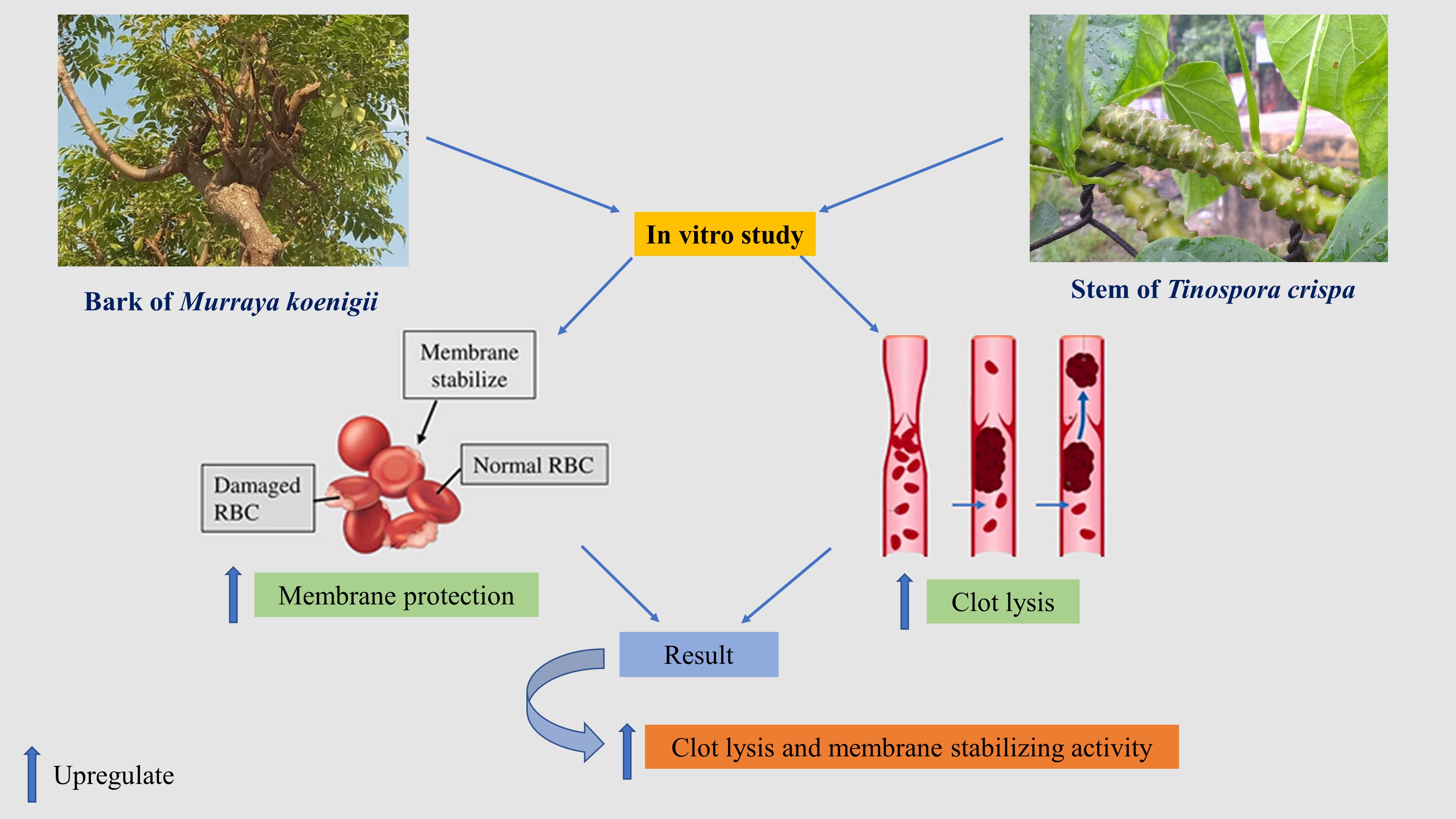
Thrombosis and inflammation are interrelated pathological processes that play a central role in the development of cardiovascular complications. This study evaluated the thrombolytic and membrane stabilizing activities of the bark of Murraya koenigiiand the stem of Tinospora crispa, two widely used medicinal plants with recognized traditional medicinal value. Crude methanolic extracts of both plant materials were first prepared. These extracts were then subjected to solvent–solvent partitioning to yield petroleum ether, dichloromethane, ethyl acetate, and aqueous fractions. Thrombolytic potential was investigated using an in vitroclot lysis assay, while membrane stabilizing activity was assessed through hypotonic solution and heat-induced hemolysis models employing human erythrocytes. The ethyl acetate fraction of M. koenigii bark exhibited the highest thrombolytic activity among the tested samples, whereas fractions derived from T. crispastem demonstrated moderate clot lysis. In membrane stabilization assays, M. koenigiibark extracts showed strong inhibition of hypotonic solution–induced hemolysis, while T. crispastem fractions were more effective against heat-induced hemolysis. The observed bioactivities varied across solvent fractions, indicating the influence of solvent polarity on the distribution of active constituents. These findings support the traditional use of M. koenigiiand T. crispa and suggest their potential as sources of plant-derived thrombolytic and membrane stabilizing agents for managing inflammation-associated thrombotic disorders.
