Background: The role of genetic testing in sudden cardiac arrest (SCA) among young and middle-aged adults remains incompletely understood, particularly in pre-hospital settings and in individuals with established clinical diagnoses such as coronary artery disease. Methods: We performed whole-exome sequencing with targeted analysis in a city-wide cohort of individuals who experienced pre-hospital SCA in Hangzhou, China. Variants were evaluated using a virtual panel of 2151 cardiovascular-related genes. Variant classification followed the American College of Medical Genetics and Genomics guidelines. Results: A total of 69 individuals (mean age 38 ± 14.6 years) were included. Pathogenic or likely pathogenic variants were identified in 13 individuals, corresponding to a diagnostic yield of 18.8%. One individual carried two variants. The identified variants involved genes associated with cardiomyopathies, arrhythmia syndromes, metabolic disorders, and lipid metabolism. Genetic findings were observed both in individuals without a clear clinical diagnosis and in those with apparently established causes of cardiac arrest, including coronary artery disease. When rare variants of uncertain significance with supportive evidence were considered, up to 47.8% of individuals carried potentially relevant variants. Broader gene panels identified more variants than narrower panels limited to established sudden cardiac death genes. Conclusions: Systematic exome sequencing in young and middle-aged individuals with pre-hospital SCA identifies clinically relevant genetic variants in a substantial proportion of cases. Genetic testing may complement conventional clinical investigation and may contribute to molecular autopsy and family-based risk assessment in selected patients.
- Open Access
- Article
Targeted Exome Sequencing in Pre-Hospital Sudden Cardiac Arrest Reveals a High Genetic Diagnostic Yield
- Jiangang Wang 1,†,
- Jingcai Xu 2,†,
- Lingye Hong 3,
- Xiaoling Chen 1,
- Meili Lu 1,
- Shuangyong Dong 4,
- Jian Huang 5,
- Wei Shi 6,
- Jianwei Lin 7,
- Jianying Wang 8,
- Yao Li 8,
- Qianyuan Zhu 9,
- Yue Wang 9,
