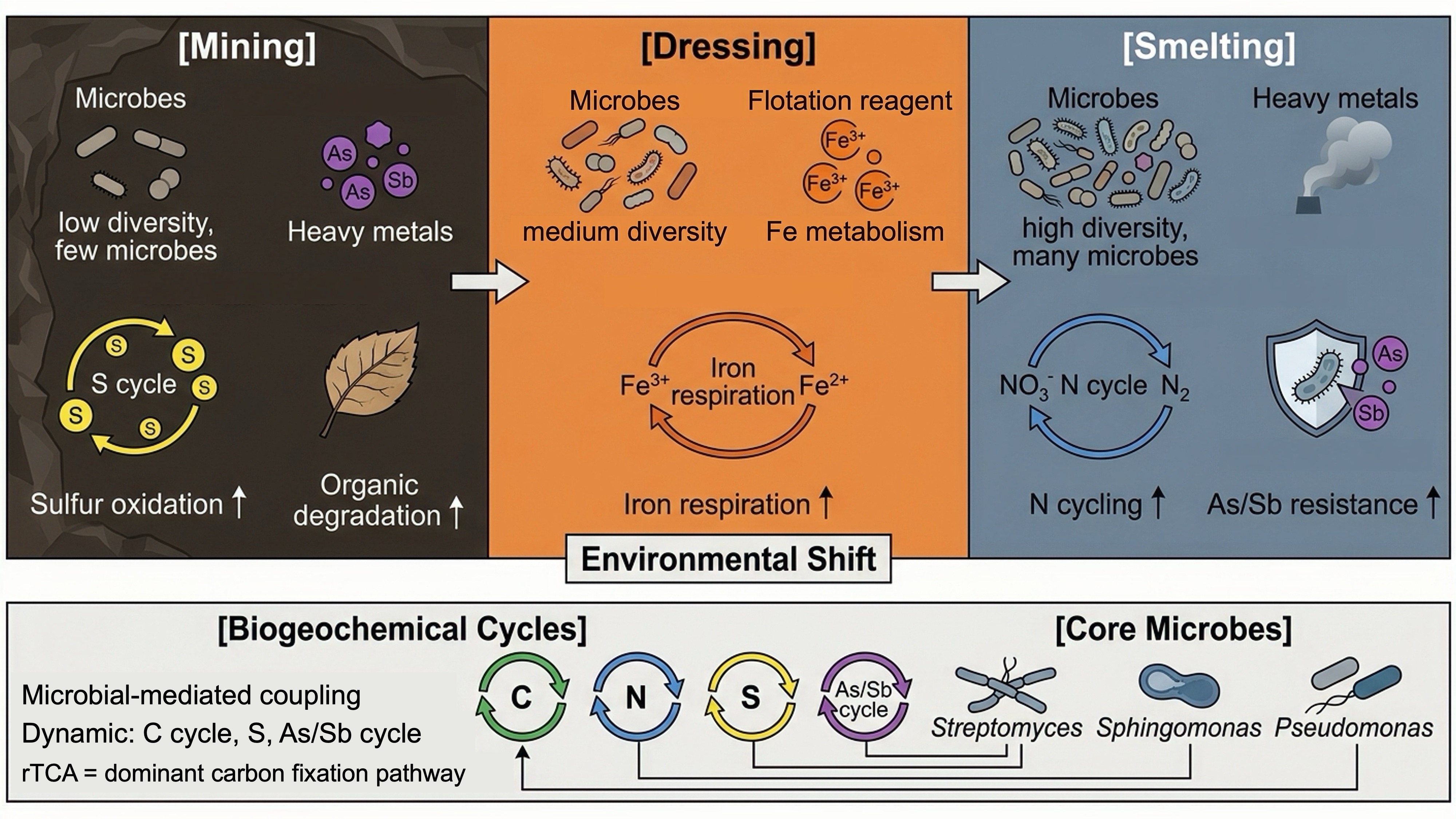
- Mining soils showed the lowest diversity and stronger sulfur oxidation.
- Dressing soils were characterized by enhanced iron-related respiration.
- Smelting soils enriched genes for N cycling, S metabolism and As/Sb resistance.
- Core taxa linked C, N, S and As/Sb cycling across all processing stages.
