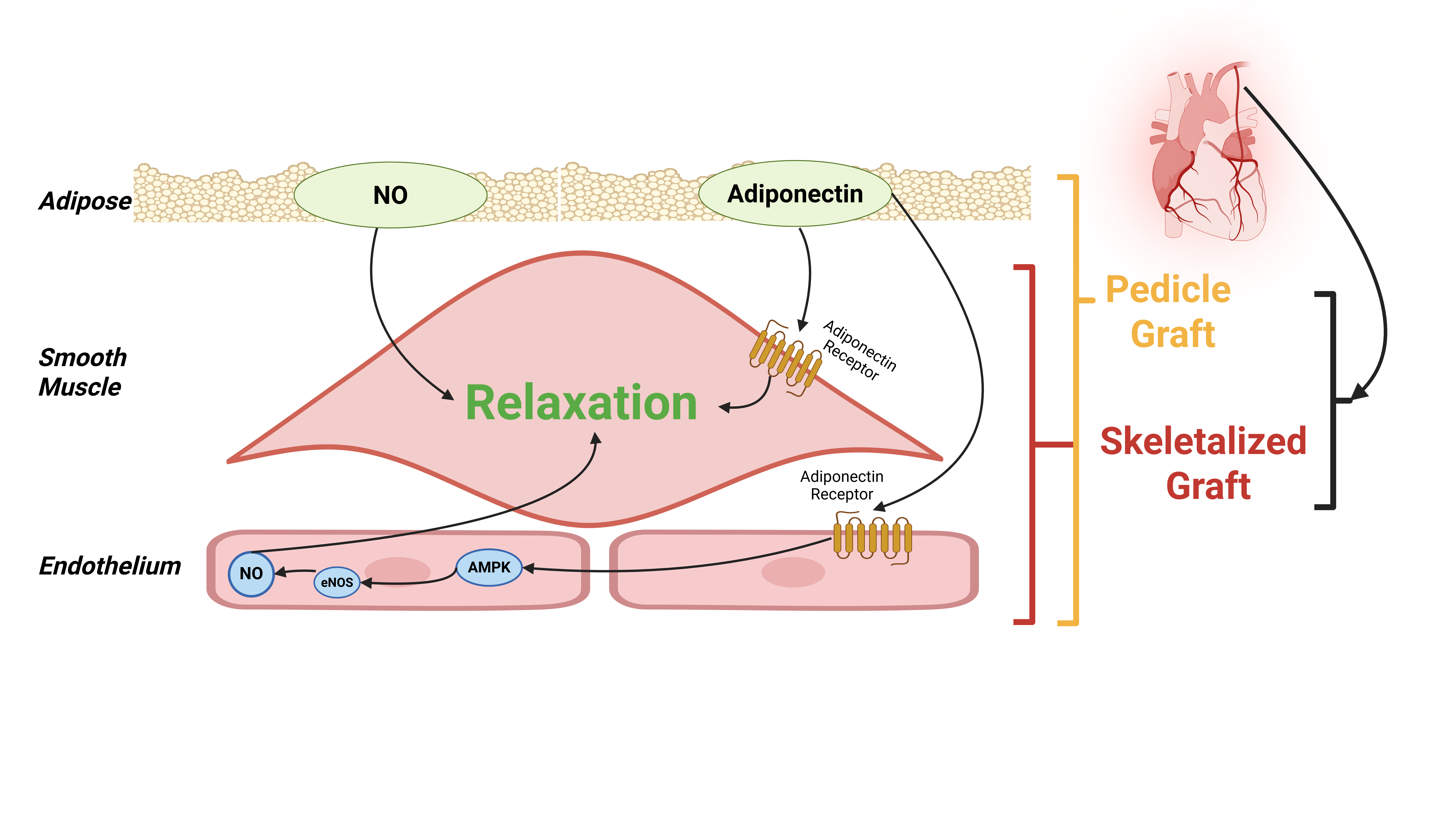
Background: The internal mammary artery (IMA) is the conduit of choice for coronary artery bypass grafting (CABG). While it can be harvested as a pedicled (with surrounding tissue) or skeletonized (stripped bare) graft, the optimal technique remains debated. The perivascular adipose tissue (PVAT) surrounding the IMA is no longer considered inert but is a bioactive organ secreting vasoactive adipokines. This review synthesizes the evidence on the role of PVAT in IMA vasoreactivity and its implications for the choice of harvesting technique. Methods: A systematic literature search was conducted in PubMed/MEDLINE, Embase, and CENTRAL up to 1 December 2025 for original studies investigating PVAT function in human IMA or comparing pedicled vs. skeletonized harvesting with relevant vasomotor or clinical outcomes. Data were extracted and synthesized narratively. Results: The included studies consistently demonstrate that IMA-PVAT exerts a potent anticontractile and vasorelaxant effect. This is mediated through multiple pathways, including the release of adiponectin, which enhances endothelial nitric oxide synthase function, and the production of non-nitric oxide, non-prostanoid relaxing factors that act directly on vascular smooth muscle. Ex vivo studies show conflicting results that skeletonization, by removing PVAT, may or may not augment vasoconstrictive responses and increase susceptibility to spasm but preservation of PVAT definitively preserves vasorelaxation properties of IMA. Though less definitive and conflicting, clinical studies, particularly a recent large post-hoc RCT analysis, suggest that harvesting of the IMA during CABG surgery using a skeletonized technique was associated with a higher rate of graft occlusion and worse clinical outcomes than the traditional pedicled technique. Conclusions: PVAT is an integral, protective component of the human IMA, providing a multimodal defense against vasospasm. The pedicled harvesting technique, by conserving PVAT, leverages these inherent vasorelaxant properties and is physiologically superior for minimizing graft spasm. Surgical strategy should consider the antispastic benefit of the pedicled IMA, balanced against the potential sternal wound healing and length advantages of skeletonization in high-risk patients.