- Xutao Hong 7,10,*,
- Ming Qi 7,10,11,*
Author Information
Received: 30 Dec 2025 | Revised: 16 Mar 2026 | Accepted: 30 Mar 2026 | Published: 15 Apr 2026
Abstract
Graphical Abstract
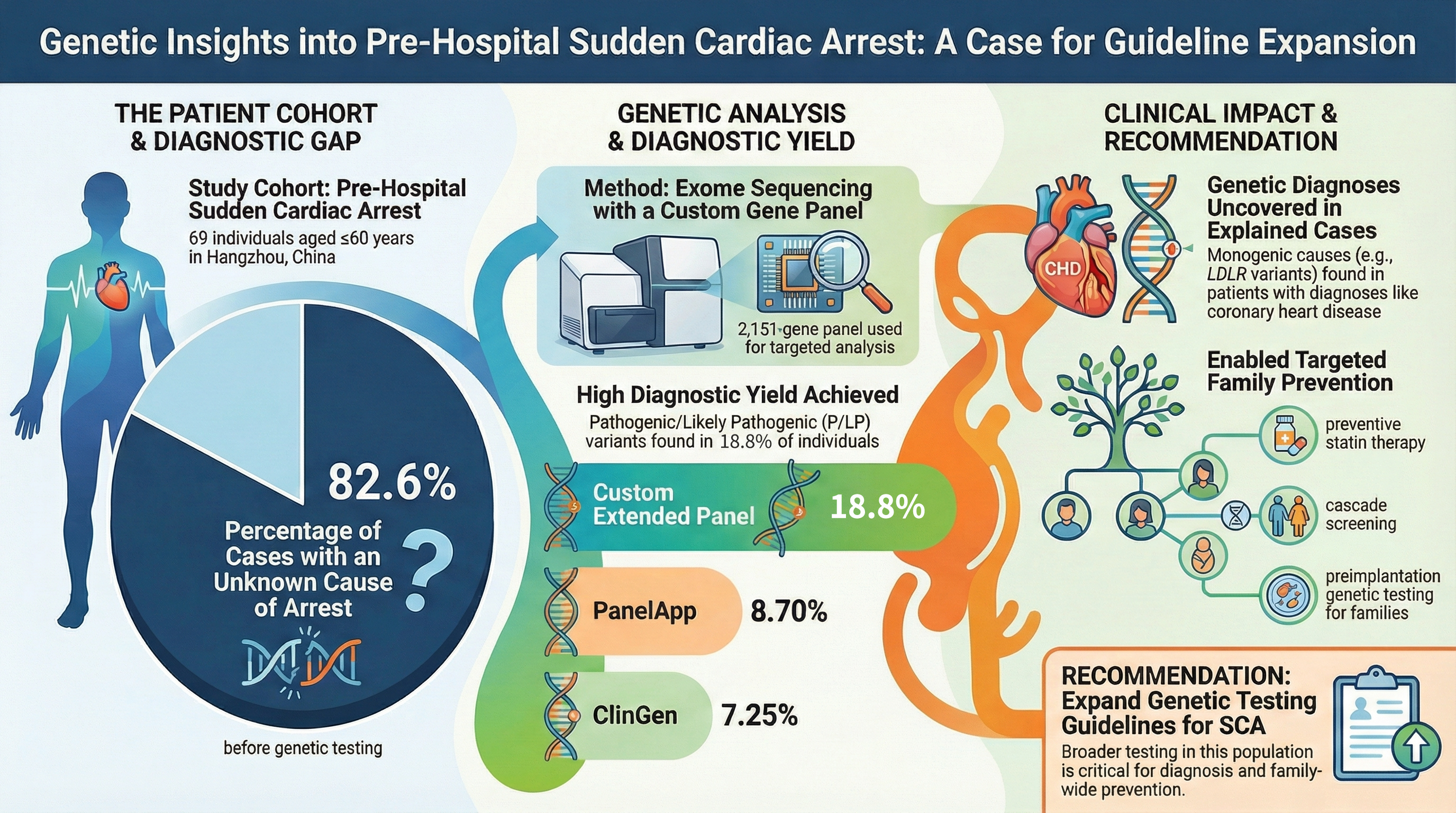
Diagnostic Yield and Clinical Implications of Exome Sequencing in Pre-Hospital Sudden Cardiac Arrest
This Graphical Abstract summarizes the study design, key genetic findings, and potential clinical implications. A cohort of young and middle-aged individuals experiencing pre-hospital sudden cardiac arrest (SCA) underwent systematic exome sequencing with targeted analysis. Pathogenic or likely pathogenic variants were identified in 18.8% of individuals, demonstrating a substantial genetic contribution to SCA in this population.
Genetic variants were detected both in individuals without a clear clinical diagnosis and in those with apparently established causes of cardiac arrest, including coronary artery disease. In addition to pathogenic variants, a number of rare variants of uncertain significance were identified that may warrant further investigation.
These findings highlight the potential role of genetic testing as a complementary approach in the investigation of sudden cardiac arrest, including molecular autopsy and family-based risk assessment. Identification of disease-associated variants may facilitate cascade screening and preventive clinical management in affected families.
Finally, identification of pathogenic variants prompted follow up clinical evaluation in several families. In one family with a history of sudden cardiac death, detection of a frameshift variant in MYBPC3 in a surviving individual led to targeted cardiac evaluation and implementation of preventive management strategies for relatives.
Abbreviations: ES, exome sequencing; SCA, sudden cardiac arrest; P/LP, pathogenic or likely pathogenic; VUS, variant of uncertain significance.
Keywords
sudden cardiac arrest | pre-hospital | exome sequencing | molecular autopsy | diagnostic yield
References
- 1.
Zhang, S. Sudden cardiac death in China. Pacing Clin. Electrophysiol. 2009, 32, 1159–1162.
- 2.