- Open Access
- Article
Microbial Response to Mining, Dressing and Smelting Activities in Antimony-Mining Area: Insights into C, N, S and As Cycling
- Weisong Pan 1,
- Boyan Zhang 1,
- Chuan Wu 2,3,
- Yayuan Huang 2,*,
- Xiaoran Xiong 1,
- Yahui Wu 2,
- Tsang Po Keung 3,
- Waichin Li 3,*
Author Information
Received: 22 Dec 2025 | Revised: 26 Mar 2026 | Accepted: 17 Apr 2026 | Published: 15 May 2026
Highlights
Abstract
Arsenic (As) and antimony (Sb) are toxic metalloids with similar physicochemical properties, and their microbial transformation is closely linked to carbon (C), nitrogen (N), and sulfur (S) cycling. Mining-related anthropogenic activities substantially alter the bioavailability of As/Sb and microbial metabolism, but the specific impacts of sequential mining, dressing, and smelting processes on microbial communities and C, N, S, and As/Sb cycling remain unclear. In this study, we systematically investigated soil physicochemical properties, microbial communities, and functional potentials in an abandoned Sb mine area. The results showed that different mineral processing stages significantly altered soil nutrient status and heavy metal distribution, and induced distinct shifts in microbial diversity, composition, and metabolic potential. The mining area exhibited the lowest bacterial α-diversity, whereas the smelting area supported the highest diversity. Metal-tolerant taxa, including Proteobacteria, Actinobacteria, and Firmicutes, were enriched in the mining area, with dominant genera such as Pseudomonas, Bacillus, and Bradyrhizobium. Functional analyses indicated clear stage-specific differentiation of microbial metabolism: the dressing area was characterized by enhanced iron-related respiratory potential, the smelting area by increased methyl compound utilization, and the mining area by stronger sulfur oxidation and organic matter degradation potential. Metagenomic analysis further revealed that the reductive citric acid cycle was the dominant carbon fixation pathway, while smelting activities increased the relative abundance of genes involved in nitrate transformation, sulfur metabolism, and As/Sb resistance. Core taxa including Streptomyces, Sphingopyxis, Sphingomonas, and Pseudomonas were broadly associated with C, N, S, and As/Sb cycling across all sites. This study provides new insights into how sequential mining, dressing, and smelting activities shape microbial community assembly and biogeochemical functions in metalloid-contaminated soils. The findings have broader relevance for understanding microbial adaptation and for developing site-specific bioremediation strategies in similar mining-impacted environments worldwide.
Graphical Abstract
Keywords
mining | dressing and smelting | metagenome | arsenic | antimony
References
- 1.
He, M.; Wang, N.; Long, X.; et al. Antimony Speciation in the Environment: Recent Advances in Understanding the Biogeochemical Processes and Ecological Effects. J. Environ. Sci. 2019, 75, 14–39.
- 2.
Zhu, Y.; Yang, J.; Wang, L.; et al. Factors Influencing the Uptake and Speciation Transformation of Antimony in the Soil‑Plant System, and the Redistribution and Toxicity of Antimony in Plants. Sci. Total Environ. 2020, 738, 140232.
- 3.
Long, J.; Tan, D.; Deng, S.; et al. Antimony Accumulation and Iron Plaque Formation at Different Growth Stages of Rice (Oryza sativa L.). Environ. Pollut. 2019, 249, 414–422.
- 4.
Ren, J.; Ma, L.Q.; Sun, H.; et al. Antimony Uptake, Translocation and Speciation in Rice Plants Exposed to Antimonite and Antimonate. Sci. Total Environ. 2014, 475, 83–89.
- 5.
Deng, J.; Xiao, T.; Fan, W.; et al. Relevance of the Microbial Community to Sb and As Biogeochemical Cycling in Natural Wetlands. Sci. Total Environ. 2022, 818, 151826.
- 6.
Hao, C.; Sun, X.; Peng, Y.; et al. Geochemical Impact of Dissolved Organic Matter on Antimony Mobilization in Shallow Groundwater of the Xikuangshan Antimony Mine, Hunan Province, China. Sci. Total Environ. 2023, 860, 160292.
- 7.
Wu, T.; Zhang, N.; Liu, C.; et al. Factors Driving Antimony Accumulation in Soil-Pakchoi and Wheat Agroecosystems: Insights and Predictive Models. Environ. Pollut. 2024, 351, 124016.
- 8.
Pan, W.; Zou, Q.; Hu, M.; et al. Microbial Community Composition and Co‑Occurrence Patterns Driven by Co-Contamination of Arsenic and Antimony in Antimony-Mining Area. J. Hazard. Mater. 2023, 454, 131535.
- 9.
Yu, H.; Liu, S.; Weng, W.; et al. Generational Specific Recruitment of Arsenic- and Antimony-Reducing Microorganisms in Plant Root‑Associated Niches for Adapting to Metalloid‑Metal Pollution. Environ. Sci. Technol. 2024, 58, 19567–19578.
- 10.
Tang, S.; Song, X.; Chen, J.; et al. Widespread Distribution of the arsO Gene Confers Bacterial Resistance to Environmental Antimony. Environ. Sci. Technol. 2023, 57, 14579–14588.
- 11.