- Open Access
- Review
Which Is Better? Pedicled versus Skeletonized Internal Mammary Artery Grafts in Coronary Artery Bypass Grafting: Emphasis on the Antispastic Role of Perivascular Adipose
Author Information
Received: 02 Dec 2025 | Revised: 13 Feb 2026 | Accepted: 25 Feb 2026 | Published: 07 Apr 2026
Abstract
Graphical Abstract
Keywords
References
- 1.
Head, S.J.; Milojevic, M.; Daemen, J.; et al. Mortality after coronary artery bypass grafting versus percutaneous coronary intervention with stenting for coronary artery disease: A pooled analysis of individual patient data. Lancet 2018, 391, 939–948.
- 2.
Neumann, F.J.; Sousa-Uva, M.; Ahlsson, A.; et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur. Heart J. 2019, 40, 87–165.
- 3.
Byrne, R.A.; Fremes, S.; Capodanno, D.; et al. 2022 Joint ESC/EACTS review of the 2018 guideline recommendations on the revascularization of left main coronary artery disease in patients at low surgical risk and anatomy suitable for PCI or CABG. Eur. J. Cardio-Thorac. Surg. 2023, 64, ezad286.
- 4.
Kolessov, V.I. Mammary artery-coronary artery anastomosis as method of treatment for angina pectoris. J. Thorac. Cardiovasc. Surg. 1967, 54, 535–544.
- 5.
Tatoulis, J.; Buxton, B.F.; Fuller, J.A. Patencies of 2127 arterial to coronary conduits over 15 years. Ann. Thorac. Surg. 2004, 77, 93–101.
- 6.
Buxton, B.F.; Fuller, J.A.; Tatoulis, J. Evolution of complete arterial grafting. For coronary artery disease. Tex. Heart Inst. J. 1998, 25, 17–23.
- 7.
Lüscher, T.F.; Diederich, D.; Siebenmann, R.; et al. Difference between endothelium-dependent relaxation in arterial and in venous coronary bypass grafts. N. Engl. J. Med. 1988, 319, 462–467.
- 8.
He, G.W.; Liu, Z.G. Comparison of nitric oxide release and endothelium-derived hyperpolarizing factor-mediated hyperpolarization between human radial and internal mammary arteries. Circulation 2001, 104, I344–I349.
- 9.
Liu, Z.G.; Ge, Z.D.; He, G.W. Difference in endothelium-derived hyperpolarizing factor-mediated hyperpolarization and nitric oxide release between human internal mammary artery and saphenous vein. Circulation 2000, 102, 296–301. https://doi.org/10.1161/01.cir.102.suppl_3.iii-296.
- 10.
He, G.W.; Yang, C.Q. Comparison among arterial grafts and coronary artery. An attempt at functional classification. J. Thorac. Cardiovasc. Surg. 1995, 109, 707–715.
- 11.
Deja, M.A.; Woś, S.; Gołba, K.S.; et al. Intraoperative and laboratory evaluation of skeletonized versus pedicled internal thoracic artery. Ann. Thorac. Surg. 1999, 68, 2164–2168.
- 12.
Van den Eynde, J.; Heeren, A.; Szecel, D.; et al. Skeletonisation contributing to a reduction of sternal wound complications: A retrospective study in OPCAB patients. J. Cardiothorac. Surg. 2019, 14, 162. https://doi.org/10.1186/s13019-019-0985-9.
- 13.
Ohira, S.; Miyata, H.; Yamazaki, S.; et al. Deep sternal wound infection after bilateral internal thoracic artery grafting: Insights from a Japanese national database. J. Thorac. Cardiovasc. Surg. 2019, 157, 166–173.e1. https://doi.org/10.1016/j.jtcvs.2018.06.101.
- 14.
