Arsenic is a highly toxic element found in liquid effluents and can pose serious health and environmental risks. Conversely, coconut fiber, an abundant byproduct of the agrifood industry, has strong potential as an adsorbent for removing arsenic from water. Its lignocellulosic structure, which includes cellulose, hemicellulose, lignin, and functional groups such as hydroxyls and carbonyls, enhances interactions with various arsenic species. Research indicates that both raw fiber and chemically treated fiber, using methods such as acid, alkali, biochar conversion, or metal/oxide impregnation, can remove contaminants like dyes, heavy metals, and arsenic. This review shows that chemical treatments significantly improve adsorption efficiency, reaching up to 99% arsenic removal, whereas raw fibers typically achieve less than 25%. Factors such as pH, temperature, and contact time directly influence process effectiveness, with neutral or slightly acidic pH and temperatures near room temperature being ideal for better removal. Kinetic and isotherm models, including the Langmuir and the Freundlich models, help elucidate adsorption mechanisms and estimate the maximum biosorbent capacity. Additionally, coconut fiber can be regenerated and reused after adsorption and desorption cycles, making the process potentially sustainable and cost-effective. Despite promising results with synthetic solutions, challenges remain for real-world applications, such as in industrial effluents and groundwater, due to the presence of competing ions and organic matter. Developing technologies based on coconut fiber promotes the valorization of agro-industrial waste, supports green chemistry and the circular economy by enabling the reuse of low-cost, abundant materials, minimizes environmental impacts, and fosters sustainable treatment of arsenic-contaminated water.
- Open Access
- Mini Review
Arsenic Removal from Aqueous Solutions Using Coconut Fiber (Cocos nucifera): A Brief Review
- Cris L. B. S. Lima 1,
- Luiz A. M. Pontes 1,2,
- Valfredo A. Lemos 3,4,
- Sérgio L. C. Ferreira 4,5,
- Leonardo S. G. Teixeira 4,5,*
Author Information
Received: 29 Oct 2025 | Revised: 16 Feb 2026 | Accepted: 25 Feb 2026 | Published: 06 Mar 2026
Abstract
Graphical Abstract
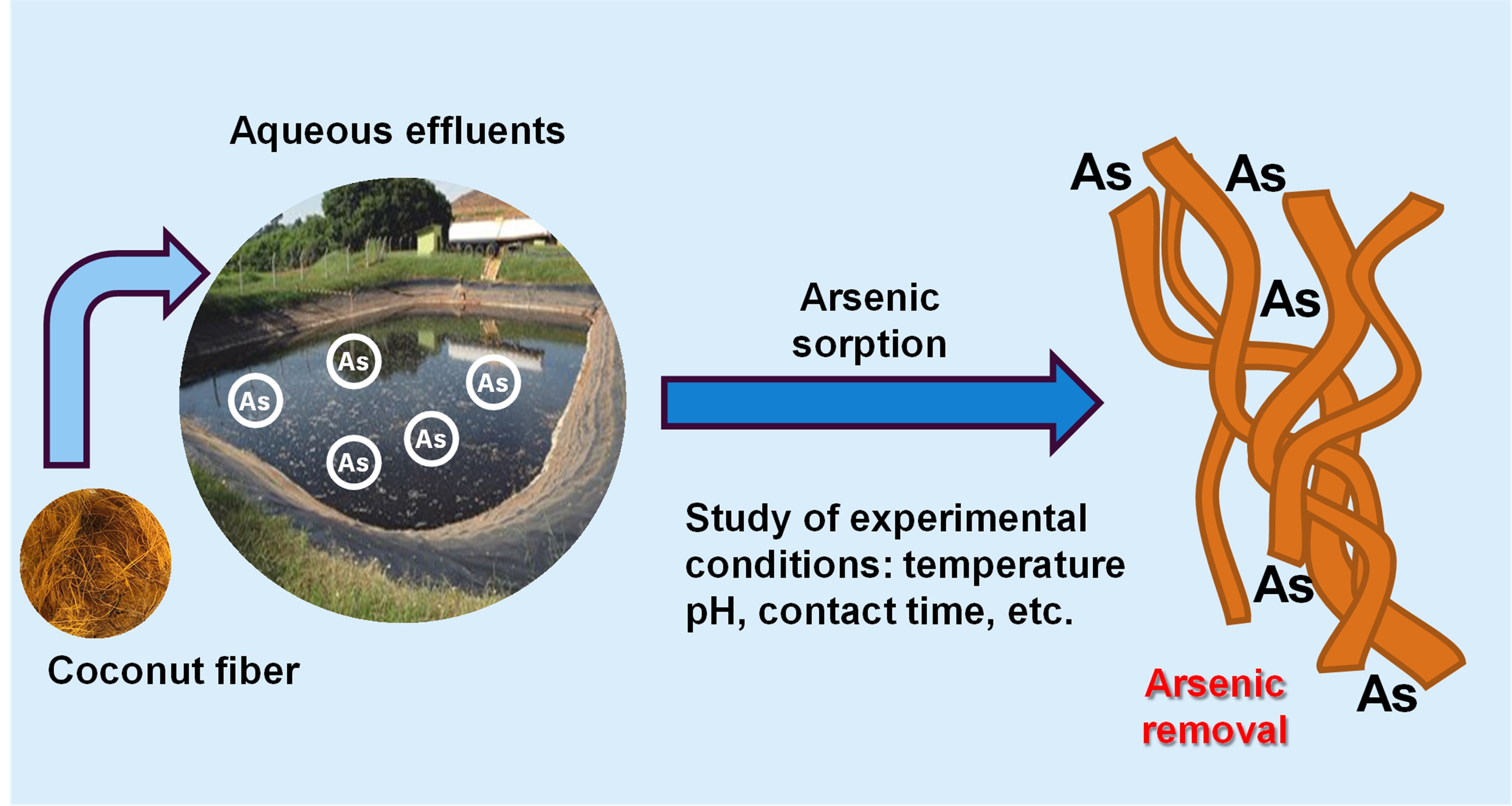
Keywords
