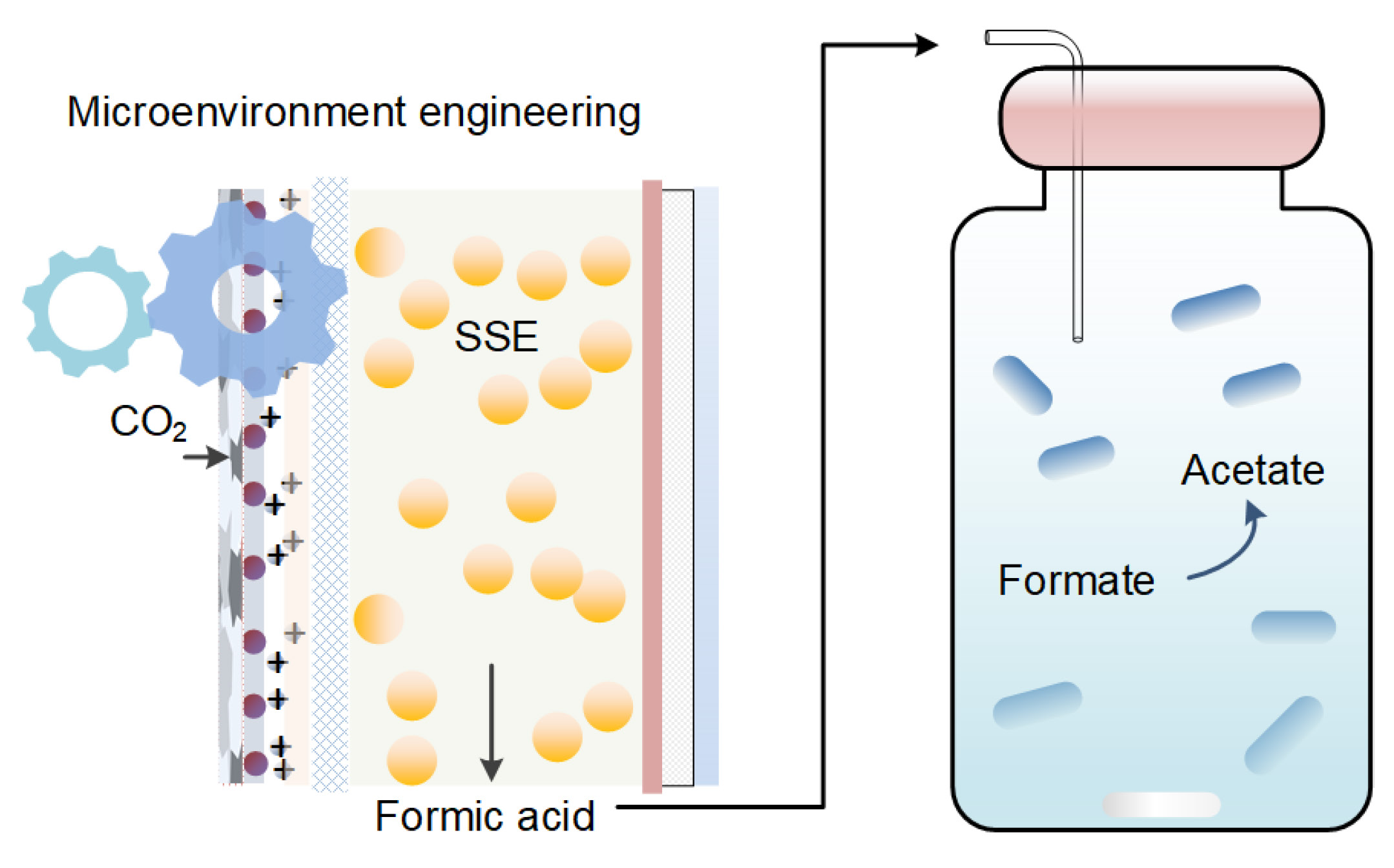
Tandem systems coupling electrochemical CO2 reduction (CO2RR) with microbial conversion offer a promising strategy to overcome the limitations of conventional microbial electrosynthesis (MES), which typically suffers from low reaction rates due to its reliance on electrode–biofilm architectures. In this study, we develop a solid-state electrolyte (SSE) reactor through microenvironmental engineering, eliminating the need for prefabricated anion exchange membranes (AEMs) and advanced electrocatalysts—both of which have constrained the practical deployment of SSE systems. A potassium-infused, sandwich-structured gas diffusion electrode (GDE) is fabricated via a three-layer, layer-by-layer assembly, an approach shown to be superior to the physical mixing of any two of the constituent layers. Furthermore, a simple filter paper separator is employed in place of pristine or porous AEMs to mitigate hydrogen accumulation and maintain a pH gradient at the GDE–SSE interface. Using commercial bismuth nanoparticles as the electrocatalyst, this configuration enables stable operation for over 130 h, yielding more than 3 litres of pure formic acid solution at concentrations exceeding 0.1 M. For microbial conversion, the integrated system achieves a near-theoretical formate-to-acetate molar ratio of 4.1:1 with a pure acetogenic culture, while a mixed consortium yields a slightly higher ratio of 5.0:1. This work underscores the potential of microenvironmental engineering in advancing cost-effective tandem systems for CO2 valorization.
- Open Access
- Article
Microenvironmental Engineering of a Solid-State Electrolyte Reactor for Efficient Microbial Electrosynthesis of Acetate from CO2
- Na Chu 1,†,
- Xiaobing Wu 2,†,
