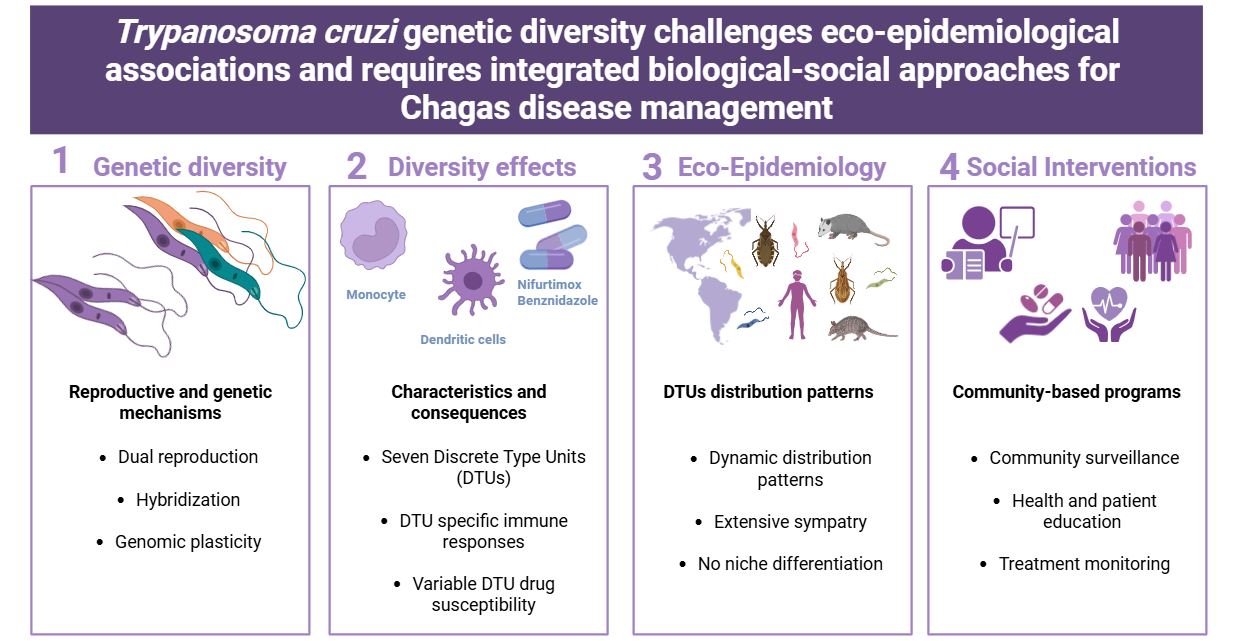
Trypanosoma cruzi, the etiological agent of Chagas disease, exhibits extensive genetic diversity with important implications for parasite biology, disease manifestation, and control strategies. This review synthesizes current knowledge on T. cruzi genetic diversity. It emphasizes how these findings inform both biological and community-based interventions. The parasite employs a dual reproductive strategy combining clonal propagation and sexual reproduction, with evidence of meiotic sex, hybridization, and genetic recombination in natural populations. Genomic plasticity is characterized by chromosomal aneuploidies, gene amplification, and extensive copy number variations, predominantly in gene family-rich regions including mucins, trans-sialidases, and mucin-associated surface proteins. T. cruzi is classified into seven discrete typing units: TcI–TcVI and TcBat. Although DTUs have been historically associated with specific epidemiological and ecological scenarios, recent comprehensive analyses demonstrated extensive sympatry among all clades across the Americas with no significant niche differences. This challenges assumptions about strong associations between parasite diversity and ecology which requires adaptive surveillance strategies. Different DTUs elicit distinct innate and adaptive immune responses, with variations in cytokine production, cell invasion rates, and surface antigen expression affecting disease outcomes and vaccine development approaches. Drug resistance patterns are influenced by both inter-DTUs and intra-DTU genetic variability, with no exclusive association between natural resistance and particular DTUs, highlighting the need for community-based treatment monitoring. Understanding these complex interactions is essential for developing effective therapeutic strategies, vaccines, and diagnostic tools. Future research should integrate social sciences approaches including health education programs, community surveillance, and sustainable control interventions tailored to local parasite populations.
- Open Access
- Review
Trypanosoma cruzi Genetic Diversity Challenges Eco-Epidemiological Associations and Requires Integrated Biological-Social Approaches for Chagas Disease Management
Author Information
Received: 28 Oct 2025 | Revised: 27 Jan 2026 | Accepted: 28 Jan 2026 | Published: 10 Apr 2026
Abstract
Graphical Abstract
Keywords
Trypanosoma cruzi | Chagas disease | DTU | immune response | drug resistance
References
- 1.
Tibayrenc, M.; Ayala, F.J. The Population Genetics of Trypanosoma cruzi Revisited in the Light of the Predominant Clonal Evolution Model. Acta Trop. 2015, 151, 156–165. https://doi.org/10.1016/j.actatropica.2015.05.006.
- 2.
Valadares, H.M.S.; Pimenta, J.R.; Segatto, M.; et al. Unequivocal Identification of Subpopulations in Putative Multiclonal Trypanosoma cruzi Strains by FACs Single Cell Sorting and Genotyping. PLoS Negl. Trop. Dis. 2012, 6, e1722. https://doi.org/10.1371/journal.pntd.0001722.
- 3.