Liu, B.; Yao, J.; Chen, Z.; et al. Biogeography, Assembly Processes and Species Coexistence Patterns of Microbial Communities in Metalloids-Laden Soils around Mining and Smelting Sites. J. Hazard. Mater. 2022, 425, 127945.
- 12.
Wang, N.; Wang, A.; Xie, J.; et al. Responses of Soil Fungal and Archaeal Communities to Environmental Factors in an Ongoing Antimony Mine Area. Sci. Total Environ. 2019, 652, 1030–1039.
- 13.
Li, B.; Xu, R.; Sun, X.; et al. Microbiome–Environment Interactions in Antimony-Contaminated Rice Paddies and the Correlation of Core Microbiome with Arsenic and Antimony Contamination. Chemosphere 2021, 263, 128227.
- 14.
Lin, H.; Qin, K.; Dong, Y.; et al. A Newly-Constructed Bifunctional Bacterial Consortium for Removing Butyl Xanthate and Cadmium Simultaneously from Mineral Processing Wastewater: Experimental Evaluation, Degradation and Biomineralization. J. Environ. Manage. 2022, 316, 115304.
- 15.
Bararunyeretse, P.; Zhang, Y.; Ji, H. Molecular Biology‑Based Analysis of the Interactive Effect of Nickel and Xanthates on Soil Bacterial Community Diversity and Structure. Sustainability 2019, 11, 3888.
- 16.
Li, H.; Yao, J.; Min, N.; et al. New Insights on the Effect of Non-Ferrous Metal Mining and Smelting Activities on Microbial Activity Characteristics and Bacterial Community Structure. J. Hazard. Mater. 2023, 453, 131301.
- 17.
Luo, Y.; Xing, R.; Wan, Z.; et al. Vertical Distribution of Nutrients, Enzyme Activities, Microbial Properties, and Heavy Metals in Zinc Smelting Slag Site Revegetated with Two Herb Species: Implications for Direct Revegetation. Sci. Total Environ. 2023, 879, 163206.
- 18.
Zhao, X.; Huang, J.; Lu, J.; et al. Study on the Influence of Soil Microbial Community on the Long-Term Heavy Metal Pollution of Different Land Use Types and Depth Layers in Mine. Ecotoxicol. Environ. Saf. 2019, 170, 218–226.
- 19.
Li, Y.; Zhang, M.; Xu, R.; et al. Arsenic and Antimony Co‑Contamination Influences on Soil Microbial Community Composition and Functions: Relevance to Arsenic Resistance and Carbon, Nitrogen, and Sulfur Cycling. Environ. Int. 2021, 153, 106522.
- 20.
Li, Y.; Lin, H.; Gao, P.; et al. Synergistic Impacts of Arsenic and Antimony Co-Contamination on Diazotrophic Communities. Microb. Ecol. 2022, 84, 44–58.
- 21.
Sun, W.; Sun, X.; Li, B.; et al. Bacterial Response to Sharp Geochemical Gradients Caused by Acid Mine Drainage Intrusion in a Terrace: Relevance of C, N, and S Cycling and Metal Resistance. Environ. Int. 2020, 138, 105601.
- 22.
Li, Y.; Guo, L.; Haggblom, M.M.; et al. Serratia spp. Are Responsible for Nitrogen Fixation Fueled by As(III) Oxidation, a Novel Biogeochemical Process Identified in Mine Tailings. Environ. Sci. Technol. 2022, 56, 2033–2043.
- 23.
Liang, J.L.; Liu, J.; Jia, P.; et al. Novel Phosphate-Solubilizing Bacteria Enhance Soil Phosphorus Cycling Following Ecological Restoration of Land Degraded by Mining. ISME J. 2020, 14, 1600–1613.
- 24.
Zou, Q.; An, W.; Wu, C.; et al. Red Mud-Modified Biochar Reduces Soil Arsenic Availability and Changes Bacterial Composition. Environ. Chem. Lett. 2018, 16, 615–622.
- 25.
Zou, Q.; Wei, H.; Chen, Z.; et al. Soil Particle Size Fractions Affect Arsenic (As) Release and Speciation: Insights into Dissolved Organic Matter and Functional Genes. J. Hazard. Mater. 2023, 443, 130100.
- 26.
Huang, D.; Zhou, L.; Fan, H.; et al. Responses of Aggregates and Associated Soil Available Phosphorus, and Soil Organic Matter in Different Slope Aspects, to Seasonal Freeze-Thaw Cycles in Northeast China. Geoderma 2021, 402, 115184.
- 27.
Wu, Z.; Yang, X.; Huang, L.; et al. In Situ Enrichment of Sulphate-Reducing Microbial Communities with Different Carbon Sources Stimulating the Acid Mine Drainage Sediments. Sci. Total Environ. 2023, 898, 165584.
