; Zhang, W.; et al. Square-Pyramidal Fe–N4 with Defect-Modulated O-Coordination: Two-Tier Electronic Structure Fine-Tuning for Enhanced Oxygen Reduction. Chem Catal. 2022, 2, 816–835.
- 30.
Gong, L.; Qiu, X.; Tratnyek, P.G.; et al. FeNX(C)-Coated Microscale Zero-Valent Iron for Fast and Stable Trichloroethylene Dechlorination in Both Acidic and Basic pH Conditions. Environ. Sci. Technol. 2021, 55, 5393–5402.
- 31.
Meng, F.; Xu, J.; Dai, H.; et al. Even Incorporation of Nitrogen into Fe0 Nanoparticles as Crystalline Fe4N for Efficient and Selective Trichloroethylene Degradation. Environ. Sci. Technol. 2022, 56, 4489–4497.
- 32.
Zhang, L.; Sun, Y.; Ge, R.; et al. Mechanical Insight into Direct Singlet Oxygen Generation Pathway: Pivotal Role of FeN4 Sites and Selective Organic Contaminants Removal. Appl. Catal. B 2023, 339, 123130.
- 33.
Fan, P.; Sun, Y.; Zhou, B.; et al. Coupled Effect of Sulfidation and Ferrous Dosing on Selenate Removal by Zerovalent Iron Under Aerobic Conditions. Environ. Sci. Technol. 2019, 53, 14577–14585.
- 34.
Gong, L.; Qiu, X.; Cheng, D.; et al. Coincorporation of N and S into Zero-Valent Iron to Enhance TCE Dechlorination: Kinetics, Electron Efficiency, and Dechlorination Capacity. Environ. Sci. Technol. 2021, 55, 16088–16098.
- 35.
Li, X.; Zhang, X.; Zhang, P.; et al. Incorporation of N-Doped Biochar into Zero-Valent Iron for Efficient Reductive Degradation of Neonicotinoids: Mechanism and Performance. Biochar 2023, 5, 78.
- 36.
Smit, E.; Manoun, B.; Waal, D. Low-Wavenumber Raman Spectra of the Spin-Transition Complexes [Fe(NH2 trz)3](ClO4)2 and [Fe(Htrz)3](ClO4)2. J. Raman Spectrosc. 2001, 32, 339–344.
- 37.
Zu, J.; Zhang, N.; Liu, X.; et al. Mechanochemical Thioglycolate Modification of Microscale Zero-Valent Iron for Superior Heavy Metal Removal. Angew. Chem. Int. Ed. 2025, 64, e202415051.
- 38.
Song, J.; Hou, N.; Liu, X.; et al. Directional Formation of Reactive Oxygen Species via a Non-Redox Catalysis Strategy That Bypasses Electron Transfer Process. Adv. Mater. 2024, 36, 2405832.
- 39.
Deng, S.; Liu, L.; Cagnetta, G.; et al. Mechanochemically Synthesized S-ZVIbm Composites for the Activation of Persulfate in the pH-Independent Degradation of Atrazine: Effects of Sulfur Dose and Ball-Milling Conditions. Chem. Eng. J. 2021, 423, 129789.
- 40.
Zitolo, A.; Goellner, V.; Armel, V.; et al. Identification of Catalytic Sites for Oxygen Reduction in Iron- and Nitrogen-Doped Graphene Materials. Nat. Mater. 2015, 14, 937–942.
- 41.
Li, Q.; Luo, L.; Xu, C.; et al. Palladium Enhanced Iron Active Site—An Efficient Dual-Atom Catalyst for Oxygen Electroreduction. Small 2023, 19, 2303321.
- 42.
Fan, P.; Wu, X.; Zeng, J.; et al. Resolve the Species-Specific Effects of Iron (Hydr)oxides on the Performance of Underlying Zerovalent Iron for Metalloid Removal: Identification of Their Key Properties. J. Hazard. Mater. 2024, 477, 135378.
- 43.
Qin, H.; Sun, Y.; Yang, H.; et al. Unexpected Effect of Buffer Solution on Removal of Selenite and Selenate by Zerovalent Iron. Chem. Eng. J. 2018, 334, 296–304.
- 44.
Liu, H.; Qian, T.; Zhao, D. Reductive Immobilization of Perrhenate in Soil and Groundwater Using Starch-Stabilized ZVI Nanoparticles. Chin. Sci. Bull. 2013, 58, 275–281.
- 45.
Boglaienko, D.; Emerson, H.P.; Katsenovich, Y.P.; et al. Comparative Analysis of ZVI Materials for Reductive Separation of 99Tc(VII) from Aqueous Waste Streams. J. Hazard. Mater. 2019, 380, 120836.
- 46.
Shang, Y.; Ding, Y.; Zhang, P.; et al. Pyrrolic N or Pyridinic N: The Active Center of N-Doped Carbon for CO2 Reduction. Chin. J. Catal. 2022, 43, 2405–2413.
- 47.
Ha, Y.; Fei, B.; Yan, X.; et al. Atomically Dispersed Co-Pyridinic N-C for Superior Oxygen Reduction Reaction. Adv. Energy Mater. 2020, 10, 2002592.
- 48.
Qin, M.; Chen, C.; Zhang, B.; et al. Ultrahigh Pyridinic/Pyrrolic N Enabling N/S Co-Doped Holey Graphene with Accelerated Kinetics for Alkali-Ion Batteries. Adv. Mater. 2024, 36, 2407570.
- 49.
Yang, S.-M.; Guo, L.-L.; Nan, B.; et al. Tender Energy Spectroscopy Beamline at the Shanghai Synchrotron Radiation Facility. Nucl. Sci. Tech. 2025, 37, 11.

This work is licensed under a Creative Commons Attribution 4.0 International License.