Zhang, S. Sudden cardiac death in China: Current status and future perspectives. Europace 2015, 17, ii14–ii18.
- 3.
Zheng, J.; Lv, C.; Zheng, W.; et al. Incidence, process of care, and outcomes of out-of-hospital cardiac arrest in China: A prospective study of the BASIC-OHCA registry. Lancet Public Health 2023, 8, e923–e932.
- 4.
Chugh, S.S.; Jui, J.; Gunson, K.; et al. Current burden of sudden cardiac death: Multiple source surveillance versus retrospective death certificate-based review in a large U.S. community. J. Am. Coll. Cardiol. 2004, 44, 1268–1275.
- 5.
Nichol, G.; Thomas, E.; Callaway, C.W.; et al. Regional variation in out-of-hospital cardiac arrest incidence and outcome. JAMA 2008, 300, 1423–1431.
- 6.
Lin, X.; Ma, Y.; Cai, Z.; et al. Next-generation sequencing identified novel Desmoplakin frame-shift variant in patients with Arrhythmogenic cardiomyopathy. BMC Cardiovasc. Disord. 2020, 20, 74.
- 7.
Wang, Y.; Han, B.; Fan, Y.; et al. Next-Generation Sequencing Reveals Novel Genetic Variants for Dilated Cardiomyopathy in Pediatric Chinese Patients. Pediatr. Cardiol. 2022, 43, 110–120.
- 8.
Zhao, Y.; Feng, Y.; Ding, X.; et al. Identification of a novel hypertrophic cardiomyopathy-associated mutation using targeted next-generation sequencing. Int. J. Mol. Med. 2017, 40, 121–129.
- 9.
Bagnall, R.D.; Weintraub, R.G.; Ingles, J.; et al. A Prospective Study of Sudden Cardiac Death among Children and Young Adults. N. Engl. J. Med. 2016, 374, 2441–2452.
- 10.
Semsarian, C.; Ingles, J.; Wilde, A.A.M. Sudden cardiac death in the young: The molecular autopsy and a practical approach to surviving relatives. Eur. Heart J. 2015, 36, 1290–1296.
- 11.
Alders, M.; Bikker, H.; Christiaans, I. Long QT Syndrome. In GeneReviews((R)); Adam, M.P., Ed.; University of Washington: Seattle, WA, USA, 1993.
- 12.
Riera, A.R.; Zhang, L.; Uchida, A.H.; et al. The management of Brugada syndrome patients. Cardiol. J. 2007, 14, 97–106.
- 13.
Priori, S.G.; Blomström-Lundqvist, C.; Mazzanti, A.; et al. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: The Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the European Society of Cardiology (ESC)Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC). Eur. Heart J. 2015, 36, 2793–2867.
- 14.
Writing Group for Practice Guidelines for the Diagnosis; Treatment of Genetic Diseases Medical Genetics Branch of Chinese Medical Association; Han, S.; et al. [Clinical practice guidelines for Long Q-T interval syndrome]. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 2020, 37, 289–294.
- 15.
Miles, C.J.; Behr, E.R. The role of genetic testing in unexplained sudden death. Transl. Res. 2016, 168, 59–73.
- 16.
Grondin, S.; Davies, B.; Cadrin-Tourigny, J.; et al. Importance of genetic testing in unexplained cardiac arrest. Eur. Heart J. 2022, 43, 3071–3081.
- 17.
Guo, L.; Torii, S.; Fernandez, R.; et al. Genetic Variants Associated With Unexplained Sudden Cardiac Death in Adult White and African American Individuals. JAMA Cardiol. 2021, 6, 1013.
- 18.
Schon, U.; Holzer, A.; Laner, A.; et al. HPO-driven virtual gene panel: A new efficient approach in molecular autopsy of sudden unexplained death. BMC Med. Genom. 2021, 14, 94.
- 19.
Neves, R.; Tester, D.J.; Simpson, M.A.; et al. Exome Sequencing Highlights a Potential Role for Concealed Cardiomyopathies in Youthful Sudden Cardiac Death. Circ. Genom. Precis. Med. 2022, 15, e003497.