Ramírez, J.D.; Guhl, F.; Messenger, L.A.; et al. Contemporary Cryptic Sexuality in Trypanosoma cruzi. Mol. Ecol. 2012, 21, 4216–4226. https://doi.org/10.1111/j.1365-294X.2012.05699.x.
- 4.
Llewellyn, M.S.; Rivett-Carnac, J.B.; Fitzpatrick, S.; et al. Extraordinary Trypanosoma cruzi Diversity within Single Mammalian Reservoir Hosts Implies a Mechanism of Diversifying Selection. Int. J. Parasitol. 2011, 41, 609–614. https://doi.org/10.1016/j.ijpara.2010.12.004.
- 5.
Messenger, L.A.; Miles, M.A. Evidence and Importance of Genetic Exchange among Field Populations of Trypanosoma cruzi. Acta Trop. 2015, 151, 150–155. https://doi.org/10.1016/j.actatropica.2015.05.007.
- 6.
Ocana-Mayorga, S.; Llewellyn, M.S.; Costales, J.A.; et al. Sex, Subdivision, and Domestic Dispersal of Trypanosoma cruzi Lineage I in Southern Ecuador. PLoS Negl. Trop. Dis. 2010, 4, e915. https://doi.org/10.1371/journal.pntd.0000915.
- 7.
Schwabl, P.; Imamura, H.; Van den Broeck, F.; et al. Meiotic Sex in Chagas Disease Parasite Trypanosoma cruzi. Nat. Commun. 2019, 10, 3972. https://doi.org/10.1038/s41467-019-11771-z.
- 8.
Jansen, A.M.; Xavier, S.C.C.; Roque, A.L.R. Landmarks of the Knowledge and Trypanosoma cruzi Biology in the Wild Environment. Front. Cell. Infect. Microbiol. 2020, 10, 10. https://doi.org/10.3389/fcimb.2020.00010.
- 9.
Lewis, M.D.; Llewellyn, M.S.; Gaunt, M.W.; et al. Flow Cytometric Analysis and Microsatellite Genotyping Reveal Extensive DNA Content Variation in Trypanosoma cruzi Populations and Expose Contrasts between Natural and Experimental Hybrids. Int. J. Parasitol. 2009, 39, 1305–1317. https://doi.org/10.1016/j.ijpara.2009.04.001.
- 10.
Matos, G.M.; Lewis, M.D.; Talavera-López, C.; et al. Microevolution of Trypanosoma cruzi Reveals Hybridization and Clonal Mechanisms Driving Rapid Genome Diversification. eLife 2022, 11, e75237. https://doi.org/10.7554/eLife.75237.
- 11.
Flores-López, C.A.; Machado, C.A. Analyses of 32 Loci Clarify Phylogenetic Relationships among Trypanosoma cruzi Lineages and Support a Single Hybridization Prior to Human Contact. PLoS Negl. Trop. Dis. 2011, 5, e1272. https://doi.org/10.1371/journal.pntd.0001272.
- 12.
Cortez, D.R.; Lima, F.M.; Reis-Cunha, J.L.; et al. Trypanosoma cruzi Genomic Variability: Array Comparative Genomic Hybridization Analysis of Clone and Parental Strain. Front. Cell. Infect. Microbiol. 2022, 12, 760830. https://doi.org/10.3389/fcimb.2022.760830.
- 13.
Zingales, B.; Bartholomeu, D.C. Trypanosoma cruzi Genetic Diversity: Impact on Transmission Cycles and Chagas Disease. Mem. Inst. Oswaldo Cruz 2022, 117, e210193. https://doi.org/10.1590/0074-02760210193.
- 14.
Alves, C.L.; Repolês, B.M.; da Silva, M.S.; et al. The Recombinase Rad51 Plays a Key Role in Events of Genetic Exchange in Trypanosoma cruzi. Sci. Rep. 2018, 8, 13335. https://doi.org/10.1038/s41598-018-31541-z.
- 15.
Ruvalcaba-Trejo, L.I.; Sturm, N.R. The Trypanosoma cruzi Sylvio X10 Strain Maxicircle Sequence: The Third Musketeer. BMC Genomics 2011, 12, 58. https://doi.org/10.1186/1471-2164-12-58.
- 16.
Rusman, F.; Floridia-Yapur, N.; Ragone, P.G.; et al. Evidence of Hybridization, Mitochondrial Introgression and Biparental Inheritance of the KDNA Minicircles in Trypanosoma cruzi I. PLoS Negl. Trop. Dis. 2020, 14, e0007770. https://doi.org/10.1371/journal.pntd.0007770.
- 17.
Barnabé, C.; Brenière, S.F. Scarce Events of Mitochondrial Introgression in Trypanosoma cruzi: New Case with a Bolivian Strain. Infect. Genet. Evol. 2012, 12, 1879–1883. https://doi.org/10.1016/j.meegid.2012.08.018.
- 18.
Messenger, L.A.; Garcia, L.; Vanhove, M.; et al. Ecological Host Fitting of Trypanosoma cruzi TcI in Bolivia: Mosaic Population Structure, Hybridization and a Role for Humans in Andean Parasite Dispersal. Mol. Ecol. 2015, 24, 2406–2422. https://doi.org/10.1111/mec.13186.
- 19.
Messenger, L.A.; Miles, M.A.; Bern, C. Between a Bug and a Hard Place: Trypanosoma cruzi Genetic Diversity and the Clinical Outcomes of Chagas Disease. Expert Rev. Anti. Infect. Ther. 2015, 13, 995–1029. https://doi.org/10.1586/14787210.2015.1056158.