- Weihong Zheng 2,
- Yanwei Luo 2,
- Hang Lin 2,
- Bingjie Sun 2,
- Huilin Chen 2,
- Chenghao Xu 2,
- Yan Wang 2,
- Xintong Xu 2,
- Jiping Tang 2,*,
- Yaoxing Liu 1,
- Yong Jiang 2,*
Author Information
Received: 17 Feb 2026 | Revised: 27 Mar 2026 | Accepted: 31 Mar 2026 | Published: 13 Apr 2026
Abstract
Graphical Abstract
Keywords
CO2 electrolysis | microenvironmental engineering | acetogen | gas diffusion electrode | formic acid
References
- 1.
Kuang, M.; Li, B.; Zhou, L.; et al. Carbon Dioxide Upgrading to Biodegradable Plastics through Photo/Electro-Synthetic Biohybrid Systems. Angew. Chem. Int. Ed. 2025, 64, e202422357.
- 2.
Ying, Z.; Qiu, Q.; Ye, J.; et al. Mechanism, Performance Enhancement, and Economic Feasibility of CO2 Microbial Electrosynthesis Systems: A Data-Driven Analysis of Research Topics and Trends. Renew. Sust. Energy Rev. 2024, 202, 114704.
- 3.
Lovat, S.J.; Ben-Nissan, R.; Milshtein, E.; et al. Electro-Microbial Production Techno-Economic Viability and Environmental Implications. Nat. Biotechnol. 2025, 43, 848–853.
- 4.
Liu, Z.; Wang, K.; Chen, Y.; et al. Third-Generation Biorefineries as the Means to Produce Fuels and Chemicals from CO2. Nat. Catal. 2020, 3, 274–288.
- 5.
Jourdin, L.; Grieger, T.; Monetti, J.; et al. High Acetic Acid Production Rate Obtained by Microbial Electrosynthesis from Carbon Dioxide. Environ. Sci. Technol. 2015, 49, 13566–13574.
- 6.
Chu, N.; Jiang, Y.; Liang, Q.; et al. Electricity-Driven Microbial Metabolism of Carbon and Nitrogen: A Waste-to-Resource Solution. Environ. Sci. Technol. 2023, 57, 4379–4395.
- 7.
Shakeel, S.; Khan, M.Z.; Shakeel, M. Microbial Electrosynthesis of Acetic Acid from Carbon Dioxide Using a Bimetallic-Granular Activated Carbon Cathode. Bioelectrochemistry 2026, 168, 109158.