bioremediation | lignocellulosic materials | inorganic contaminants | biomass | solid waste | liquid effluents
References
- 1.
Soudani, A.; Youcef, L.; Chebbi, M.; et al. Agricultural waste-based biochars for sustainable removal of heavy metals from stabilized landfill leachate. Environ. Sci. Pollut. Res. Int. 2024, 31, 57733–57747. https://doi.org/10.1007/s11356-024-34946-8.
- 2.
Ni, H.; Arslan, M.; Qian, J.; et al. Application of basalt fibers in a biological contact oxidation reactor for the treatment of landfill leachate. J. Clean. Prod. 2021, 297, 126648. https://doi.org/10.1016/j.jclepro.2021.126648.
- 3.
Maia, L.C.; Soares, L.C.; Alves Gurgel, L.V. A review on the use of lignocellulosic materials for arsenic adsorption. J. Environ. Manag. 2021, 288, 112397. https://doi.org/10.1016/j.jenvman.2021.112397.
- 4.
Ge, Y.; Li, Z. Application of Lignin and Its Derivatives in Adsorption of Heavy Metal Ions in Water: A Review. ACS Sustain. Chem. Eng. 2018, 6, 7181–7192. https://doi.org/10.1021/acssuschemeng.8b01345.
- 5.
Khaskheli, M.I.; Memon, S.Q.; Siyal, A.N.; et al. Use of Orange Peel Waste for Arsenic Remediation of Drinking Water. Waste Biomass Valorization 2011, 2, 423–433. https://doi.org/10.1007/s12649-011-9081-7.
- 6.
Brainer, M.S.C.P.; Ximenes, L.F. Coconut Production: Recovery of Traditional Growing Areas in Northeast Brazil; Technical Report No. 127; Bank of the Northeast of Brazil (BNB): Fortaleza, Brazil, 2020. (In Portuguese). Available online: https://www.bnb.gov.br/s482-dspace/handle/123456789/387 (accessed on 3 August 2025).
- 7.
Cardoso, M.G.S.; de Oliveira, D.N.P.S.; Bufalino, L.; et al. Physical and chemical treatments of residual fibers from Cocos nucifera L. aiming application in cement composites. Res. Soc. Dev. 2022, 11, e57311831259. https://doi.org/10.33448/rsd-v11i8.31259.
- 8.
Suleman, R.; Zahoor, M.A.; Qarni, M.A.; et al. Assessment of heavy metals and microbial loads in Nile tilapia (Oreochromis niloticus) from different farms and rivers. Sci. Rep. 2025, 15, 5055. https://doi.org/10.1038/s41598-025-87152-y.
- 9.
Berslin, D.; Reshmi, A.; Sivaprakash, B.; et al. Remediation of emerging metal pollutants using environment friendly biochar- Review on applications and mechanism. Chemosphere 2022, 290, 133384. https://doi.org/10.1016/j.chemosphere.2021.133384.
