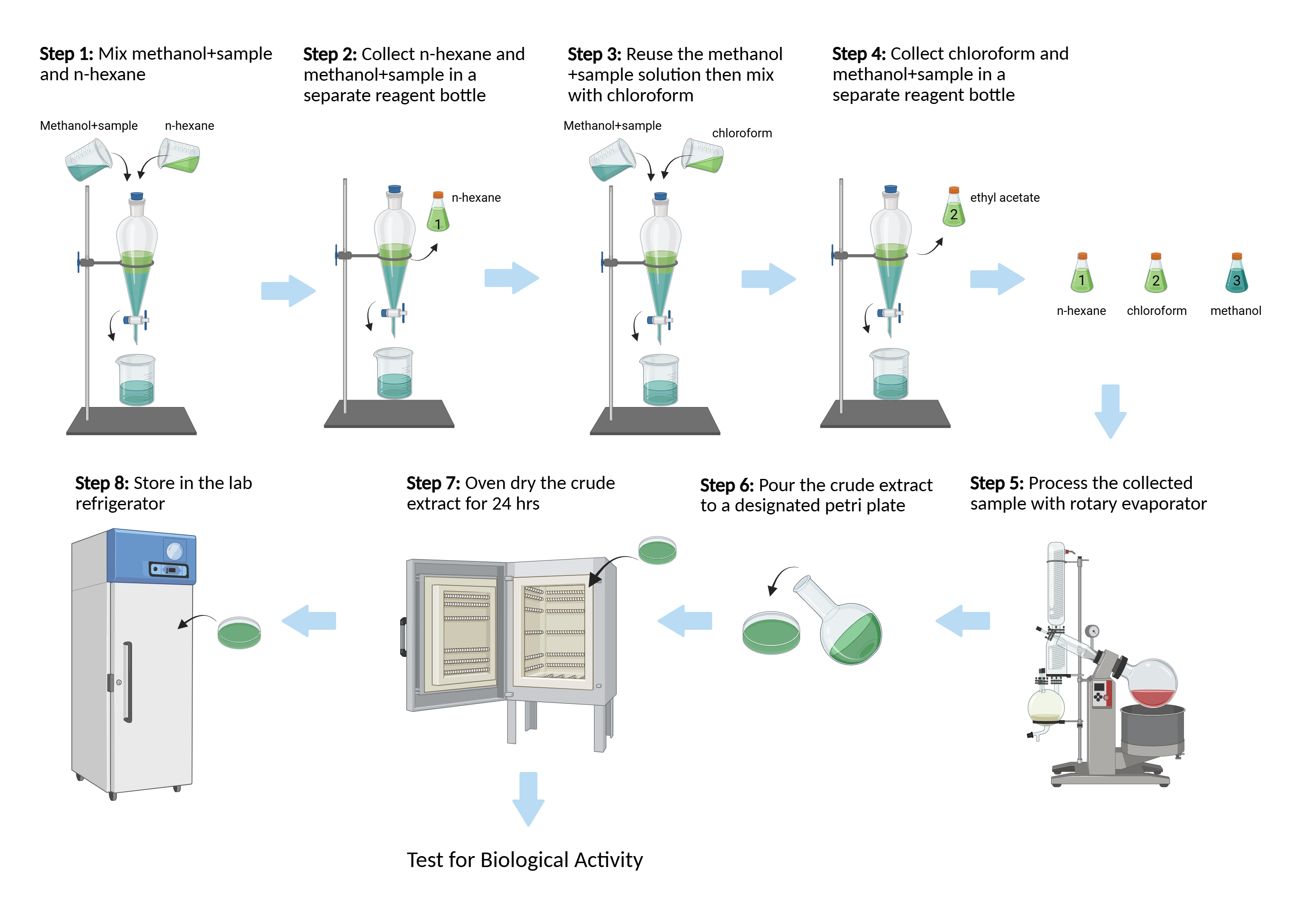
Background: Premna obtusifolia (Lamiaceae) and Oxalis corniculata L. (Oxalidaceae) are two ethnomedicinal plants widely used in Bangladesh for the management of infections and inflammatory disorders. Although they belong to distinct botanical families and differ chemically, P. obtusifolia being rich in diterpenoids and O. corniculata abundant in flavonoids, both are traditionally associated with antioxidant and antimicrobial effects. The present study comparatively evaluated their methanolic extracts and solvent fractions using DPPH radical scavenging assay, total phenolic content (TPC) determination, and disc diffusion antimicrobial screening. Materials and Methods: After powdering the leaves and extracting them with methanol, the fractions were separated into n-hexane and chloroform. Using the DPPH radical scavenging experiment, antioxidant activity was evaluated, and IC50 values were computed. This was done using the disc diffusion method to measure antimicrobial activity. To determine the total phenolic content (TPC), the Folin–Ciocalteu technique was employed. To provide mechanistic insight, previously reported 14 phytochemicals from these species were subjected to molecular docking against human carbonyl reductase 1 (CBR-1; PDB ID: 4Z3D) and Staphylococcus aureus dihydrofolate reductase (DHFR; PDB ID: 2W9S). Results: The crude methanolic extract of P. obtusifolia showed an IC50 of 31 µg/mL, while O. corniculata exhibited stronger antioxidant activity (IC50 = 15.61 µg/mL). Both extracts demonstrated significant antimicrobial effects, whereas n-hexane and chloroform fractions showed moderate antimicrobial activity. The antioxidant potential of P. obtusifolia and O. corniculata was correlated with TPC values of 185.82 mg GAE/g and 154.90 mg GAE/g, respectively. Diosmetin-7-O-β-D-glucopyranoside, Quercetin, Arucadiol, and Swertisin demonstrated binding affinities comparable to reference ligands under the same docking conditions, according to molecular docking, indicating their potential as antioxidants and antimicrobials. Conclusion: The substantial antibacterial and antioxidant properties of the crude methanolic extracts support the traditional usage of Oxalis corniculata and Premna obtusifolia and suggest that these plants may be used as natural sources of bioactive chemicals for medicinal purposes.
