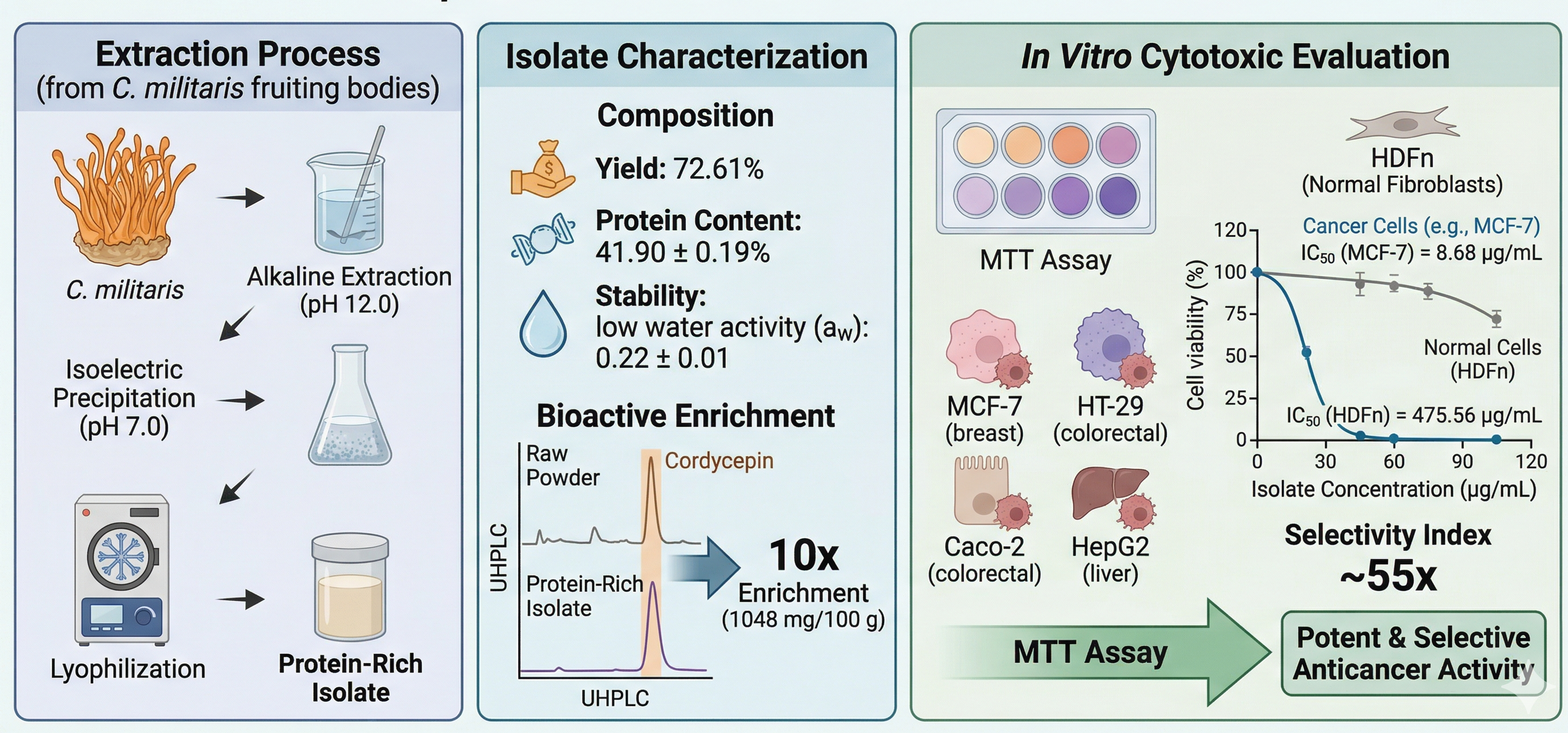
Cancer remains a global health challenge due to the limitations of conventional therapies, including severe side effects and drug resistance. While Cordyceps militaris (L.) Fr. is well-regarded in traditional medicine for its antitumor effects, research has primarily focused on small molecules like cordycepin. The anticancer potential of its total protein-rich isolate remains less characterized. This study aimed to extract, characterize, and evaluate the in vitro cytotoxic activities of the protein-rich isolate from C. militaris fruiting bodies against various human cancer cell lines. Proteins were extracted from dried fruiting bodies using an alkaline extraction method (pH 12.0) followed by isoelectric precipitation (pH 7.0) and lyophilization. The isolate was analyzed for yield, protein content, water activity, and moisture. Bioactive markers (cordycepin and adenosine) were quantified using UHPLC. Cytotoxicity was assessed via MTT assay against MCF-7 (breast), HT-29 and Caco-2 (colorectal), and HepG2 (liver) cancer cells, with HDFn (normal fibroblasts) used to determine selectivity. Results showed that the extraction process achieved a high yield of 72.61% with a protein content of 41.90 ± 0.19%. The isolate demonstrated excellent stability with a low water activity (aw) of 0.22 ± 0.01. Notably, cordycepin was significantly enriched in the isolate, increasing tenfold to 1048 mg/100 g compared to the raw powder. The protein-rich isolate showed dose-dependent cytotoxicity, with the highest potency against MCF-7 cells (IC50 = 8.68 µg/mL). Crucially, the isolate exhibited promising selectivity in this model, being nearly 55 times less toxic to normal HDFn cells (IC50 = 475.56 µg/mL). It is concluded that C. militaris protein-rich isolate is a stable, bioactive-rich fraction with potent and selective anticancer activity. The significant enrichment of cordycepin and the wide therapeutic window suggest its potential as a safe candidate for functional foods or adjuvant cancer therapies.