- 8.
Bian, B.; Ma, X.; Li, S.; et al. Microbial Electrosynthesis of Methane in an Up-Scaled Zero-Gap Cell. Water Res. 2026, 297, 125723.
- 9.
Li, L.; Ye, W.; Liu, Q.; et al. Operando Raman Characterization of Unique Electroinduced Molecular Tautomerization in Zero-Gap Electrolyzers Promotes CO2 Reduction. Proc. Natl. Acad. Sci. USA 2025, 122, e2418144122.
- 10.
Pham, T.H.; Lai, H.D.T.; Dang, N.K.; et al. Ambipolar Ion Transport Membranes Enable Stable Noble-Metal-Free CO2 Electrolysis in Neutral Media. Adv. Energy Mater. 2025, 15, e04286.
- 11.
Wang, Z.; Wang, W.; Yang, Q.; et al. Highly Selective Acidic CO2 Electroreduction with Large Current on Polypyrrole-Modified Ag Catalyst by Local Microenvironment Modulation. Adv. Energy Mater. 2025, 15, 2405419.
- 12.
Lv, Z.; Wang, C.; Liu, W.; et al. Enhanced CO2 Adsorption and Conversion in Diethanolamine-Cu Interfaces Achieving Stable Neutral Ethylene Electrosynthesis. Adv. Energy Mater. 2024, 14, 2402551.
- 13.
Li, H.; Opgenorth, P.H.; Wernick, D.G.; et al. Integrated Electromicrobial Conversion of CO2 to Higher Alcohols. Science 2012, 335, 1596.
- 14.
Hann, E.C.; Overa, S.; Harland-Dunaway, M.; et al. A Hybrid Inorganic–Biological Artificial Photosynthesis System for Energy-Efficient Food Production. Nat. Food 2022, 3, 461–471.
- 15.
Staerz, A.F.; van Leeuwen, M.; Priamushko, T.; et al. Effects of Iron Species on Low Temperature CO2 Electrolyzers. Angew. Chem. Int. Ed. 2024, 63, e202306503.
- 16.
Cui, H.; Liu, W.; Ma, C.; et al. Converting CO2 to Single-Cell Protein via an Integrated Electrocatalytic-Biosynthetic System. Appl. Catal. B Environ. Energy 2024, 350, 123946.
- 17.
Lee, G.; Jo, H.J.; Choi, J.; et al. CO2 Upgrading into Bioproducts Using a Two-Step Abiotic-Biotic System. Proc. Natl. Acad. Sci. USA 2025, 122, e2512565122.
- 18.
Jiang, Y.; Wu, G.; Pu, Y.; et al. Flow-Electrode Capacitive Separation of Organic Acid Products and Recovery of Alkali Cations after Acidic CO2 Electrolysis. Proc. Natl. Acad. Sci. USA 2024, 121, e2408205121.
- 19.
Zhu, P.; Wu, Z.Y.; Elgazzar, A.; et al. Continuous Carbon Capture in an Electrochemical Solid-Electrolyte Reactor. Nature 2023, 618, 959–966.
- 20.
- 21.
Chu, N.; Jiang, Y.; Zeng, R.J.; et al. Solid Electrolytes for Low-Temperature Carbon Dioxide Valorization: A Review. Environ. Sci. Technol. 2024, 58, 10881–10896.
- 22.
Sun, Y.; Dai, L.; Sui, N.L.D.; et al. Direct Parallel Electrosynthesis of High-Value Chemicals from Atmospheric Components on Symmetry-Breaking Indium Sites. Proc. Natl. Acad. Sci. USA 2024, 121, e2409620121.
- 23.
Yuan, C.Y.; Feng, L.; Qin, X.; et al. Constructing Metal(II)-Sulfate Site Catalysts toward Low Overpotential Carbon Dioxide Electroreduction to Fuel Chemicals. Angew. Chem. Int. Ed. 2024, 63, e202405255.
- 24.