Sahar, G.; Shavit, R.; Yosibash, Z.; et al. The physiologic and histologic properties of the distal internal thoracic artery and its subdivisions. J. Thorac. Cardiovasc. Surg. 2015, 149, 1042–1050.
- 15.
Gaudino, M.; Toesca, A.; Nori, S.L.; et al. Effect of skeletonization of the internal thoracic artery on vessel wall integrity. Ann. Thorac. Surg. 1999, 68, 1623–1627.
- 16.
Gaudino, M.; Trani, C.; Glieca, F.; et al. Early vasoreactive profile of skeletonized versus pedicled internal thoracic artery grafts. J. Thorac. Cardiovasc. Surg. 2003, 125, 638–641.
- 17.
Löhn, M.; Dubrovska, G.; Lauterbach, B.; et al. Periadventitial fat releases a vascular relaxing factor. FASEB J. 2002, 16, 1057–1063.
- 18.
Gollasch, M. Vasodilator signals from perivascular adipose tissue. Br. J. Pharmacol. 2012, 165, 633–642.
- 19.
Fésüs, G.; Dubrovska, G.; Gorzelniak, K.; et al. Adiponectin is a novel humoral vasodilator. Cardiovasc. Res. 2007, 75, 719–727.
- 20.
Momin, A.U.; Melikian, N.; Shah, A.M.; et al. Leptin is an endothelial-independent vasodilator in humans with coronary artery disease: Evidence for tissue specificity of leptin resistance. Eur. Heart J. 2006, 27, 2294–2299.
- 21.
Fang, L.; Zhao, J.; Chen, Y.; et al. Hydrogen sulfide derived from periadventitial adipose tissue is a vasodilator. J. Hypertens. 2009, 27, 2174–2185.
- 22.
Ozen, G.; Topal, G.; Gomez, I.; et al. Control of human vascular tone by prostanoids derived from perivascular adipose tissue. Prostaglandins Other Lipid Mediat. 2013, 107, 13–17.
- 23.
Gao, Y.J.; Zeng, Z.H.; Teoh, K.; et al. Perivascular adipose tissue modulates vascular function in the human internal thoracic artery. J. Thorac. Cardiovasc. Surg. 2005, 130, 1130–1136.
- 24.
Malinowski, M.; Deja, M.A.; Gołba, K.S.; et al. Perivascular tissue of internal thoracic artery releases potent nitric oxide and prostacyclin-independent anticontractile factor. Eur. J. Cardiothorac. Surg. 2008, 33, 225–231.
- 25.
Numaguchi, R.; Furuhashi, M.; Matsumoto, M.; et al. Differential Phenotypes in Perivascular Adipose Tissue Surrounding the Internal Thoracic Artery and Diseased Coronary Artery. J. Am. Heart Assoc. 2019, 8, e011147. https://doi.org/10.1161/JAHA.118.011147.
- 26.
Zhang, Y.F.; Hou, H.T.; Yang, Q.; et al. Perivascular adipose on relaxation of internal mammary artery with surgical implications: Better antispastic effect of pedicle than skeletal grafts. J. Thorac. Cardiovasc. Surg. 2025, in press. https://doi.org/10.1016/j.jtcvs.2025.10.033.
- 27.
Margaritis, M.; Antonopoulos, A.S.; Digby, J.; et al. Interactions between vascular wall and perivascular adipose tissue reveal novel roles for adiponectin in the regulation of endothelial nitric oxide synthase function in human vessels. Circulation 2013, 127, 2209–2221.
- 28.
Niinami, H.; Endo, Y.; Morita, K.; et al. Long-Term Outcomes Following Off-pump Coronary Artery Bypass Grafting Using Only 3 In situ Arterial Grafts. Eur. J. Cardiothorac. Surg. 2025, 67, ezaf289.
- 29.
Zhao, T.Y.; Wang, B.J.; Liu, C.; et al. Comparing 3-month and 1-year patency rates of no-touch great saphenous vein and pedicled left internal mammary artery in off-pump coronary artery bypass grafting: A prospective non-inferiority study. Front. Cardiovasc. Med. 2025, 12, 1547482.
- 30.
Calafiore, A.M.; Vitolla, G.; Iaco, A.L.; et al. Bilateral internal mammary artery grafting: Midterm results of pedicled versus skeletonized conduits. Ann. Thorac. Surg. 1999, 67, 1637–1642.
- 31.
Mannacio, V.; Di Tommaso, L.; De Amicis, V.; et al. Randomized flow capacity comparison of skeletonized and pedicled left internal mammary artery. Ann. Thorac. Surg. 2011, 91, 24–30.
- 32.
Zientara, A.; Komminoth, P.; Seifert, B.; et al. Skeletonized internal thoracic artery harvesting: A low thermal damage electrosurgical device provides improved endothelial layer and tendency to better integrity of the vessel wall compared to conventional electrosurgery. J. Cardiothorac. Surg. 2018, 13, 105.