- Open Access
- Article
Thrombolytic and Membrane Stabilizing Activities of Murraya koenigii Bark and Tinospora crispa Stem
Author Information
Received: 05 Jan 2026 | Revised: 26 Feb 2026 | Accepted: 04 Mar 2026 | Published: 12 Mar 2026
Abstract
Graphical Abstract
Keywords
Murraya koenigii | Tinospora crispa | thrombolytic activity | membrane stabilizing activity | medicinal plants | inflammation | erythrocyte hemolysis
References
- 1.
Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. https://doi.org/10.1021/acs.jnatprod.9b01285.
- 2.
Poli, M.A.; Hossain, M.J.; Kholil, I.; et al. Traditional Medicine Use and Associated Factors in Chronic Patients in Jamalpur, Bangladesh: A Cross-Sectional Study. Front. Public Health 2025, 13, 1548728. https://doi.org/10.3389/fpubh.2025.1548728.
- 3.
Libby, P.; Ridker, P.M.; Hansson, G.K.; et al. Inflammation in Atherosclerosis: From Pathophysiology to Practice. J. Am. Coll. Cardiol. 2009, 54, 2129–2138. https://doi.org/10.1016/j.jacc.2009.09.009.
- 4.
Theofilis, P.; Sagris, M.; Antonopoulos, A.S.; et al. Inflammatory Mediators of Platelet Activation: Focus on Atherosclerosis and COVID-19. Int. J. Mol. Sci. 2021, 22, 11170. https://doi.org/10.3390/ijms222011170.
- 5.
Shebuski, R.J.; Kilgore, K.S. Role of Inflammatory Mediators in Thrombogenesis. J. Pharmacol. Exp. Ther. 2002, 300, 729–735. https://doi.org/10.1124/jpet.300.3.729.
- 6.
Tisdale, J.E.; Colucci, R.D.; Ujhelyi, M.R.; et al. Evaluation and Comparison of the Adverse Effects of Streptokinase and Alteplase. Pharmacotherapy 1992, 12, 440–444.
- 7.
Anosike, C.A.; Obidoa, O.; Ezeanyika, L.U. Membrane Stabilization as a Mechanism of the Anti-Inflammatory Activity of Methanol Extract of Garden Egg (Solanum aethiopicum). DARU J. Pharm. Sci. 2012, 20, 76. https://doi.org/10.1186/2008-2231-20-76.
- 8.
Aidoo, D.B.; Konja, D.; Henneh, I.T.; et al. Protective Effect of Bergapten against Human Erythrocyte Hemolysis and Protein Denaturation In Vitro. Int. J. Inflamm. 2021, 2021, 1279359. https://doi.org/10.1155/2021/1279359.
- 9.
Suthar, P.; Kumar, S.; Kumar, V.; et al. Murraya koenigii (L.) Spreng: Speculative Ethnobotanical Perspectives of Ubiquitous Herb with Versatile Nutra/Functional Properties. S. Afr. J. Bot. 2022, 145, 111–134. https://doi.org/10.1016/j.sajb.2021.11.025.
- 10.
Nandy, S.; Das, S. Unveiling the Diverse Medicinal Properties of Murraya koenigii. Sci. Phytochem. 2023, 2, 211–225. https://doi.org/10.58920/sciphy02020107.
- 11.