Zhao, Z.H.; Huang, J.R.; Huang, D.S.; et al. Efficient Capture and Electroreduction of Dilute CO2 into Highly Pure and Concentrated Formic Acid Aqueous Solution. J. Am. Chem. Soc. 2024, 146, 14349–14356.
- 25.
Ma, M.; Seger, B. Rational Design of Local Reaction Environment for Electrocatalytic Conversion of CO2 into Multicarbon Products. Angew. Chem. Int. Ed. 2024, 63, e202401185.
- 26.
Zeng, M.; Fang, W.; Cen, Y.; et al. Reaction Environment Regulation for Electrocatalytic CO2 Reduction in Acids. Angew. Chem. Int. Ed. 2024, 63, e202404574.
- 27.
Liu, J.; Xie, S.; Li, D.; et al. Microenvironmental Hydrophobicity-Regulated Evolution of Reaction Sites for Boosting Electrochemical CO2 Reduction. Appl. Catal. B Environ. Energy 2025, 379, 125700.
- 28.
Pu, Y.; Wang, Y.; Wu, G.; et al. Tandem Acidic CO2 Electrolysis Coupled with Syngas Fermentation: A Two-Stage Process for Producing Medium-Chain Fatty Acids. Environ. Sci. Technol. 2024, 58, 7445–7456.
- 29.
Disch, J.; Ingenhoven, S.; Vierrath, S. Bipolar Membrane with Porous Anion Exchange Layer for Efficient and Long-Term Stable Electrochemical Reduction of CO2 to CO. Adv. Energy Mater. 2023, 13, 2301614.
- 30.
Chu, N.; Wu, X.; Zhao, Z.; et al. Biohybrid CO2 Electrolysis under External Mode: Using Pure Formic Acid Extracted from CO2 Electroreduction for Diverse Microbial Conversion. Fundam. Res. 2025, 5, 2597–2606.
- 31.
Chu, N.; Jiang, Y.; Wang, D.; et al. Super-Fast Charging Biohybrid Batteries through a Power-to-Formate-to-Bioelectricity Process by Combining Microbial Electrochemistry and CO2 Electrolysis. Angew. Chem. Int. Ed. 2023, 62, e202312147.
- 32.
Xia, C.; Zhu, P.; Jiang, Q.; et al. Continuous Production of Pure Liquid Fuel Solutions via Electrocatalytic CO2 Reduction Using Solid-Electrolyte Devices. Nat. Energy 2019, 4, 776–785.
- 33.
Rashid, M.; Nabil, S.K.; Adnan, M.A.; et al. Cation-Infused Bilayer Ionomer Coating Enables High Partial Current Density toward Multi Carbon Products in CO2 Electrolysis. Adv. Energy Mater. 2024, 14, 2400570.
- 34.
Kim, C.; Bui, J.C.; Luo, X.; et al. Tailored Catalyst Microenvironments for CO2 Electroreduction to Multicarbon Products on Copper Using Bilayer Ionomer Coatings. Nat. Energy 2021, 6, 1026–1034.
- 35.
Li, W.; Yin, Z.; Gao, Z.; et al. Bifunctional Ionomers for Efficient Co-Electrolysis of CO2 and Pure Water towards Ethylene Production at Industrial-Scale Current Densities. Nat. Energy 2022, 7, 835–843.
- 36.
Liu, J.; Zhang, B.; Chen, D.; et al. Steering the Selectivity of CORR from Acetate to Ethanol via Tailoring the Thermodynamic Activity of Water. Angew. Chem. Int. Ed. 2024, 63, e202412266.
- 37.
Zhang, M.; Zhu, W.; Liu, Z.; et al. Selective Sieving Effect of Multi-Atomic Bismuth Interfaces for Efficient Formate Electrosynthesis and Evolution at Industrial Current Density. Angew. Chem. Int. Ed. 2025, 64, e202510206.
- 38.
Zhu, J.; Li, J.; Lu, R.; et al. Surface Passivation for Highly Active, Selective, Stable, and Scalable CO2 Electroreduction. Nat. Commun. 2023, 14, 4670.