- 33.
Lamy, A.; Browne, A.; Sheth, T.; et al. Skeletonized vs. Pedicled Internal Mammary Artery Graft Harvesting in Coronary Artery Bypass Surgery: A Post Hoc Analysis from the COMPASS Trial. JAMA Cardiol. 2021, 6, 1042–1049.
- 34.
Abfalterer, H.; Ruttmann-Ulmer, E.; Grimm, M.; et al. Randomized comparison of HARVesting the Left Internal Thoracic Artery in a skeletonized versus pedicled technique: The HARVITA trial-study protocol. Interdiscip. Cardiovasc. Thorac. Surg. 2024, 38, ivae045.
- 35.
Formica, F.; Ferro, O.; Brustia, M.; et al. Effects of papaverine and glycerylnitrate-verapamil solution as topical and intraluminal vasodilators for internal thoracic artery. Ann. Thorac. Surg. 2006, 81, 120–124.
- 36.
Fu, Y.; Hong, Y.; Ding, T.; et al. Early outcomes and follow-up of robotic-assisted multivessel coronary artery bypass with in situ bilateral internal thoracic artery: Report of 221 patients in a single center. J. Thorac. Cardiovasc. Surg. 2025, 170, 1639–1647.
- 37.
Jung, Y.C.; Chong, Y.; Kang, M.W.; et al. Clipless internal mammary artery harvesting for minimally invasive coronary artery bypass grafting using the shear-tip harmonic scalpel. J. Thorac. Dis. 2024, 16, 3711–3721.
- 38.
Nisivaco, S.; Kitahara, H.; Abutaleb, A.; et al. Robotic Totally Endoscopic Coronary Bypass to the Left Anterior Descending Artery: Left Versus Right Internal Thoracic Artery Grafts. J. Surg. Res. 2023, 291, 139–150.
- 39.
Marín-Cuartas, M.; Kang, J.; Davierwala, P.M.; et al. Step-by-step harvesting of various grafts for coronary artery bypass surgery. Multimed. Man. Cardiothorac. Surg. 2021, 2021. https://doi.org/10.1510/mmcts.2021.076.
- 40.
Cerrato, R.; Crabtree, M.; Antoniades, C.; et al. Effects of Endothelin-1 on Intracellular Tetrahydrobiopterin Levels in Vascular Tissue. Scand. Cardiovasc. J. 2018, 52, 163–169.
- 41.
Ji, Q.; Xia, L.; Shi, Y.; et al. Sequential Grafting of in Situ Skeletonized Left Internal Mammary Artery to the Left Coronary System. Int. Heart J. 2018, 59, 727–735.
- 42.
Ji, Q.; Xia, L.; Shi, Y.; et al. In Situ Skeletonized Right Internal Mammary Artery Bypass Grafting to Left Anterior Descending Artery. Int. Heart J. 2018, 59, 35–42.
- 43.
Ji, Q.; Shi, Y.; Xia, L.; et al. Revascularization of Left Coronary System Using a Skeletonized Left Internal Mammary Artery—Sequential vs. Separate Grafting. Circ. J. 2017, 82, 102–109.
- 44.
Ray Mohapatra, C.K.; Mishra, P.; Saxena, P.; et al. Use of nitroglycerin and verapamil solution by organ bath technique in preparation of left internal thoracic artery for coronary artery bypass surgery. Indian. Heart J. 2017, 69, 772–776.
- 45.
Fukuda, S.; Nakamura, Y.; Egi, K.; et al. Comparison of direct effects of clinically available vasodilators; nitroglycerin, nifedipine, cilnidipine and diltiazem, on human skeletonized internal mammary harvested with ultrasonic scalpel. Heart Vessel. 2016, 31, 1681–1684.
- 46.
Cybularz, M.; Langbein, H.; Zatschler, B.; et al. Endothelial function and gene expression in perivascular adipose tissue from internal mammary arteries of obese patients with coronary artery disease. Atheroscler. Suppl. 2017, 30, 149–158.
- 47.
Rieß, F.C.; Behrendt, C.A.; Amin, W.; et al. Complete arterial revascularization using bilateral internal mammary artery in T-graft technique for multivessel coronary artery disease in on- or off-pump approach: Does gender lose its historical impact on clinical outcome? Eur. J. Cardio-Thorac. Surg. 2017, 52, 917–923.
- 48.
Gu, C.X.; Yang, J.F.; Zhang, H.C.; et al. Off-pump coronary artery bypass grafting using a bilateral internal mammary artery Y graft. J. Geriatr. Cardiol. 2012, 9, 247–251.
- 49.
Cho, W.C.; Kim, J.B.; Jung, S.H.; et al. Revascularization of the left anterior descending artery area using a single left internal thoracic artery: Auto-Y composite grafting or sequential bypassing. J. Thorac. Cardiovasc. Surg. 2011, 142, 1464–1468.