- 10.
Raj, K.; Vora, T.; Padmapriya, G.; et al. A comprehensive review of sustainable hydrogels from lignin for advanced wastewater solutions. Int. J. Biol. Macromol. 2025, 301, 139963. https://doi.org/10.1016/j.ijbiomac.2025.139963.
- 11.
Wae AbdulKadir, W.A.F.; Che Omar, R.; Roslan, R.; et al. Landfill leachate treatment in Malaysia: Continuous circulation motion using mixed agricultural wastes with an open grid-like Luffa's configuration. J. Water Process Eng. 2023, 56, 104532. https://doi.org/10.1016/j.jwpe.2023.104532.
- 12.
Li, X.; Shi, J.; Luo, X. Enhanced adsorption of rhodamine B from water by Fe-N co-modified biochar: Preparation, performance, mechanism and reusability. Bioresour. Technol. 2022, 343, 126103. https://doi.org/10.1016/j.biortech.2021.126103.
- 13.
Zakaria, W.A.N.; Mohd Ghazi, R.; Muhammad, M.; et al. Optimization of Malachite green removal using activated carbon derived from coconut shell. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2021; Volume 842, p. 012033. https://doi.org/10.1088/1755-1315/842/1/012033.
- 14.
Bokil, S.A.; Topare, N.S.; Khedkar, S.V. Batch adsorption studies on treatment of textile industry effluent using bamboo and green coconut shell activated carbon. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2020; Volume 983, p. 012005. https://doi.org/10.1088/1757-899X/983/1/012005.
- 15.
Wang, S.; Zhong, S.; Zheng, X.; et al. Calcite modification of agricultural waste biochar highly improves the adsorption of Cu(II) from aqueous solutions. J. Environ. Chem. Eng. 2021, 9, 106215. https://doi.org/10.1016/j.jece.2021.106215.
- 16.
Wu, J.; Wang, T.; Wang, J.; et al. A novel modified method for the efficient removal of Pb and Cd from wastewater by biochar: Enhanced the ion exchange and precipitation capacity. Sci. Total Environ. 2021, 754, 142150. https://doi.org/10.1016/j.scitotenv.2020.142150.
- 17.
Kamran, U.; Park, S.-J. Hybrid biochar supported transition metal doped MnO2 composites: Efficient contenders for lithium adsorption and recovery from aqueous solutions. Desalination 2022, 522, 115387. https://doi.org/10.1016/j.desal.2021.115387.
- 18.
Lazim, Z.M.; Salmiati; Hadibarata, T.; et al. Bisphenol A Removal by Adsorption Using Waste Biomass: Isotherm and Kinetic Studies. Biointerface Res. Appl. Chem. 2021, 11, 8467–8481. https://doi.org/10.33263/briac111.84678481.
- 19.
Esfandiar, N.; Suri, R.; McKenzie, E.R. Competitive sorption of Cd, Cr, Cu, Ni, Pb and Zn from stormwater runoff by five low-cost sorbents; Effects of co-contaminants, humic acid, salinity and pH. J. Hazard. Mater. 2022, 423, 126938. https://doi.org/10.1016/j.jhazmat.2021.126938.
- 20.
Chwastowski, J.; Staroń, P. Influence of Saccharomyces cerevisiae yeast cells immobilized on Cocos nucifera fibers for the adsorption of Pb(II) ions. Colloids Surf. A Physicochem. Eng. Asp. 2022, 632, 127735. https://doi.org/10.1016/j.colsurfa.2021.127735.
- 21.
Bal, D.K.; Bhasarkar, J.B. Adsorptive degradation of hexavalent chromium from aqueous solution using coconut shell as a green adsorbent. Environ. Prog. Sustain. Energy 2021, 40, e13594. https://doi.org/10.1002/ep.13594.
- 22.
Hanafiah, S.; Salleh, N.; Ghafar, N.; et al. Efficiency of coconut husk as agricultural adsorbent in removal of chromium and nickel ions from aqueous solution. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2020; Volume 596, p. 012048. https://doi.org/10.1088/1755-1315/596/1/012048.
- 23.
Thinh, N.V.; Yusuke, S.; Tu, P.H.; et al. Novel co-doped iron oxide and graphitic carbon nanosheets on biochar for arsenite removal from contaminated water: Synthesis, applicability and mechanism. Bioresour. Technol. Rep. 2022, 17, 100929. https://doi.org/10.1016/j.biteb.2021.100929.