- Open Access
- Article
Exploring the Therapeutic Potential of Premna obtusifolia and Oxalis corniculata: A Combined in Vitro Bioassay and Molecular Docking Study
- Arifa Sultana 1,
- Rajib Das 2,3,
- Amit Roy 1,
- Mst. Nowsad Zahan Sathi 4,
- Najmus Sakib Minhaj 4,
- Md. Reaz Uddin 5,6,
- Sitesh Chandra Bachar 1,*
Author Information
Received: 31 Dec 2025 | Revised: 17 Feb 2026 | Accepted: 26 Feb 2026 | Published: 16 Mar 2026
Abstract
Graphical Abstract
Keywords
Premna obtusifolia | Oxalis corniculate | antioxidant | antimicrobial | total phenolic content
References
- 1.
Akinmoladun, A.C.; Ibukun, E.O.; Afor, E.; et al. Phytochemical Constituent and Antioxidant Activity of Extract from the Leaves of Ocimum Gratissimum. Sci. Res. Essay 2007, 2, 163–166.
- 2.
Hall, C.A.; Cuppet, S.L. Antioxidant Methodology: In Vivo and in Vitro Concepts; AOCS Press: Champaign, IL, USA, 1997.
- 3.
Barry, A.L.; Hoeprich, P.D.; Saubolle, M.A. The Antimicrobic Susceptibility Test: Principles and Practices; Lea & Febiger: Philadelphia, PA, USA, 1976.
- 4.
Arbab, S.; Ullah, H.; Bano, I.; et al. Evaluation of in Vitro Antibacterial Effect of Essential Oil and Some Herbal Plant Extract Used against Mastitis Pathogens. Vet. Med. Sci. 2022, 8, 2655–2661. https://doi.org/10.1002/vms3.959.
- 5.
Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; et al. Natural Products in Drug Discovery: Advances and Opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216.
- 6.
Gulcin, İ.; Alwasel, S.H. DPPH Radical Scavenging Assay. Processes 2023, 11, 2248. https://doi.org/10.3390/pr11082248.
- 7.
Harminder; Singh, V.; Chaudhary, A.K. A Review on the Taxonomy, Ethnobotany, Chemistry and Pharmacology of Oroxylum Indicum Vent. Indian J. Pharm. Sci. 2011, 73, 483–490. https://doi.org/10.4103/0250-474X.98981.
- 8.
Christenhusz, M.J.M.; Byng, J.W. The Number of Known Plants Species in the World and Its Annual Increase. Phytotaxa 2016, 261, 201–217. https://doi.org/10.11646/phytotaxa.261.3.1.
- 9.