- 50.
Glineur, D.; Djaoudi, S.; D’horre, W.; et al. Endothelium-dependent and endothelium-independent vasodilator response of left and right internal mammary and internal thoracic arteries used as a composite Y-graft. Eur. J. Cardiothorac. Surg. 2011, 40, 389–393.
- 51.
Asai, T.; Suzuki, T.; Nota, H.; et al. Off-pump coronary artery bypass grafting using skeletonized in situ arterial grafts. Ann. Cardiothorac. Surg. 2013, 2, 552–556.
- 52.
Obed, D.; Fleissner, F.; Martens, A.; et al. Total Arterial Revascularization with Radial Artery and Internal Thoracic Artery T-Grafts Is Associated with Superior Long-Term Survival in Patients Undergoing Coronary Artery Bypass Grafting. Ann. Thorac. Cardiovasc. Surg. 2020, 26, 30–39. https://doi.org/10.5761/atcs.oa.19-00226.
- 53.
Kim, K.B.; Cho, K.R.; Chang, W.I.; et al. Bilateral skeletonized internal thoracic artery graftings in off-pump coronary artery bypass: Early result of Y versus in situ grafts. Ann. Thorac. Surg. 2002, 74, S1371–S1376.
- 54.
Andreasen, J.J.; Aardestrup, I.V.; Eschen, R.B.; et al. Fatty acid composition of the internal mammary artery in relation to dietary intake of marine n-3 polyunsaturated fatty acids and association with flow-mediated vasodilation. Cell Mol. Biol. 2010, 56, 10–17.
- 55.
He, G.W.; Ryan, W.H.; Acuff, T.E.; et al. Risk factors for operative mortality and sternal wound infection in bilateral internal mammary artery grafting. J. Thorac. Cardiovasc. Surg. 1994, 107, 196–202.
- 56.
Sakic, A.; Chevtchik, O.; Kilo, J.; et al. Simple adaptations of surgical technique to critically reduce the risk of postoperative sternal complications in patients receiving bilateral internal thoracic arteries. Interact. Cardiovasc. Thorac. Surg. 2013, 17, 378–382.
- 57.
Kinoshita, T.; Asai, T.; Suzuki, T. Preoperative SYNTAX score and graft patency after off-pump coronary bypass surgery. Eur. J. Cardiothorac. Surg. 2013, 44, e25–e31.
- 58.
Gaudino, M.; Toesca, A.; Glieca, F.; et al. Skeletonization does not influence internal thoracic artery innervation. Ann. Thorac. Surg. 2004, 77, 1257–1261.
- 59.
Une, D.; Shimizu, S.; Kamiya, A.; et al. Both skeletonized and pedicled internal thoracic arteries supply adequate graft flow after coronary artery bypass grafting even during intense sympathoexcitation. J. Physiol. Sci. 2010, 60, 407–413. https://doi.org/10.1007/s12576-010-0109-8.
- 60.
Maruyama, T.; Kohno, H.; Ishida, K.; et al. Change of luminal diameter of skeletonized and non-skeletonized radial artery graft at early and late postoperative period. Heart Vessel. 2016, 31, 474–481.
- 61.
Amano, A.; Takahashi, A.; Hirose, H. Skeletonized radial artery grafting: Improved angiographic results. Ann. Thorac. Surg. 2002, 73, 1880–1887.
- 62.
Shi, J.; Iesaki, T.; Kubota, N.; et al. Skeletonization with an ultrasonic scalpel is as safe as a non-skeletonized dissection in preserving the endothelial function of the human gastroepiploic artery. Interact. Cardiovasc. Thorac. Surg. 2009, 8, 216–220.
- 63.
Kamiya, H.; Watanabe, G.; Takemura, H.; et al. Skeletonization of gastroepiploic artery graft in off-pump coronary artery bypass grafting: Early clinical and angiographic assessment. Ann. Thorac. Surg. 2004, 77, 2046–2050.
- 64.
Eren, E.; Balkanay, M.; Toker, M.E.; et al. Pedicled right internal mammary artery for reoperative off-pump revascularization of left anterior descending coronary artery. Tex. Heart Inst. J. 2006, 33, 143–147.
- 65.
Agrifoglio, M.; Di Matteo, S.; Antona, C.; et al. Pedicled arterial grafts in coronary surgery: Postoperative echo color-Doppler study. J. Cardiovasc. Surg. 1996, 37, 53–57.
- 66.
Lust, R.M.; Zeri, R.S.; Spence, P.A.; et al. Effect of chronic native flow competition on internal thoracic artery grafts. Ann. Thorac. Surg. 1994, 57, 45–50.
- 67.
Kushwaha, S.S.; Bustami, M.; Tadjkarimi, S.; et al. Late endothelial function of free and pedicled internal mammary artery grafts. J. Thorac. Cardiovasc. Surg. 1995, 110, 453–462.