- 20.
Minning, T.A.; Weatherly, D.B.; Flibotte, S.; et al. Widespread, Focal Copy Number Variations (CNV) and Whole Chromosome Aneuploidies in Trypanosoma cruzi Strains Revealed by Array Comparative Genomic Hybridization. BMC Genomics 2011, 12, 139. https://doi.org/10.1186/1471-2164-12-139.
- 21.
Reis-Cunha, J.L.; Rodrigues-Luiz, G.F.; Valdivia, H.O.; et al. Chromosomal Copy Number Variation Reveals Differential Levels of Genomic Plasticity in Distinct Trypanosoma cruzi Strains. BMC Genomics 2015, 16, 499. https://doi.org/10.1186/s12864-015-1680-4.
- 22.
Wang, W.; Peng, D.; Baptista, R.P.; et al. Strain-Specific Genome Evolution in Trypanosoma cruzi, the Agent of Chagas Disease. PLoS Pathog. 2021, 17, e1009254. https://doi.org/10.1371/journal.ppat.1009254.
- 23.
Cruz-Saavedra, L.; Schwabl, P.; Vallejo, G.A.; et al. Genome Plasticity Driven by Aneuploidy and Loss of Heterozygosity in Trypanosoma cruzi. Microb. Genom. 2022, 8, e000843. https://doi.org/10.1099/mgen.0.000843.
- 24.
Campo, V.; Di Noia, J.M.; Buscaglia, C.A.; et al. Differential Accumulation of Mutations Localized in Particular Domains of the Mucin Genes Expressed in the Vertebrate Host Stage of Trypanosoma cruzi. Mol. Biochem. Parasitol. 2004, 133, 81–91. https://doi.org/10.1016/j.molbiopara.2003.09.006.
- 25.
Jäger, A.V.; Muiá, R.P.; Campetella, O. Stage-Specific Expression of Trypanosoma cruzi Trans-Sialidase Involves Highly Conserved 3′ Untranslated Regions. FEMS Microbiol. Lett. 2008, 283, 182–188. https://doi.org/10.1111/j.1574-6968.2008.01170.x.
- 26.
Herreros-Cabello, A.; Callejas-Hernández, F.; Gironès, N.; et al. Trypanosoma cruzi Genome: Organization, Multi-Gene Families, Transcription, and Biological Implications. Genes 2020, 11, 1196. https://doi.org/10.3390/genes11101196.
- 27.
Clayton, C. Regulation of Gene Expression in Trypanosomatids: Living with Polycistronic Transcription. Open Biol. 2019, 9, 190072. https://doi.org/10.1098/rsob.190072.
- 28.
Reis-Cunha, J.L.; Pimenta-Carvalho, S.A.; Almeida, L.V.; et al. Ancestral Aneuploidy and Stable Chromosomal Duplication Resulting in Differential Genome Structure and Gene Expression Control in Trypanosomatid Parasites. Genome Res. 2024, 34, 441–453. https://doi.org/10.1101/gr.278550.123.
- 29.
Herreros-Cabello, A.; Callejas-Hernández, F.; Gironès, N.; et al. Trypanosoma cruzi: Genomic Diversity and Structure. Pathogens 2025, 14, 61. https://doi.org/10.3390/pathogens14010061.
- 30.
Ackermann, A.A.; Panunzi, L.G.; Cosentino, R.O.; et al. A Genomic Scale Map of Genetic Diversity in Trypanosoma cruzi. BMC Genomics 2012, 13, 736. https://doi.org/10.1186/1471-2164-13-736.
- 31.
Brenière, S.F.; Waleckx, E.; Barnabé, C. Over Six Thousand Trypanosoma cruzi Strains Classified into Discrete Typing Units (DTUs): Attempt at an Inventory. PLoS Negl. Trop. Dis. 2016, 10, e0004792. https://doi.org/10.1371/journal.pntd.0004792.
- 32.
Zingales, B.; Andrade, S.G.; Briones, M.R.S.; et al. A New Consensus for Trypanosoma cruzi Intraspecific Nomenclature: Second Revision Meeting Recommends TcI to TcVI. Mem. Inst. Oswaldo Cruz 2009, 104, 1051–1054. https://doi.org/10.1590/S0074-02762009000700021.
- 33.
Zingales, B.; Miles, M.A.; Campbell, D.A.; et al. The Revised Trypanosoma cruzi Subspecific Nomenclature: Rationale, Epidemiological Relevance and Research Applications. Infect. Genet. Evol. 2012, 12, 240–253. https://doi.org/10.1016/j.meegid.2011.12.009.
- 34.
Izeta-Alberdi, A.; Ibarra-Cerdeña, C.N.; Moo-Llanes, D.A.; et al. Geographical, Landscape and Host Associations of Trypanosoma cruzi DTUs and Lineages. Parasit. Vectors 2016, 9, 631. https://doi.org/10.1186/s13071-016-1918-2.
- 35.
Velásquez-Ortiz, N.; Herrera, G.; Hernández, C.; et al. Discrete Typing Units of Trypanosoma cruzi: Geographical and Biological Distribution in the Americas. Sci. Data 2022, 9, 360. https://doi.org/10.1038/s41597-022-01452-w.
- 36.