- 24.
Gautam, S.B.; Alam, M.S.; Kamsonlian, S. Adsorptive Removal of As(III) from Aqueous Solution by Raw Coconut Husk and Iron Impregnated Coconut Husk: Kinetics and Equilibrium Analyses. Int. J. Chem. React. Eng. 2017, 15, 20160097. https://doi.org/10.1515/ijcre-2016-0097.
- 25.
Raut, E.R.; Thakur, A.B.; Chaudhari, A.R. Review on toxic metal ions removal by using activated carbon prepared from natural biomaterials. In Journal of Physics: Conference Series; IOP Publishing: Bristol, UK, 2021; Volume 1913, p. 012091. https://doi.org/10.1088/1742-6596/1913/1/012091.
- 26.
Fernando, J.A.K.M.; Yalegama, C.; Fernando, N.; et al. Characterization of the coir fiber from different coconut varieties to enhance the performance of coir fiber-reinforced natural rubber composites. J. Elastomers Plast. 2025, 57, 475–497. https://doi.org/10.1177/00952443251323933.
- 27.
Chand, O.; Chopra, L.; Raju, N.N.; et al. Extraction of micro fibrous cellulose from coconut husk by using chlorine free process: Potential utilization application as a filter aid. J. Mol. Struct. 2025, 1319, 139325. https://doi.org/10.1016/j.molstruc.2024.139325.
- 28.
Hidalgo-Salazar, M.A.; Correa-Aguirre, J.P.; Roman, A.J.; et al. Colombian natural fibers: Potential applications in sustainable natural fiber reinforced composites materials. Polym. Compos. 2025, 46, 5599–5617. https://doi.org/10.1002/pc.29313.
- 29.
Schiavon, J.Z.; Andrade, J.J.d.O. Comparison between alternative chemical treatments on coir fibers for application in cementitious materials. J. Mater. Res. Technol. 2023, 25, 4634–4649. https://doi.org/10.1016/j.jmrt.2023.06.210.
- 30.
Fauzi, W.W.; Fairuz, F.M.; Shukri, N.M.; et al. Removal of As and Cd Ions from Aqueous Solution Using Biosorption Technique. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2022; Volume 1102, p. 012088. https://doi.org/10.1088/1755-1315/1102/1/012088.
- 31.
Bruckmann, F.S.; Gonçalves, J.O.; Silva, L.F.O.; et al. Chitosan-based adsorbents for wastewater treatment: A comprehensive review. Int. J. Biol. Macromol. 2025, 309, 143173. https://doi.org/10.1016/j.ijbiomac.2025.143173.
- 32.
Ferreira, V.F.; da Rocha, D.R.; da Silva, F.d C. Potentiality and opportunity in the chemistry of sucrose and other sugars. Quim. Nova 2009, 32, 623–638. https://doi.org/10.1590/s0100-40422009000300007.
- 33.
Bezerra, P.K.S.B.; Silva, O.L.; Júnior, S.D.O.; et al. Cellulolytic enzymes behavior in delignified green coconut residues and enzymatic hydrolysis with enzyme recovery. Ind. Crop. Prod. 2021, 172, 114037. https://doi.org/10.1016/j.indcrop.2021.114037.
- 34.
Rahim, A.R.A.; Mohsin, H.M.; Chin, K.B.L.; et al. Promising Low-cost Adsorbent from Desiccated Coconut Waste for Removal of Congo Red Dye from Aqueous Solution. Water Air Soil Pollut. 2021, 232, 357. https://doi.org/10.1007/s11270-021-05308-8.
- 35.
Ghosh, I.; Kar, S.; Chatterjee, T.; et al. Adsorptive removal of Safranin-O dye from aqueous medium using coconut coir and its acid-treated forms: Adsorption study, scale-up design, MPR and GA-ANN modeling. Sustain. Chem. Pharm. 2021, 19, 100374. https://doi.org/10.1016/j.scp.2021.100374.
- 36.
Chong, S.N.; Hadibarata, T. Adsorption of Phenol Red and Remazol Brilliant Blue R by Coconut Shells (Cocos nucifera) and Ambarella Peels (Spondias dulcis). Biointerface Res. Appl. Chem. 2020, 11, 8564–8576. https://doi.org/10.33263/BRIAC111.85648576.
- 37.