- 68.
Isomura, T.; Hisatomi, K.; Hirano, A.; et al. Use of the right gastroepiploic artery as a pedicled arterial graft for coronary revascularization. Eur. J. Cardiothorac. Surg. 1993, 7, 38–41.
- 69.
Srivastava, S.; Gadasalli, S.; Agusala, M.; et al. Use of bilateral internal thoracic arteries in CABG through lateral thoracotomy with robotic assistance in 150 patients. Ann. Thorac. Surg. 2006, 81, 800–806.
- 70.
Cheng, K.K.; Lam, K.S.; Wang, Y.; et al. Adiponectin-induced endothelial nitric oxide synthase activation and nitric oxide production are mediated by APPL1 in endothelial cells. Diabetes 2007, 56, 1387–1394; Erratum in Diabetes 2016, 65, 3218. https://doi.org/10.2337/db16-er10a.
- 71.
Lembo, G.; Vecchione, C.; Fratta, L.; et al. Leptin induces direct vasodilation through distinct endothelial mechanisms. Diabetes 2000, 49, 293–297.
- 72.
Gao, Y.J.; Lu, C.; Su, L.Y.; et al. Modulation of vascular function by perivascular adipose tissue: The role of endothelium and hydrogen peroxide. Br. J. Pharmacol. 2007, 151, 323–331.
- 73.
Emilova, R.; Dimitrova, D.; Mladenov, M.; et al. Cystathionine gamma-lyase of perivascular adipose tissue with reversed regulatory effect in diabetic rat artery. Biotechnol. Biotechnol. Equip. 2015, 29, 147–151. https://doi.org/10.1080/13102818.2014.991565.
- 74.
Soltis, E.E.; Cassis, L.A. Influence of perivascular adipose tissue on rat aortic smooth muscle responsiveness. Clin. Exp. Hypertens. A 1991, 13, 277–296.
- 75.
Antonopoulos, A.S.; Margaritis, M.; Coutinho, P.; et al. Adiponectin as a link between type 2 diabetes and vascular NADPH oxidase activity in the human arterial wall: The regulatory role of perivascular adipose tissue. Diabetes 2015, 64, 2207–2219.
- 76.
Wei, J.H.; Qi, H.; Zhou, Y.; et al. Homocysteine impairs the anticontractile/vasorelaxing activity of perivascular adipose tissue surrounding human internal mammary artery. Eur. J. Cardiothorac. Surg. 2023, 64, ezad406.
- 77.
Wei, J.H.; Qi, H.; Wang, X.C.; et al. Inhibition of soluble epoxide hydrolase ameliorates hyperhomocysteinemia-induced perivascular adipose tissue dysfunction: Revealing epoxyeicosatrienoic acid as a perivascular adipose tissue-derived relaxing factor. Eur. J. Pharmacol. 2025, 1006, 178181.
- 78.
Zierold, S.; Buschmann, K.; Gachkar, S.; et al. Brain-Derived Neurotrophic Factor Expression and Signaling in Different Perivascular Adipose Tissue Depots of Patients with Coronary Artery Disease. J. Am. Heart Assoc. 2021, 10, e018322. https://doi.org/10.1161/JAHA.120.018322.
- 79.
Takami, Y.; Ina, H. Effects of skeletonization on intraoperative flow and anastomosis diameter of internal thoracic arteries in coronary artery bypass grafting. Ann. Thorac. Surg. 2002, 73, 1441–1445.
- 80.
Dalén, M.; Dismorr, M.; Glaser, N.; et al. Skeletonized Versus Pedicled Harvesting of the Internal Thoracic Artery and Long-Term Clinical Outcomes in Coronary Artery Bypass Surgery. J. Am. Heart Assoc. 2024, 13, e034354. https://doi.org/10.1161/JAHA.124.034354.
- 81.
He, G.W. Contractility of the human internal mammary artery at the distal section increases toward the end. Emphasis on not using the end of the internal mammary artery for grafting. J. Thorac. Cardiovasc. Surg. 1993, 106, 406–411.
- 82.
He, G.W.; Ryan, W.H.; Acuff, T.E.; et al. Greater contractility of internal mammary artery bifurcation: Possible cause of low patency rates. Ann. Thorac. Surg. 1994, 58, 529–532.
- 83.
He, G.W.; Acuff, T.E.; Yang, C.Q.; et al. Middle and proximal sections of the human internal mammary artery are not “passive conduits”. J. Thorac. Cardiovasc. Surg. 1994, 108, 741–746.
- 84.
Sun, W.T.; Chen, H.X.; Hou, H.T.; et al. Protein post-translational modification crotonylation of TXN and GLO1 in artery and vein grafts for coronary artery surgery. Redox Biol. 2025, 82, 103608. https://doi.org/10.1016/j.redox.2025.103608.