- 20.
Kransdorf, E.P.; Mathias, M.; Nakamura, K.; et al. Genetic Causes of Sudden Cardiac Arrest in the Community. medRxiv 2024, 12, 24318665.
- 21.
Miura, A.; Yamamoto, T.; Funayama, K.; et al. Postmortem Identification of Genetic Variations Associated with Sudden Unexpected Death in Young People. Int. Heart J. 2024, 65, 55–62.
- 22.
Tseng, Z.H.; Olgin, J.E.; Vittinghoff, E.; et al. Prospective Countywide Surveillance and Autopsy Characterization of Sudden Cardiac Death: POST SCD Study. Circulation 2018, 137, 2689–2700.
- 23.
Hamilton, R.M.; Cunningham, K.S.; Behr, E.R. Surviving Sudden Death: Where Does Next-Generation Sequencing Fit in the Assessment of Sudden Death Victims and Their Families. Circulation. Cardiovasc. Genet. 2017, 10, e002015.
- 24.
Martin, A.R.; Williams, E.; Foulger, R.E.; et al. PanelApp crowdsources expert knowledge to establish consensus diagnostic gene panels. Nat. Genet. 2019, 51, 1560–1565.
- 25.
Stark, Z.; Foulger, R.E.; Williams, E.; et al. Scaling national and international improvement in virtual gene panel curation via a collaborative approach to discordance resolution. Am. J. Hum. Genet. 2021, 108, 1551–1557.
- 26.
Richards, S.; Aziz, N.; Bale, S.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424.
- 27.
Whiffin, N.; Walsh, R.; Govind, R.; et al. CardioClassifier: Disease- and gene-specific computational decision support for clinical genome interpretation. Genet. Med. 2018, 20, 1246–1254.
- 28.
Ioannidis, N.M.; Rothstein, J.H.; Pejaver, V.; et al. REVEL: An Ensemble Method for Predicting the Pathogenicity of Rare Missense Variants. Am. J. Hum. Genet. 2016, 99, 877–885.
- 29.
Pejaver, V.; Byrne, A.B.; Feng, B.J.; et al. Calibration of computational tools for missense variant pathogenicity classification and ClinGen recommendations for PP3/BP4 criteria. Am. J. Hum. Genet. 2022, 109, 2163–2177.
- 30.
Tordai, H.; Torres, O.; Csepi, M.; et al. Analysis of AlphaMissense data in different protein groups and structural context. Sci. Data 2024, 11, 495.
- 31.
Frazer, J.; Notin, P.; Dias, M.; et al. Disease variant prediction with deep generative models of evolutionary data. Nature 2021, 599, 91–95.
- 32.
Modena, M.; Giannoni, A.; Aimo, A.; et al. Whole-exome sequencing to identify causative variants in juvenile sudden cardiac death. Hum. Genom. 2024, 18, 102.
- 33.
Grundy, S.M.; Stone, N.J.; Bailey, A.L.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2019, 73, e285–e350.
- 34.
Fadoni, J.; Santos, A.; Amorim, A.; et al. Sudden Cardiac Death: The Role of Molecular Autopsy with Next-Generation Sequencing. Diagnostics 2025, 15, 460.
- 35.
Isbister, J.C.; Semsarian, C. The role of the molecular autopsy in sudden cardiac death in young individuals. Nat. Rev. Cardiol. 2024, 21, 215–216.
- 36.
Coll, M.; Alcalde, M.; Fernandez-Falgueras, A.; et al. Value of Molecular Autopsy in Suspected Sudden Cardiac Death in the Young. J. Mol. Diagn. 2025, 27, 859–868.
- 37.
Wang, J.; Hong, L.; Li, Y.; et al. Case Report: Lethal neonatal hypertrophic cardiomyopathy from compound heterozygous MYBPC3 variants. Front. Cardiovasc. Med. 2025, 12, 1726463.

This work is licensed under a Creative Commons Attribution 4.0 International License.