- Open Access
- Article
Protein-rich Isolate Extracted from Cordyceps militaris Fruiting Body and Anticancer Activities on Cancer Cells
- Sarayut Radapong 1,*,
- Tiyanee Sahad 1,
- Nathaphat Harnkit 1,
- Yanit Harntaweesup 2,
- Wannaporn Payao 2,
- Sumate Sanchoo 3,
- Praw Suppajariyawat 1
Author Information
Received: 09 Mar 2026 | Revised: 17 Apr 2026 | Accepted: 22 Apr 2026 | Published: 30 Apr 2026
Abstract
Graphical Abstract
Keywords
Cordyceps militaris | protein-rich isolate | cordycepin | anticancer activity | cytotoxicity
References
- 1.
Sung, H.; Ferlay, J.; Siegel, R.L.; et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249.
- 2.
Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803.
- 3.
Paterson, R.R.M. Cordyceps: A Traditional Chinese Medicine and Another Fungal Therapeutic Biofactory? Phytochemistry 2008, 69, 1469–1495.
- 4.
Das, S.K.; Masuda, M.; Sakurai, A.; et al. Medicinal Uses of the Mushroom Cordyceps militaris: Current State and Prospects. Fitoterapia 2010, 81, 961–968.
- 5.
Lee, H.H.; Lee, S.; Lee, K.; et al. Anti-cancer Effect of Cordyceps militaris in Human Colorectal Carcinoma RKO Cells via Cell Cycle Arrest and Mitochondrial Apoptosis. Daru 2015, 23, 35.
- 6.
Tuli, H.S.; Sharma, A.K.; Sandhu, S.S.; et al. Cordycepin: A Bioactive Metabolite with Therapeutic Potential. Life Sci. 2013, 93, 863–869.
- 7.
Xu, X.; Yan, H.; Chen, J.; et al. Bioactive Proteins from Mushrooms. Biotechnol. Adv. 2011, 29, 667–674.
- 8.
Magnusson, B.; Örnemark, U. The Fitness for Purpose of Analytical Methods—A Laboratory Guide to Method Validation and Related Topics, 2nd ed.; Eurachem: Bucharest, Romania, 2014.
- 9.
Mosmann, T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J. Immunol. Methods 1983, 65, 55–63.
- 10.
Shi, R.; Chen, Z.; Fan, W.; et al. Research on the physicochemical and digestive properties of Pleurotus eryngii protein. Int. J. Food Prop. 2018, 21, 2785–2806.
- 11.
Yoon, S.Y.; Park, S.J.; Park, Y.J. The Anticancer Properties of Cordycepin and Their Underlying Mechanisms. Int. J. Mol. Sci. 2018, 19, 3027.
- 12.
Tian, X.; Li, Y.; Shen, Y.; et al. Apoptosis and inhibition of proliferation of cancer cells induced by cordycepin. Oncol Lett 2015, 10, 595–599.
- 13.
Hwang, J.H.; Park, S.J.; Ko, W.G.; et al. Cordycepin induces human lung cancer cell apoptosis by inhibiting nitric oxide mediated ERK/Slug signaling pathway. Am. J. Cancer Res. 2017, 7, 417–432.

This work is licensed under a Creative Commons Attribution 4.0 International License.