- 28.
Filella, M.; Belzile, N.; Chen, Y.-W. Antimony in the Environment: A Review Focused on Natural Waters I. Occurrence. Earth Sci. Rev. 2002, 57, 125–176.
- 29.
Wilson, S.C.; Lockwood, P.V.; Ashley, P.M.; et al. The Chemistry and Behaviour of Antimony in the Soil Environment with Comparisons to Arsenic: A Critical Review. Environ. Pollut. 2010, 158, 1169–1181.
- 30.
Leuz, A.-K.; Johnson, C.A. Oxidation of Sb(III) to Sb(V) by O2 and H2O2 in Aqueous Solutions. Geochim. Cosmochim. Acta 2005, 69, 1165–1172.
- 31.
Li, J.; Zheng, Q.; Liu, J.; et al. Bacterial-Fungal Interactions and Response to Heavy Metal Contamination of Soil. Front. Microbiol. 2024, 15, 1395154.
- 32.
Zeng, K.; Huang, X.; Guo, J.; et al. Microbial-Driven Mechanisms for the Effects of Heavy Metals on Soil Organic Carbon Storage: A Global Analysis. Environ. Int. 2024, 190, 108467.
- 33.
Zhao, X.; Huang, J.; Zhu, X.; et al. Ecological Effects of Heavy Metal Pollution on Soil Microbial Community Structure and Diversity on both Sides of a River around a Mining Area. Int. J. Environ. Res. Public. Health. 2020, 17, 5680.
- 34.
Chen, Q.; Ding, J.; Zhu, D.; et al. Rare Microbial Taxa as the Major Drivers of Ecosystem Multifunctionality in Long‑Term Fertilized Soils. Soil Biol. Biochem. 2020, 141, 107686.
- 35.
Gong, Y.; Yang, S.; Chen, S.; et al. Soil Microbial Responses to Simultaneous Contamination of Antimony and Arsenic in the Surrounding Area of an Abandoned Antimony Smelter in Southwest China. Environ. Int. 2023, 174, 107897.
- 36.
Philippot, L.; Chenu, C.; Kappler, A.; et al. The Interplay between Microbial Communities and Soil Properties. Nat. Rev. Microbiol. 2024, 22, 226–239.
- 37.
Okkenhaug, G.; Zhu, Y.; He, J.; et al. Antimony (Sb) and Arsenic (As) in Sb Mining Impacted Paddy Soil from Xikuangshan, China: Differences in Mechanisms Controlling Soil Sequestration and Uptake in Rice. Environ. Sci. Technol. 2012, 46, 3155–3162.
- 38.
Yin, Y.; Wang, X.; Hu, Y.; et al. Soil Bacterial Community Structure in the Habitats with Different Levels of Heavy Metal Pollution at an Abandoned Polymetallic Mine. J. Hazard. Mater. 2023, 442, 130063.
- 39.
Diepens, N.J.; Dimitrov, M.R.; Koelmans, A.A.; et al. Molecular Assessment of Bacterial Community Dynamics and Functional End Points during Sediment Bioaccumulation Tests. Environ. Sci. Technol. 2015, 49, 13586–13595.
- 40.
Fakhar, A.; Gul, B.; Gurmani, A.R.; et al. Heavy Metal Remediation and Resistance Mechanism of Aeromonas, Bacillus, and Pseudomonas: A Review. Crit. Rev. Environ. Sci. Technol. 2022, 52, 1868–1914.
- 41.
Yin, K.; Lv, M.; Wang, Q.; et al. Simultaneous Bioremediation and Biodetection of Mercury Ion through Surface Display of Carboxylesterase E2 from Pseudomonas aeruginosa PA1. Water Res. 2016, 103, 383–390.
- 42.
Reichman, S.M. Probing the Plant Growth-Promoting and Heavy Metal Tolerance Characteristics of Bradyrhizobium japonicum CB1809. Eur. J. Soil Biol. 2014, 63, 7–13.
- 43.
Chen, X.; Wang, J.; Pan, C.; et al. Metagenomic Analysis Reveals the Response of Microbial Community in River Sediment to Accidental Antimony Contamination. Sci. Total Environ. 2022, 813, 152484.
- 44.
Sun, W.; Sun, X.; Li, B.; et al. Bacterial Response to Antimony and Arsenic Contamination in Rice Paddies during Different Flooding Conditions. Sci. Total Environ. 2019, 675, 273–285.
- 45.
He, Q.; Liu, Y.; Wan, D.; et al. Enhanced Biological Antimony Removal from Water by Combining Elemental Sulfur Autotrophic Reduction and Disproportionation. J. Hazard. Mater. 2022, 434, 128926.

This work is licensed under a Creative Commons Attribution 4.0 International License.