Maffey, L.; Cardinal, M.V.; Ordóñez-Krasnowski, P.C.; et al. Direct Molecular Identification of Trypanosoma cruzi Discrete Typing Units in Domestic and Peridomestic Triatoma infestans and Triatoma sordida from the Argentine Chaco. Parasitology 2012, 139, 1570–1579. https://doi.org/10.1017/S0031182012000856.
- 37.
Monje-Rumi, M.M.; Brandán, C.P.; Ragone, P.G.; et al. Trypanosoma cruzi Diversity in the Gran Chaco: Mixed Infections and Differential Host Distribution of TcV and TcVI. Infect. Genet. Evol. 2015, 29, 53–59. https://doi.org/10.1016/j.meegid.2014.11.001.
- 38.
Barros, J.H.S.; Xavier, S.C.C.; Bilac, D.; et al. Identification of Novel Mammalian Hosts and Brazilian Biome Geographic Distribution of Trypanosoma cruzi TcIII and TcIV. Acta Trop. 2017, 172, 173–179. https://doi.org/10.1016/j.actatropica.2017.05.003.
- 39.
Ledezma, A.P.; Blandon, R.; Schijman, A.G.; et al. Mixed Infections by Different Trypanosoma cruzi Discrete Typing Units among Chagas Disease Patients in an Endemic Community in Panama. PLoS ONE 2020, 15, e0241921. https://doi.org/10.1371/journal.pone.0241921.
- 40.
Dorn, P.L.; McClure, A.G.; Gallaspy, M.D.; et al. The Diversity of the Chagas Parasite, Trypanosoma cruzi, Infecting the Main Central American Vector, Triatoma dimidiata, from Mexico to Colombia. PLoS Negl. Trop. Dis. 2017, 11, e0005878. https://doi.org/10.1371/journal.pntd.0005878.
- 41.
Carrasco, H.J.; Segovia, M.; Llewellyn, M.S.; et al. Geographical Distribution of Trypanosoma cruzi Genotypes in Venezuela. PLoS Negl. Trop. Dis. 2012, 6, e1707. https://doi.org/10.1371/journal.pntd.0001707.
- 42.
Maiguashca Sánchez, J.; Sueto, S.O.B.; Schwabl, P.; et al. Remarkable Genetic Diversity of Trypanosoma cruzi and Trypanosoma Rangeli in Two Localities of Southern Ecuador Identified via Deep Sequencing of Mini-Exon Gene Amplicons. Parasit. Vectors 2020, 13, 279. https://doi.org/10.1186/s13071-020-04079-1.
- 43.
Calvopina, M.; Segovia, G.; Cevallos, W.; et al. Fatal Acute Chagas Disease by Trypanosoma cruzi DTU TcI, Ecuador. BMC Infect. Dis. 2020, 20, 143. https://doi.org/10.1186/s12879-020-4851-0.
- 44.
Brenière, S.F.; Aliaga, C.; Waleckx, E.; et al. Genetic Characterization of Trypanosoma cruzi DTUs in Wild Triatoma infestans from Bolivia: Predominance of TcI. PLoS Negl. Trop. Dis. 2012, 6, e1650. https://doi.org/10.1371/journal.pntd.0001650.
- 45.
Campos-Soto, R.; Ortiz, S.; Cordova, I.; et al. Interactions between Trypanosoma cruzi the Chagas Disease Parasite and Naturally Infected Wild Mepraia Vectors of Chile. Vector Borne Zoonotic Dis. 2016, 16, 181–192. https://doi.org/10.1089/vbz.2015.1850.
- 46.
Ihle-Soto, C.; Costoya, E.; Correa, J.P.; et al. Spatio-Temporal Characterization of Trypanosoma cruzi Infection and Discrete Typing Units Infecting Hosts and Vectors from Non-Domestic Foci of Chile. PLoS Negl. Trop. Dis. 2019, 13, e0007170. https://doi.org/10.1371/journal.pntd.0007170.
- 47.
Ortiz, S.; Ceballos, M.J.; González, C.R.; et al. Trypanosoma cruzi Diversity in Infected Dogs from Areas of the North Coast of Chile. Vet. Parasitol. Reg. Stud. Reports 2016, 5, 42–47. https://doi.org/10.1016/j.vprsr.2016.09.004.
- 48.
Llewellyn, M.S.; Miles, M.A.; Carrasco, H.J.; et al. Genome-Scale Multilocus Microsatellite Typing of Trypanosoma cruzi Discrete Typing Unit I Reveals Phylogeographic Structure and Specific Genotypes Linked to Human Infection. PLoS Pathog. 2009, 5, e1000410. https://doi.org/10.1371/journal.ppat.1000410.
- 49.
Roman, F.; das Chagas Xavier, S.; Messenger, L.A.; et al. Dissecting the Phyloepidemiology of Trypanosoma cruzi I (TcI) in Brazil by the Use of High Resolution Genetic Markers. PLoS Negl. Trop. Dis. 2018, 12, e0006466. https://doi.org/10.1371/journal.pntd.0006466.
- 50.
Costales, J.A.; Jara-Palacios, M.A.; Llewellyn, M.S.; et al. Trypanosoma cruzi Population Dynamics in the Central Ecuadorian Coast. Acta Trop. 2015, 151, 88–93. https://doi.org/10.1016/j.actatropica.2015.07.017.
- 51.
Gómez-Palacio, A.; Lopera, J.; Rojas, W.; et al. Multilocus Analysis Indicates That Trypanosoma cruzi I Genetic Substructure Associated with Sylvatic and Domestic Cycles Is Not an Attribute Conserved throughout Colombia. Infect. Genet. Evol. 2016, 38, 35–43. https://doi.org/10.1016/j.meegid.2015.11.026.