Dias, D.A.; Urban, S.; Roessner, U. A Historical Overview of Natural Products in Drug Discovery. Metabolites 2012, 2, 303–336. https://doi.org/10.3390/metabo2020303.
- 10.
El Beyrouthy, M.; Dhifi, W.; Arnold-Apostolides, N. Ethnopharmacological Survey of the Indigenous Lamiaceae from Lebanon. Acta Hortic. 2013, 997, 257–276. https://doi.org/10.17660/actahortic.2013.997.33.
- 11.
Elisabetsky, E.; Etkin, N.L. Ethnopharmacology. In Encyclopedia of Life Support Systems; EOLSS Publisher: Oxford, UK; 2009; Volume 1, p. 165.
- 12.
Ghani, A. Medicinal Plants of Bangladesh with Chemical Constituents and Uses, 2nd ed.; Asiatic Society of Bangladesh: Dhaka, Bangladesh, 2003.
- 13.
Halliwell, B.; Gutteridge, J.M.C. The Definition and Measurement of Antioxidants in Biological Systems. Free Radic. Biol. Med. 1995, 18, 125–126. https://doi.org/10.1016/0891-5849(95)91457-3.
- 14.
Harley, R.M.; Atkins, S.; Budantsev, A.L.; et al. Flowering Plants Dicotyledons: Lamiales (except Acanthaceae Including Avicenniaceae). In The Families and Genera of Vascular Plants; Springer: Berlin/Heidelberg, Germany, 2004; Volume 7, pp. 167–275.
- 15.
Ingle, K.P.; Deshmukh, A.G.; Padole, D.A.; et al. Phytochemicals: Extraction Methods, Identification and Detection of Bioactive Compounds from Plant Extracts. J. Pharmacogn. Phytochem. 2017, 6, 32–36.
- 16.
Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in Vitro Evaluating Antimicrobial Activity: A Review. J. Pharm. Anal. 2016, 6, 71–79. https://doi.org/10.1016/j.jpha.2015.11.005.
- 17.
Buzayan, M.M.; El-Garbulli, F.R. Antibacterial Activity of Medicinal Aqueous Plant Extracts against Mycobacterium tuberculosis. Malays. J. Microbiol. 2012, 8, 203–206.
- 18.
Norhana, M.N.W.; Poole, S.E.; Deeth, H.C.; et al. Effects of Bilimbi (Averrhoa bilimbi L.) and Tamarind (Tamarindus indica L.) Juice on Listeria monocytogenes Scott A and Salmonella Typhimurium ATCC 14028 and the Sensory Properties of Raw Shrimps. Int. J. Food Microbiol. 2009, 136, 88–94. https://doi.org/10.1016/j.ijfoodmicro.2009.09.011.
- 19.
Rai, P.K.; Jaiswal, D.; Singh, R.K.; et al. Glycemic Properties of Trichosanthes dioica Leaves. Pharm. Biol. 2008, 46, 894–899. https://doi.org/10.1080/13880200802370167.
- 20.
Selvi, S.; Polat, R.; Çakilcioğlu, U.; et al. An Ethnobotanical Review on Medicinal Plants of the Lamiaceae Family in Turkey. Turk. J. Bot. 2022, 46, 283–332. https://doi.org/10.55730/1300-008X.2712.
- 21.
Sánchez-Moreno, C. Methods Used to Evaluate the Free Radical Scavenging Activity in Foods and Biological Systems. Food Sci. Technol. Int. 2002, 8, 121–137. https://doi.org/10.1106/108201302026770.
- 22.
Pan, S.Y.; Zhou, S.F.; Gao, S.H.; et al. New Perspectives on How to Discover Drugs from Herbal Medicines: CAM’S Outstanding Contribution to Modern Therapeutics. Evid. Based Complement. Altern. Med. 2013, 2013, 627375. https://doi.org/10.1155/2013/627375.
- 23.
Nikolaeva, T.N.; Lapshin, P.V.; Zagoskina, N.V. Method for Determining the Total Content of Phenolic Compounds in Plant Extracts with Folin–Denis Reagent and Folin–Ciocalteu Reagent: Modification and Comparison. Russ. J. Bioorg. Chem. 2022, 48, 1519–1525. https://doi.org/10.1134/S1068162022070214.
- 24.