Droepenu, E.K.; Asare, E.A.; Dampare, S.B.; et al. Laboratory and Commercial Synthesized Zinc Oxide Nanoparticles Adsorption onto Coconut Husk: Characterization. Biointerface Res. Appl. Chem. 2021, 11, 7871–7889. https://doi.org/10.33263/BRIAC111.78717889.
- 38.
Mustapha, R.; Harun, M.H.C.; Manas, A.; et al. Preparation and Characterization of Bio-Adsorbent from Coconut Husk for Remazol Red Dye Removal. Biointerface Res. Appl. Chem. 2021, 11, 10006–10015. https://doi.org/10.33263/briac113.1000610015.
- 39.
He, R.; Peng, Z.; Lyu, H.; et al. Synthesis and characterization of an iron-impregnated biochar for aqueous arsenic removal. Sci. Total Environ. 2018, 612, 1177–1186. https://doi.org/10.1016/j.scitotenv.2017.09.016.
- 40.
Srivastava, S.; Raj, K.R.; Kardam, A. Efficient arsenic depollution in water using modified maize powder. Environ. Chem. Lett. 2013, 11, 47–53. https://doi.org/10.1007/s10311-012-0376-0.
- 41.
Kamsonlian, S.; Suresh, S.; Ramanaiah, V.; et al. Biosorptive behaviour of mango leaf powder and rice husk for arsenic(III) from aqueous solutions. Int. J. Environ. Sci. Technol. 2012, 9, 565–578. https://doi.org/10.1007/s13762-012-0054-6.
- 42.
Shafique, U.; Ijaz, A.; Salman, M.; et al. Removal of arsenic from water using pine leaves. J. Taiwan Inst. Chem. Eng. 2012, 43, 256–263. https://doi.org/10.1016/j.jtice.2011.10.006.
- 43.
Verma, L.; Siddique, M.A.; Singh, J.; et al. As(III) and As(V) removal by using iron impregnated biosorbents derived from waste biomass of Citrus limmeta (peel and pulp) from the aqueous solution and ground water. J. Environ. Manag. 2019, 250, 109452. https://doi.org/10.1016/j.jenvman.2019.109452.
- 44.
Wu, Y.; Wen, Y.; Zhou, J.; et al. Comparative and competitive adsorption of Cr(VI), As(III), and Ni(II) onto coconut charcoal. Environ. Sci. Pollut. Res. 2013, 20, 2210–2219. https://doi.org/10.1007/s11356-012-1066-y.
- 45.
Nashine, A.L.; Tembhurkar, A.R. Equilibrium, kinetic and thermodynamic studies for adsorption of As (III) on coconut (Cocos nucifera L.) fiber. J. Environ. Chem. Eng. 2016, 4, 3267–3273. https://doi.org/10.1016/j.jece.2016.06.005.
- 46.
Mon, P.P.; Cho, P.P.; Vidyasagar, D.; et al. Synergistic sorption: Enhancing arsenic (V) removal using biochar decorated with cerium oxide composite. Mater. Today Sustain. 2024, 25, 100675. https://doi.org/10.1016/j.mtsust.2024.100675.
- 47.
Lima, C.L.B.S.; Moreira, I.T.A.; Campos, L.M.A.; et al. Removal of arsenic from landfill leachate using green coconut fiber. Sci. Rep. 2025, 15, 37064. https://doi.org/10.1038/s41598-025-20861-6.
- 48.
Priyadharshini, B.; Abraham, M.; Nirmala, R.; et al. Optimization Study of Adsorption parameter for the removal of Zinc (Zn) and Arsenic (As) on Activated Coconut Shell (ACS) Adsorbent using Response Surface Methodology. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2024; Volume 1365, p. 012008. https://doi.org/10.1088/1755-1315/1365/1/012008.
- 49.
Anirudhan, T.S.; Unnithan, M.R. Arsenic(V) removal from aqueous solutions using an anion exchanger derived from coconut coir pith and its recovery. Chemosphere 2007, 66, 60–66. https://doi.org/10.1016/j.chemosphere.2006.05.031.
- 50.
Jimenez, R.S.; Dal Bosco, S.M.; Carvalho, W.A. Heavy metals removal from wastewater by the natural zeolite scolecite—Temperature and pH influence in single-metal solutions. Quim. Nova 2004, 27, 734–738. https://doi.org/10.1590/s0100-40422004000500011.

This work is licensed under a Creative Commons Attribution 4.0 International License.