- 85.
Shin, H.; Yozu, R.; Hashizume, K.; et al. Free right internal thoracic artery as a second arterial conduit: Modification of proximal anastomosis for improvement of graft patency. Ann. Thorac. Cardiovasc. Surg. 2001, 7, 155–158.
- 86.
Sahar, G.; Wolak, A.; Matsa, M.; et al. Concomitant grafting of both postbifurcation internal thoracic artery branches. Innovations 2015, 10, 14–20.
- 87.
Li, J.Y.; Chen, M.R. Complete myocardial revascularization using only pedicled arterial conduits in Kawasaki disease. J. Formos. Med. Assoc. 2000, 99, 507–509.
- 88.
Nishida, H.; Tomizawa, Y.; Endo, M.; et al. Coronary artery bypass with only in situ bilateral internal thoracic arteries and right gastroepiploic artery. Circulation 2001, 104, I76–I80.
- 89.
Zhang, G.; Zhao, Z.; Chen, Y.; et al. Use of the right internal mammary artery and the great saphenous vein for left anterior descending artery revascularization in patients whose left internal mammal artery cannot be used: A study based on transit-time flow measurement. J. Cardiothorac. Surg. 2020, 15, 126.
- 90.
Chan, J.; Comanici, M.; Dong, T.; et al. National Variation in the Use of Multiple Arterial Grafting in Isolated Coronary Artery Bypass Grafting in the United Kingdom. Eur. J. Cardiothorac. Surg. 2025, 67, ezaf402. https://doi.org/10.1093/ejcts/ezaf402.
- 91.
Chan, J.; Dimagli, A.; Dong, T.; et al. Trend and early clinical outcomes of off-pump coronary artery bypass grafting in the UK. Eur. J. Cardiothorac. Surg. 2023, 64, ezad272. https://doi.org/10.1093/ejcts/ezad272.
- 92.
Lorusso, R.; Crudeli, E.; Luca, F.; et al. Refractory spasm of coronary arteries and grafted conduits after isolated coronary artery bypass surgery. Ann. Thorac. Surg. 2012, 93, 545–551.
- 93.
He, G.W.; Rosenfeldt, F.L.; Buxton, B.F.; et al. Reactivity of human isolated internal mammary artery to constrictor and dilator agents: Implications for treatment of internal mammary artery spasm. Circulation 1989, 80, I141–I150.
- 94.
He, G.W.; Buxton, B.F.; Rosenfeldt, F.L.; et al. Pharmacologic dilatation of the internal mammary artery during coronary bypass grafting. J. Thorac. Cardiovasc. Surg. 1994, 107, 1440–1444.
- 95.
Yin, X.Y.; Hou, H.T.; Li, M.R.; et al. A New Method for Antispastic Effect in Coronary Artery Bypass Grafts by Using a L- and T-Type Calcium Channel Blocker Efonidipine. Basic. Clin. Pharmacol. Toxicol. 2025, 137, e70077. https://doi.org/10.1111/bcpt.70077.
- 96.
Duman, I.; Tanyeli, Ö.; Dereli, Y.; et al. The effects of botulinum toxin A and papaverine on human saphenous vein and internal mammary artery grafts: An in vitro study. Arch. Med. Sci. 2021, 18, 1607–1616. https://doi.org/10.5114/aoms/110157.
- 97.
Hou, H.T.; Wang, Z.Q.; Wang, J.; et al. Antispastic Effect of Fasudil and Cocktail of Fasudil and Nitroglycerin in Internal Thoracic Artery. Ann. Thorac. Surg. 2023, 115, 1152–1161. https://doi.org/10.1016/j.athoracsur.2021.11.079.
- 98.
Sun, W.T.; Hou, H.T.; Chen, H.X.; et al. Calcium-activated potassium channel family in coronary artery bypass grafts. J. Thorac. Cardiovasc. Surg. 2021, 161, e399–e409. https://doi.org/10.1016/j.jtcvs.2019.11.016.
- 99.
Kinoshita, T.; Tawa, M.; Suzuki, T.; et al. Suppression of Graft Spasm by the Particulate Guanylyl Cyclase Activator in Coronary Bypass Surgery. Ann. Thorac. Surg. 2017, 104, 122–129. https://doi.org/10.1016/j.athoracsur.2016.10.003.
- 100.
Ertuna, E.; Turkseven, S.; Amanvermez, D.; et al. Effects of levosimendan on isolated human internal mammary artery and saphenous vein: Concurrent use with conventional vasodilators. Fundam. Clin. Pharmacol. 2016, 30, 226–234. https://doi.org/10.1111/fcp.12185.
- 101.
Hou, H.T.; Wang, J.; Wang, Z.Q.; et al. Effect of Benidipine in Human Internal Mammary Artery and Clinical Implications. Ann. Thorac. Surg. 2016, 101, 1789–1795. https://doi.org/10.1016/j.athoracsur.2015.10.029.