Absar, K.M.B.; Md. Rifat, H.B.S.; Das, S.; et al. Phytochemical and Pharmacological Properties of Oxalis corniculata: A Review. Trop. J. Phytochem. Pharm. Sci. 2024, 3, 364–374. https://doi.org/10.26538/tjpps/v3i7.2.
- 25.
Salae, A.W.; Rodjun, A.; Karalai, C.; et al. Potential Anti-Inflammatory Diterpenes from Premna obtusifolia. Tetrahedron 2012, 68, 819–829. https://doi.org/10.1016/j.tet.2011.11.058.
- 26.
Borah, A.; Yadav, R. Evaluation of Antioxidant Activity of Different Solvent Extracts of Oxalis corniculata L. J. Pharm. Res. 2012, 5, 91–93.
- 27.
Ahmed, D.; Zara, S.; Baig, H. In Vitro Analysis of Antioxidant Activities of Oxalis corniculata Linn. Fractions in Various Solvents. Afr. J. Tradit. Complement. Altern. Med. 2012, 10, 158–165. https://doi.org/10.4314/ajtcam.v10i1.21.
- 28.
Zhong, T.; He, J.; Zhao, H.; et al. Oxalis corniculata L. As a Source of Natural Antioxidants: Phytochemistry, Bioactivities, and Application Potential. Antioxidants 2025, 14, 1352. https://doi.org/10.3390/antiox14111352.
- 29.
Palariya, D.; Singh, A.; Dhami, A.; et al. Phytochemical Analysis and Screening of Antioxidant, Antibacterial and Antiinflammatory Activity of Essential Oil of Premna mucronata Roxb. Leaves. Trends Phytochem. Res. 2019, 3, 275–286.
- 30.
Simamora, A.; Santoso, A.W.; Timotius, K.H.; et al. Antioxidant Activity, Enzyme Inhibition Potentials, and Phytochemical Profiling of Premna serratifolia L. Leaf Extracts. Int. J. Food Sci. 2020, 2020, 3436940. https://doi.org/10.1155/2020/3436940.
- 31.
Mali, P. Pharmacological Potentials of Premna integrifolia L. Anc. Sci. Life 2016, 35, 132–142. https://doi.org/10.4103/0257-7941.179864.
- 32.
Hossan, M.S.; Hanif, A.; Khan, M.; et al. Ethnobotanical Survey of the Tripura Tribe of Bangladesh. Am. J. Sustain. Agric. 2009, 3, 253–261.
- 33.
Rahman, M.; Uddin, S. Medicinal Plants Used by Chakma Tribe in Hill Tracts Districts of Bangladesh. Indian J. Tradit. Knowl. 2007, 6, 508–517.
- 34.
Al Aboody, M.S.; Mickymaray, S. Anti-Fungal Efficacy and Mechanisms of Flavonoids. Antibiotics 2020, 9, 45. https://doi.org/10.3390/antibiotics9020045.
- 35.
Nyemb, J.N.; Njock, G.B.B.; Demissie, T.B.; et al. Bioactive Constituents from Gardenia aqualla (Rubiaceae) Stem Bark as Promising Antibacterial Agents: In Vitro and in Silico Insights. Microb. Pathog. 2026, 210, 108182. https://doi.org/10.1016/j.micpath.2025.108182.
- 36.
Leutcha, P.B.; Mamoudou, H.; Nganso Ditchou, Y.O.; et al. Flavonoids and Other Constituents from Jacaranda mimosifolia: In Vitro Analysis, Molecular Docking, and Molecular Dynamic Simulations of Antioxidant and Anti-Inflammatory Activities. Biomed. Pharmacother. 2025, 182, 117768. https://doi.org/10.1016/j.biopha.2024.117768.
- 37.
Elfita, E.; Muharni, M.; Munawar, M.; et al. Isolation of Antioxidant Compound from Endophytic Fungi Acremonium Sp. from the Twigs of Kandis Gajah. MAKARA Sci. Ser. 2012, 16, 46–50. https://doi.org/10.7454/mss.v16i1.1280.
- 38.
Su, R.; Ma, Q.; Zhao, Y.; et al. Deciphering the Pharmacological Mechanism of Compound Purpura Decoction in Treating Henoch-Schonlein Purpura by Network Pharmacology, Molecular Docking and Experimental Validation. Chem. Biodivers. 2025, 22, e202402793. https://doi.org/10.1002/cbdv.202402793.

This work is licensed under a Creative Commons Attribution 4.0 International License.