- 52.
Herrera, C.P.; Licon, M.H.; Nation, C.S.; et al. Genotype Diversity of Trypanosoma cruzi in Small Rodents and Triatoma sanguisuga from a Rural Area in New Orleans, Louisiana. Parasit. Vectors 2015, 8, 123. https://doi.org/10.1186/s13071-015-0730-8.
- 53.
Garcia, M.N.; Burroughs, H.; Gorchakov, R.; et al. Molecular Identification and Genotyping of Trypanosoma cruzi DNA in Autochthonous Chagas Disease Patients from Texas, USA. Infect. Genet. Evol. 2017, 49, 151–156. https://doi.org/10.1016/j.meegid.2017.01.016.
- 54.
García-López, C.; Santos-Hernández, N.G.; Gutiérrez-Jiménez, J.; et al. Identification of Discrete Typing Units of Trypanosoma cruzi Isolated from Domestic Environments in Southeastern Mexico. Vector Borne Zoonotic Dis. 2024, 24, 172–176. https://doi.org/10.1089/vbz.2023.0075.
- 55.
Díaz-Valdez, J.; Martínez, I.; Rodríguez-Moreno, Á.; et al. Multiple Discrete Typing Units of Trypanosoma cruzi Infect Sylvatic Triatoma dimidiata and Panstrongylus rufotuberculatus in Southeast Mexico. Am. J. Trop. Med. Hyg. 2021, 105, 1042–1049. https://doi.org/10.4269/ajtmh.20-1574.
- 56.
Lima, V.dos S.; Xavier, S.C.; Maldonado, I.F.; et al. Expanding the Knowledge of the Geographic Distribution of Trypanosoma cruzi TcII and TcV/TcVI Genotypes in the Brazilian Amazon. PLoS ONE 2014, 9, e116137. https://doi.org/10.1371/journal.pone.0116137.
- 57.
de Souza, T.K.M.; Westphalen, E.V.N.; Westphalen, S.R.; et al. Genetic Diversity of Trypanosoma cruzi Strains Isolated from Chronic Chagasic Patients and Non-Human Hosts in the State of São Paulo, Brazil. Mem. Inst. Oswaldo Cruz 2022, 117, e210125. https://doi.org/10.1590/0074-02760220125.
- 58.
Perez, E.; Monje, M.; Chang, B.; et al. Predominance of Hybrid Discrete Typing Units of Trypanosoma cruzi in Domestic Triatoma infestans from the Bolivian Gran Chaco Region. Infect. Genet. Evol. 2013, 13, 116–123. https://doi.org/10.1016/j.meegid.2012.09.014.
- 59.
Acosta, N.; López, E.; Lewis, M.D.; et al. Hosts and Vectors of Trypanosoma cruzi Discrete Typing Units in the Chagas Disease Endemic Region of the Paraguayan Chaco. Parasitology 2017, 144, 884–898. https://doi.org/10.1017/S0031182016002663.
- 60.
Alvedro, A.; Macchiaverna, N.P.; Murphy, N.; et al. Unusual Frequency of Trypanosoma cruzi DTU TcI and Predominance of Hybrid Lineages in Triatoma infestans before and after Control Interventions in the Argentinian Chaco. Acta Trop. 2025, 261, 107502. https://doi.org/10.1016/j.actatropica.2024.107502.
- 61.
Ramírez, J.D.; Guhl, F.; Rendón, L.M.; et al. Chagas Cardiomyopathy Manifestations and Trypanosoma cruzi Genotypes Circulating in Chronic Chagasic Patients. PLoS Negl. Trop. Dis. 2010, 4, e899. https://doi.org/10.1371/journal.pntd.0000899.
- 62.
Lozano-Arias, D.; García-Alzate, R.; Tineo, E.; et al. Ecopathogenic Complexes of American Trypanosomiasis in Endemic Areas of Venezuela: Diagnosis and Variability of Trypanosoma cruzi. J. Vector Borne Dis. 2021, 58, 18–27. https://doi.org/10.4103/0972-9062.321749.
- 63.
Ramos-Ligonio, A.; Torres-Montero, J.; López-Monteon, A.; et al. Extensive Diversity of Trypanosoma cruzi Discrete Typing Units Circulating in Triatoma dimidiata from Central Veracruz, Mexico. Infect. Genet. Evol. 2012, 12, 1341–1343. https://doi.org/10.1016/j.meegid.2012.04.024.
- 64.
Polonio, R.; López-Domínguez, J.; Herrera, C.; et al. Molecular Ecology of Triatoma dimidiata in Southern Belize Reveals Risk for Human Infection and the Local Differentiation of Trypanosoma cruzi Parasites. Int. J. Infect. Dis. 2021, 108, 320–329. https://doi.org/10.1016/j.ijid.2021.05.083.
- 65.
Vandermark, C.; Zieman, E.; Boyles, E.; et al. Trypanosoma cruzi Strain TcIV Infects Raccoons from Illinois. Mem. Inst. Oswaldo Cruz 2018, 113, 30–37. https://doi.org/10.1590/0074-02760170230.
- 66.
Rojas-Cortez, M.; Pinazo, M.J.; Garcia, L.; et al. Trypanosoma cruzi-Infected Panstrongylus Geniculatus and Rhodnius robustus Adults Invade Households in the Tropics of Cochabamba Region of Bolivia. Parasit. Vectors 2016, 9, 158. https://doi.org/10.1186/s13071-016-1445-1.