- 102.
Sarikaya, S.; Onk, A.; Boztosun, B.; et al. The effect of nebivolol on internal mammary artery blood flow during coronary artery bypass graft surgery. Perfusion 2014, 29, 315–320. https://doi.org/10.1177/0267659113517921.
- 103.
Gao, G.; Liu, X.C.; Jing, W.B.; et al. Vasorelaxation induced by new third-generation dihydropyridine calcium antagonist azelnidipine in human internal mammary artery. Ann. Thorac. Surg. 2013, 96, 1316–1321. https://doi.org/10.1016/j.athoracsur.2013.05.020.
- 104.
Bai, X.Y.; Liu, X.C.; Yang, Q.; et al. The interaction between human urotensin II and vasodilator agents in human internal mammary artery with possible clinical implications. Ann. Thorac. Surg. 2011, 92, 610–614. https://doi.org/10.1016/j.athoracsur.2011.03.094.
- 105.
Bai, X.Y.; Liu, X.C.; Jing, W.B.; et al. Effect of amlodipine in human internal mammary artery and clinical implications. Ann. Thorac. Surg. 2010, 90, 1952–1957. https://doi.org/10.1016/j.athoracsur.2010.08.007.
- 106.
Ding, R.; Feng, W.; Li, H.; et al. A comparative study on in vitro and in vivo effects of topical vasodilators in human internal mammary, radial artery and great saphenous vein. Eur. J. Cardiothorac. Surg. 2008, 34, 536–541. https://doi.org/10.1016/j.ejcts.2008.05.050.
- 107.
Onomoto, M.; Tsuneyoshi, I.; Yonetani, A.; et al. Differential pharmacologic sensitivities of phosphodiesterase-3 inhibitors among human isolated gastroepiploic, internal mammary, and radial arteries. Anesth. Analg. 2005, 101, 950–956. https://doi.org/10.1213/01.ane.0000172114.30383.23.
- 108.
Fan, L.; Yang, Q.; Xiao, X.Q.; et al. Dual actions of cilnidipine in human internal thoracic artery: Inhibition of calcium channels and enhancement of endothelial nitric oxide synthase. J. Thorac. Cardiovasc. Surg. 2011, 141, 1063–1069. https://doi.org/10.1016/j.jtcvs.2010.01.048.
- 109.
He, G.W.; Fan, L.; Furnary, A.; et al. A new antispastic solution for arterial grafting: Nicardipine and nitroglycerin cocktail in preparation of internal thoracic and radial arteries for coronary surgery. J. Thorac. Cardiovasc. Surg. 2008, 136, 673–680.e2. https://doi.org/10.1016/j.jtcvs.2007.12.019.
- 110.
Zhang, Y.F.; Hou, H.T.; Yang, Q.; et al. Significant Antispastic Effect of Combined Fasudil and Isosorbide Dinitrate on Internal Mammary Artery in Coronary Artery Bypass Grafting. Basic Clin. Pharmacol. Toxicol. 2025, 138, e70148.
- 111.
Zheng, S.Y.; Wu, M.; Huang, J.S.; et al. Use of antispastic nicardipine and nitroglycerin (NG) cocktail solution increases graft flow during off-pump coronary artery bypass grafting. J. Cardiovasc. Surg. 2012, 53, 783–788.
- 112.
Yang, Q.; Xue, H.M.; Wong, W.T.; et al. AVE3085, an enhancer of endothelial nitric oxide synthase, restores endothelial function and reduces blood pressure in spontaneously hypertensive rats. Br. J. Pharmacol. 2011, 163, 1078–1085. https://doi.org/10.1111/j.1476-5381.2011.01308.x.
- 113.
Xuan, C.; Chang, F.J.; Liu, X.C.; et al. Endothelial nitric oxide synthase enhancer for protection of endothelial function from asymmetric dimethylarginine-induced injury in human internal thoracic artery. J. Thorac. Cardiovasc. Surg. 2012, 144, 697–703. https://doi.org/10.1016/j.jtcvs.2012.01.020.
- 114.
Wei, W.; Chen, Z.W.; Yang, Q.; et al. Vasorelaxation induced by vascular endothelial growth factor in the human internal mammary artery and radial artery. Vascul Pharmacol. 2007, 46, 253–259. https://doi.org/10.1016/j.vph.2006.10.009.
- 115.
Hou, H.T.; Wang, J.; Zhang, X.; et al. Endothelial nitric oxide synthase enhancer AVE3085 reverses endothelial dysfunction induced by homocysteine in human internal mammary arteries. Nitric Oxide 2018, 81, 21–27. https://doi.org/10.1016/j.niox.2018.10.001.

This work is licensed under a Creative Commons Attribution 4.0 International License.