- 39.
Fan, L.; Xia, C.; Zhu, P.; et al. Electrochemical CO2 Reduction to High-Concentration Pure Formic Acid Solutions in an All-Solid-State Reactor. Nat. Commun. 2020, 11, 3633.
- 40.
Kang, Y.; Kim, Y.; Doh, Y.; et al. Boosting Current Density of Electrocatalytic CO2 Reduction Using Metal–Enzyme Hybrid Cathodes. Angew. Chem. Int. Ed. 2025, 64, e202504380.
- 41.
Zhang, Y.; Pan, B.; Li, Y.; et al. Electrochemical pH-Swing CO2 Capture Facilitated by Suppressed Bubble Accumulation at Electrode/Electrolyte Interfaces. Angew. Chem. Int. Ed. 2026, 65, e13456.
- 42.
Lee, G.; Rasouli, A.S.; Lee, B.-H.; et al. CO2 Electroreduction to Multicarbon Products from Carbonate Capture Liquid. Joule 2023, 7, 1277–1288.
- 43.
Yang, Y.; Zhang, J.; Tan, Z.; et al. Highly Selective Production of C2+ Oxygenates from CO2 in Strongly Acidic Condition by Rough Ag-Cu Electrocatalyst. Angew. Chem. Int. Ed. 2024, 63, e202408873.
- 44.
Liu, H.; Bai, Y.; Wu, M.; et al. A Regenerable Bi-Based Catalyst for Efficient and Stable Electrochemical CO2 Reduction to Formate at Industrial Current Densities. Angew. Chem. Int. Ed. 2024, 63, e202411575.
- 45.
Zheng, T.; Liu, C.; Guo, C.; et al. Copper-Catalysed Exclusive CO2 to Pure Formic Acid Conversion via Single-Atom Alloying. Nat. Nanotechnol. 2021, 16, 1386–1393.
- 46.
Zhang, X.; Fang, Z.; Zhu, P.; et al. Electrochemical Regeneration of High-Purity CO2 from (Bi)carbonates in a Porous Solid Electrolyte Reactor for Efficient Carbon Capture. Nat. Energy 2024, 10, 55–65.
- 47.
Wang, Z.; Zhou, Y.; Liu, D.; et al. Carbon-Confined Indium Oxides for Efficient Carbon Dioxide Reduction in a Solid-State Electrolyte Flow Cell. Angew. Chem. Int. Ed. 2022, 61, e202200552.
- 48.
Wang, X.; Zhou, M.; Wang, M.; et al. Copper-Bridge-Enhanced p-Band Center Modulation of Carbon-Bismuth Heterojunction for CO2 Electroreduction. Nano Lett. 2023, 23, 10946–10954.
- 49.
Moon, J.; Donig, J.; Kramer, S.; et al. Formate Metabolism in the Acetogenic Bacterium Acetobacterium woodii. Environ. Microbiol. 2021, 23, 4214–4227.
- 50.
Claassens, N.J.; Cotton, C.A.R.; Kopljar, D.; et al. Making Quantitative Sense of Electromicrobial Production. Nat. Catal. 2019, 2, 437–447.
- 51.
Valentini, F.; Kozell, V.; Petrucci, C.; et al. Formic Acid, a Biomass-Derived Source of Energy and Hydrogen for Biomass Upgrading. Energy Environ. Sci. 2019, 12, 2646–2664.
- 52.
Cotton, C.A.; Edlich-Muth, C.; Bar-Even, A. Reinforcing Carbon Fixation: CO2 Reduction Replacing and Supporting Carboxylation. Curr. Opin. Biotechnol. 2018, 49, 49–56.
- 53.
Hua, Y.; Kang, D.; Huang, J.; et al. Electrified CO2-to-HCOOH Valorization: A Comparative Technical Analysis on Acidic Flow Cell and Solid-State Electrolyte Cell Reactors. ACS Energy Lett. 2026, 11, 1880–1888.
- 54.