Tan, M.A.; Sharma, N.; An, S.S.A. Multi-Target Approach of Murraya koenigii Leaves in Treating Neurodegenerative Diseases. Pharmaceuticals 2022, 15, 188. https://doi.org/10.3390/ph15020188.
- 12.
Jachak, S.; Singh, M.; Ahirrao, P.; et al. Murraya koenigii (L.) Spreng. as a Natural Intervention for Diabesity: A Review. Curr. Pharm. Des. 2024, 30, 3255–3275. https://doi.org/10.2174/0113816128304471240801183021.
- 13.
Haque, E.; Bari, M.S.; Khandokar, L.; et al. An Updated and Comprehensive Review on the Ethnomedicinal Uses, Phytochemistry, Pharmacological Activity and Toxicological Profile of Tinospora crispa (L.) Hook. f. & Thomson. Phytochem. Rev. 2023, 22, 211–273. https://doi.org/10.1007/s11101-022-09843-y.
- 14.
Ahmad, W.; Jantan, I.; Bukhari, S.N.A. Tinospora crispa (L.) Hook. f. & Thomson: A Review of Its Ethnobotanical, Phytochemical, and Pharmacological Aspects. Front. Pharmacol. 2016, 7, 59. https://doi.org/10.3389/fphar.2016.00059.
- 15.
Reza, A.S.M.A.; Haque, M.A.; Sarker, J.; et al. Antiproliferative and Antioxidant Potentials of Bioactive Edible Vegetable Fraction of Achyranthes Ferruginea Roxb. in Cancer Cell Line. Food Sci. Nutr. 2021, 9, 3777–3805. https://doi.org/10.1002/fsn3.2343.
- 16.
Prasad, S.; Kashyap, R.S.; Deopujari, J.Y.; et al. Development of an in Vitro Model to Study Clot Lysis Activity of Thrombolytic Drugs. Thromb. J. 2006, 4, 14. https://doi.org/10.1186/1477-9560-4-14.
- 17.
Sonter, S.; Mishra, S.; Dwivedi, M.K.; Singh, P.K. Chemical Profiling, In Vitro Antioxidant, Membrane Stabilizing and Antimicrobial Properties of Wild Growing Murraya Paniculata from Amarkantak (M.P.). Sci. Rep. 2021, 11, 9691, https://doi.org/10.1038/s41598-021-87404-7.
- 18.
Xu, H.; Yang, J.; Wei, Z.; et al. Oxidative Stress in Vascular Surgical Diseases: Mechanisms, Impacts and Therapeutic Perspectives. Front. Pharmacol. 2025, 16, 1527684. https://doi.org/10.3389/fphar.2025.1527684.
- 19.
Franyoto, Y.D.; Nurrochmad, A.; Fakhrudin, N. Murraya koenigii L. Spreng.: An Updated Review of Chemical Composition, Pharmacological Effects, and Toxicity Studies. J. Appl. Pharm. Sci. 2024, 14, 011–027. https://doi.org/10.7324/JAPS.2024.169254.
- 20.
Balakrishnan, R.; Vijayraja, D.; Jo, S.-H.; et al. Medicinal Profile, Phytochemistry, and Pharmacological Activities of Murraya koenigii and Its Primary Bioactive Compounds. Antioxidants 2020, 9, 101. https://doi.org/10.3390/antiox9020101.
- 21.
Shinde, U.A.; Phadke, A.S.; Nair, A.M.; et al. Membrane Stabilizing Activity—A Possible Mechanism of Action for the Anti-Inflammatory Activity of Cedrus deodara Wood Oil. Fitoterapia 1999, 70, 251–257. https://doi.org/10.1016/S0367-326X(99)00030-1.
- 22.
Arora, A.; Byrem, T.M.; Nair, M.G.; et al. Modulation of Liposomal Membrane Fluidity by Flavonoids and Isoflavonoids. Arch. Biochem. Biophys. 2000, 373, 102–109. https://doi.org/10.1006/abbi.1999.1525.
- 23.
Niu, Y.; Xiang, Y. An Overview of Biomembrane Functions in Plant Responses to High-Temperature Stress. Front. Plant Sci. 2018, 9, 915. https://doi.org/10.3389/fpls.2018.00915.

This work is licensed under a Creative Commons Attribution 4.0 International License.