- 67.
Flores-López, C.A.; Esquivias-Flores, E.A.; Guevara-Carrizales, A. Phylogenetic Description of Trypanosoma cruzi Isolates from Dipetalogaster Maxima: Occurrence of TcI, TcIV, and TcIV-USA. Infect. Genet. Evol. 2023, 113, 105465. https://doi.org/10.1016/j.meegid.2023.105465.
- 68.
Zingales, B. Trypanosoma cruzi Genetic Diversity: Something New for Something Known about Chagas Disease Manifestations, Serodiagnosis and Drug Sensitivity. Acta Trop. 2018, 184, 38–52. https://doi.org/10.1016/j.actatropica.2017.09.017.
- 69.
Pérez-Cascales, E.; Sossa-Soruco, V.M.; Brenière, S.F.; et al. Reinfestation with Triatoma infestans despite Vigilance Efforts in the Municipality of Saipina, Santa Cruz, Bolivia: Situational Description Two Months after Fumigation. Acta Trop. 2020, 203, 105292. https://doi.org/10.1016/j.actatropica.2019.105292.
- 70.
Sanchez, L.; Messenger, L.A.; Bhattacharyya, T.; et al. Congenital Chagas Disease in Santa Cruz Department, Bolivia, Is Dominated by Trypanosoma cruzi Lineage V. Trans. R. Soc. Trop. Med. Hyg. 2022, 116, 80–84. https://doi.org/10.1093/trstmh/trab089.
- 71.
Arenas, M.; Campos, R.; Coronado, X.; et al. Trypanosoma cruzi Genotypes of Insect Vectors and Patients with Chagas of Chile Studied by Means of Cytochrome b Gene Sequencing, Minicircle Hybridization, and Nuclear Gene Polymorphisms. Vector Borne Zoonotic Dis. 2012, 12, 196–205. https://doi.org/10.1089/vbz.2011.0683.
- 72.
Bizai, M.L.; Romina, P.; Antonela, S.; et al. Geographic Distribution of Trypanosoma cruzi Genotypes Detected in Chronic Infected People from Argentina. Association with Climatic Variables and Clinical Manifestations of Chagas Disease. Infect. Genet. Evol. 2020, 78, 104128. https://doi.org/10.1016/j.meegid.2019.104128.
- 73.
Barnabé, C.; De Meeûs, T.; Noireau, F.; et al. Trypanosoma cruzi Discrete Typing Units (DTUs): Microsatellite Loci and Population Genetics of DTUs TcV and TcI in Bolivia and Peru. Infect. Genet. Evol. 2011, 11, 1752–1760. https://doi.org/10.1016/j.meegid.2011.07.011.
- 74.
del Pilar Fernández, M.; Cecere, M.C.; Lanati, L.A.; et al. Geographic Variation of Trypanosoma cruzi Discrete Typing Units from Triatoma infestans at Different Spatial Scales. Acta Trop. 2014, 140, 10–18. https://doi.org/10.1016/j.actatropica.2014.07.014.
- 75.
Ramírez, J.D.; Hernández, C.; Montilla, M.; et al. First Report of Human Trypanosoma cruzi Infection Attributed to TcBat Genotype. Zoonoses Public Health 2014, 61, 477–479. https://doi.org/10.1111/zph.12094.
- 76.
Pinto, C.M.; Ocaña-Mayorga, S.; Tapia, E.E.; et al. Bats, Trypanosomes, and Triatomines in Ecuador: New Insights into the Diversity, Transmission, and Origins of Trypanosoma cruzi and Chagas Disease. PLoS ONE 2015, 10, e0139999. https://doi.org/10.1371/journal.pone.0139999.
- 77.
Pinto, C.M.; Kalko, E.K.V.; Cottontail, I.; et al. TcBat a Bat-Exclusive Lineage of Trypanosoma cruzi in the Panama Canal Zone, with Comments on Its Classification and the Use of the 18S rRNA Gene for Lineage Identification. Infect. Genet. Evol. 2012, 12, 1328–1332. https://doi.org/10.1016/j.meegid.2012.04.013.
- 78.
Lima, L.; Espinosa-Álvarez, O.; Ortiz, P.A.; et al. Genetic Diversity of Trypanosoma cruzi in Bats, and Multilocus Phylogenetic and Phylogeographical Analyses Supporting Tcbat as an Independent DTU (Discrete Typing Unit). Acta Trop. 2015, 151, 166–177. https://doi.org/10.1016/j.actatropica.2015.07.015.
- 79.
Guhl, F.; Auderheide, A.; Ramírez, J.D. From Ancient to Contemporary Molecular Eco-Epidemiology of Chagas Disease in the Americas. Int. J. Parasitol. 2014, 44, 605–612. https://doi.org/10.1016/j.ijpara.2014.02.005.
- 80.
Cucunubá, Z.M.; Gutiérrez-Romero, S.A.; Ramírez, J.D.; et al. The Epidemiology of Chagas Disease in the Americas. Lancet Reg. Health Am. 2024, 37, 100881. https://doi.org/10.1016/j.lana.2024.100881.
- 81.
Magalhães, L.M.D.; Viana, A.; Chiari, E.; et al. Differential Activation of Human Monocytes and Lymphocytes by Distinct Strains of Trypanosoma cruzi. PLoS Negl. Trop. Dis. 2015, 9, e0003816. https://doi.org/10.1371/journal.pntd.0003816.