Xue, J.; Ji, B.; Zhong, K.; et al. Promoted CO2 Electrolysis to Formic Acid Using Single Atom Cobalt Alloyed Tin. Adv. Mater. 2026, 38, e72719.
- 55.
Chen, X.; Lu, R.; Li, C.; et al. Activating Inert Non-Defect Sites in Bi Catalysts Using Tensile Strain Engineering for Highly Active CO2 Electroreduction. Nat. Commun. 2025, 16, 1927.
- 56.
Adaryan, S.; Wi, T.-U.; Santos, K.; et al. Free-Standing Porous Composite Polyelectrolyte for Efficient CO2 Electrolysis. Environ. Sci. Technol. 2025, 59, 26819–26829.
- 57.
Xu, Y.; Zhao, K.; Chang, X.; et al. Emerging Roles of Cations in Electrocatalytic Reduction of CO2 and CO. Nat. Energy 2026, 11, 387–399.
- 58.
Liu, Q.; Flores-Alsina, X.; Ramin, E.; et al. Making Waves: Power-to-X for the Water Resource Recovery Facilities of the Future. Water Res. 2024, 257, 121691.
- 59.
Kormányos, A.; Szirmai, A.; Endrődi, B.; et al. Pairing Electrochemical CO2 Reduction with Glycerol Oxidation: Bottlenecks Today, Opportunities Tomorrow. Joule 2025, 9, 102096.
- 60.
Zhang, Y.; Zhang, R.; Chen, F.; et al. Mass-Transfer-Enhanced Hydrophobic Bi Microsheets for Highly Efficient Electroreduction of CO2 to Pure Formate in a Wide Potential Window. Appl. Catal. B Environ. 2023, 322, 122127.
- 61.
Tan, Z.; Zhang, J.; Yang, Y.; et al. Continuous Production of Formic Acid Solution from Electrocatalytic CO2 Reduction Using Mesoporous Bi2O3 Nanosheets as Catalyst. CCS Chem. 2024, 6, 100–109.
- 62.
Lin, L.; He, X.; Zhang, X.G.; et al. A Nanocomposite of Bismuth Clusters and Bi2O2CO3 Sheets for Highly Efficient Electrocatalytic Reduction of CO2 to Formate. Angew. Chem. Int. Ed. 2023, 62, e202214959.
- 63.
Li, L.; Liu, Z.; Yu, X.; et al. Achieving High Single-Pass Carbon Conversion Efficiencies in Durable CO2 Electroreduction in Strong Acids via Electrode Structure Engineering. Angew. Chem. Int. Ed. 2023, 62, e202300226.
- 64.
Liu, G.; Zhong, Y.; Liu, Z.; et al. Solar-Driven Sugar Production Directly from CO2 via a Customizable Electrocatalytic-Biocatalytic Flow System. Nat. Commun. 2024, 15, 2636.
- 65.
Zhang, G.; Ji, N.; Lyu, S.; et al. Artificial Synthesis of Polyesters at Ambient Condition via Consecutive CO2 Electrolysis and Fermentation. Nano Res. 2024, 17, 6016–6025.
- 66.
Li, X.; Wang, J.H.; Yuan, C.Y.; et al. A Unique Amorphous Porous BiSbOx Nanotube with Abundant Unsaturated Sb‑Stabilized BiO8−x Sites for Efficient CO2 Electroreduction in a Wide Potential Window. Adv. Funct. Mater. 2024, 34, 2402220.
- 67.
Zhang, M.; Cao, A.; Xiang, Y.; et al. Strongly Coupled Ag/Sn–SnO2 Nanosheets toward CO2 Electroreduction to Pure HCOOH Solutions at Ampere-Level Current. Nano-Micro Lett. 2023, 16, 50.
- 68.
Jia, B.; Chen, Z.; Li, C.; et al. Indium Cyanamide for Industrial-Grade CO2 Electroreduction to Formic Acid. J. Am. Chem. Soc. 2023, 145, 14101–14111.

This work is licensed under a Creative Commons Attribution 4.0 International License.