- 82.
Magalhães, L.M.D.; Passos, L.S.A.; Chiari, E.; et al. Co-Infection with Distinct Trypanosoma cruzi Strains Induces an Activated Immune Response in Human Monocytes. Parasite Immunol. 2019, 41, e12668. https://doi.org/10.1111/pim.12668.
- 83.
Magalhães, L.M.D.; Viana, A.; De Jesus, A.C.; et al. Distinct Trypanosoma cruzi Isolates Induce Activation and Apoptosis of Human Neutrophils. PLoS ONE 2017, 12, e0188083. https://doi.org/10.1371/journal.pone.0188083.
- 84.
Da Costa, T.A.; Silva, M.V.; Mendes, M.T.; et al. Immunomodulation by Trypanosoma cruzi: Toward Understanding the Association of Dendritic Cells with Infecting TcI and TcII Populations. J. Immunol. Res. 2014, 2014, 962047. https://doi.org/10.1155/2014/962047.
- 85.
Desale, H.; Tu, W.; Goff, K.; et al. PBMC Transcriptomic Signatures Reflect Trypanosoma cruzi Strain Diversity and Trained Immunity in Chronically Infected Macaques. JCI Insight 2025, 10, e186003. https://doi.org/10.1172/jci.insight.186003.
- 86.
Tarleton, R.L. CD8+ T Cells in Trypanosoma cruzi Infection. Semin. Immunopathol. 2015, 37, 233–238. https://doi.org/10.1007/s00281-015-0481-9.
- 87.
Poveda, C.; Fresno, M.; Gironès, N.; et al. Cytokine Profiling in Chagas Disease: Towards Understanding the Association with Infecting Trypanosoma cruzi Discrete Typing Units (a BENEFIT TRIAL Sub-Study). PLoS ONE 2014, 9, e91154. https://doi.org/10.1371/journal.pone.0091154.
- 88.
Ferreira, B.L.; Ferreira, É.R.; de Brito, M.V.; et al. BALB/c and C57BL/6 Mice Cytokine Responses to Trypanosoma cruzi Infection Are Independent of Parasite Strain Infectivity. Front. Microbiol. 2018, 9, 553. https://doi.org/10.3389/fmicb.2018.00553.
- 89.
Zingales, B.; Macedo, A.M. Fifteen Years after the Definition of Trypanosoma cruzi DTUs: What Have We Learned? Life 2023, 13, 2339. https://doi.org/10.3390/life13122339.
- 90.
Silvestrini, M.M.A.; Alessio, G.D.; Frias, B.E.D.; et al. New Insights into Trypanosoma cruzi Genetic Diversity, and Its Influence on Parasite Biology and Clinical Outcomes. Front. Immunol. 2024, 15, 1342431. https://doi.org/10.3389/fimmu.2024.1342431.
- 91.
Soares, R.P.; Torrecilhas, A.C.; Assis, R.R.; et al. Intraspecies Variation in Trypanosoma cruzi GPI-Mucins: Biological Activities and Differential Expression of α-Galactosyl Residues. Am. J. Trop. Med. Hyg. 2012, 87, 87–96. https://doi.org/10.4269/ajtmh.2012.12-0015.
- 92.
Bhattacharyya, T.; Brooks, J.; Yeo, M.; et al. Analysis of Molecular Diversity of the Trypanosoma cruzi Trypomastigote Small Surface Antigen Reveals Novel Epitopes, Evidence of Positive Selection and Potential Implications for Lineage-Specific Serology. Int. J. Parasitol. 2010, 40, 921–928. https://doi.org/10.1016/j.ijpara.2010.01.002.
- 93.
Risso, M.G.; Garbarino, G.B.; Mocetti, E.; et al. Differential Expression of a Virulence Factor, the Trans-Sialidase, by the Main Trypanosoma cruzi Phylogenetic Lineages. J. Infect. Dis. 2004, 189, 2250–2259. https://doi.org/10.1086/420831.
- 94.
Vela, A.; Coral-Almeida, M.; Sereno, D.; et al. In Vitro Susceptibility of Trypanosoma cruzi Discrete Typing Units (DTUs) to Benznidazole: A Systematic Review and Meta-Analysis. PLoS Negl. Trop. Dis. 2021, 15, e0009269. https://doi.org/10.1371/journal.pntd.0009269.
- 95.
Moraes, C.B.; Giardini, M.A.; Kim, H.; et al. Nitroheterocyclic Compounds Are More Efficacious than CYP51 Inhibitors against Trypanosoma cruzi: Implications for Chagas Disease Drug Discovery and Development. Sci. Rep. 2014, 4, 4703. https://doi.org/10.1038/srep04703.
- 96.
Martinez, S.J.; Nardella, G.N.; Rodríguez, M.E.; et al. Biological Features of TcM: A New Trypanosoma cruzi Isolate from Argentina Classified into TcV Lineage. Curr. Res. Microb. Sci. 2022, 3, 100152. https://doi.org/10.1016/j.crmicr.2022.100152.
- 97.
De Ornelas Toledo, M.J.; Bahia, M.T.; Carneiro, C.M.; et al. Chemotherapy with Benznidazole and Itraconazole for Mice Infected with Different Trypanosoma cruzi Clonal Genotypes. Antimicrob. Agents Chemother. 2003, 47, 223–230. https://doi.org/10.1128/AAC.47.1.223-230.2003.
- 98.
Quebrada Palacio, L.P.; González, M.N.; Hernandez-Vasquez, Y.; et al. Phenotypic Diversity and Drug Susceptibility of Trypanosoma cruzi TcV Clinical Isolates. PLoS ONE 2018, 13, e0203462. https://doi.org/10.1371/journal.pone.0203462.
- 99.
Yun, O.; Lima, M.A.; Ellman, T.; et al. Feasibility, Drug Safety, and Effectiveness of Etiological Treatment Programs for Chagas Disease in Honduras, Guatemala, and Bolivia: 10-Year Experience of Médecins Sans Frontières. PLoS Negl. Trop. Dis. 2009, 3, e488. https://doi.org/10.1371/journal.pntd.0000488.
- 100.
Villarreal, D.; Barnabé, C.; Sereno, D.; et al. Lack of Correlation between in Vitro Susceptibility to Benznidazole and Phylogenetic Diversity of Trypanosoma cruzi, the Agent of Chagas Disease. Exp. Parasitol. 2004, 108, 24–31. https://doi.org/10.1016/j.exppara.2004.07.001.
- 101.
Franco, J.; Ferreira, R.C.; Ienne, S.; et al. ABCG-like Transporter of Trypanosoma cruzi Involved in Benznidazole Resistance: Gene Polymorphisms Disclose Inter-Strain Intragenic Recombination in Hybrid Isolates. Infect. Genet. Evol. 2015, 31, 198–208. https://doi.org/10.1016/j.meegid.2015.01.030.
- 102.
Muñoz-Calderón, A.; Ramírez, J.L.; Díaz-Bello, Z.; et al. Genetic Characterization of Trypanosoma cruzi I Populations from an Oral Chagas Disease Outbreak in Venezuela: Natural Resistance to Nitroheterocyclic Drugs. ACS Infect. Dis. 2023, 9, 582–592. https://doi.org/10.1021/acsinfecdis.2c00569.
- 103.
Martínez, I.; Nogueda, B.; Martínez-Hernández, F.; et al. Microsatellite and Mini-Exon Analysis of Mexican Human DTU I Trypanosoma cruzi Strains and Their Susceptibility to Nifurtimox and Benznidazole. Vector Borne Zoonotic Dis. 2013, 13, 181–187. https://doi.org/10.1089/vbz.2012.1072.
- 104.
De Valamiel Oliveira-Silva, J.C.; Machado-De-Assis, G.F.; Oliveira, M.T.; et al. Experimental Benznidazole Treatment of Trypanosoma cruzi II Strains Isolated from Children of the Jequitinhonha Valley, Minas Gerais, Brazil, with Chagas Disease. Mem. Inst. Oswaldo Cruz 2015, 110, 86–94. https://doi.org/10.1590/0074-02760140260.
- 105.
Dickson, K.P.; Costales, J.A.; Domagalska, M.A.; et al. Innovation through Instability? Genome (Dis)Organisation in Trypanosoma cruzi. Trends Parasitol. 2025, 41, 449–459. https://doi.org/10.1016/j.pt.2025.04.008.
- 106.
Campos, M.C.O.; Leon, L.L.; Taylor, M.C.; et al. Benznidazole-Resistance in Trypanosoma cruzi: Evidence That Distinct Mechanisms Can Act in Concert. Mol. Biochem. Parasitol. 2014, 193, 17–19. https://doi.org/10.1016/j.molbiopara.2014.01.002.
- 107.
Teh-Poot, C.F.; Alfaro-Chacón, A.; Pech-Pisté, L.M.; et al. Immunogenicity of Trypanosoma cruzi Multi-Epitope Recombinant Protein as an Antigen Candidate for Chagas Disease Vaccine in Humans. Pathogens 2025, 14, 342. https://doi.org/10.3390/pathogens14040342.
- 108.
Hernández-Flores, A.; Elías-Díaz, D.; Cubillo-Cervantes, B.; et al. Fighting Strategies Against Chagas’ Disease: A Review. Pathogens 2025, 14, 183. https://doi.org/10.3390/pathogens14020183.
- 109.
de Brito, R.N.; Tanner, S.; Runk, J.V.; et al. Looking through the Lens of Social Science Approaches: A Scoping Review of Leishmaniases and Chagas Disease Research. Acta Trop. 2024, 249, 107059. https://doi.org/10.1016/j.actatropica.2023.107059.
- 110.
Gürtler, R.E.; Yadon, Z.E. Eco-Bio-Social Research on Community-Based Approaches for Chagas Disease Vector Control in Latin America. Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 91–98. https://doi.org/10.1093/trstmh/tru203.
- 111.
Bates, B.R.; Carrasco-Tenezaca, M.; Mendez-Trivino, A.M.; et al. Identifying Barriers and Facilitators for Home Reconstruction for Prevention of Chagas Disease: An Interview Study in Rural Loja Province, Ecuador. Trop. Med. Infect. Dis. 2023, 8, 228. https://doi.org/10.3390/tropicalmed8040228.
- 112.
Castro-Arroyave, D.; Monroy, M.C.; Irurita, M.I. Integrated Vector Control of Chagas Disease in Guatemala: A Case of Social Innovation in Health. Infect. Dis. Poverty 2020, 9, 25. https://doi.org/10.1186/s40249-020-00639-w.

This work is licensed under a Creative Commons Attribution 4.0 International License.


