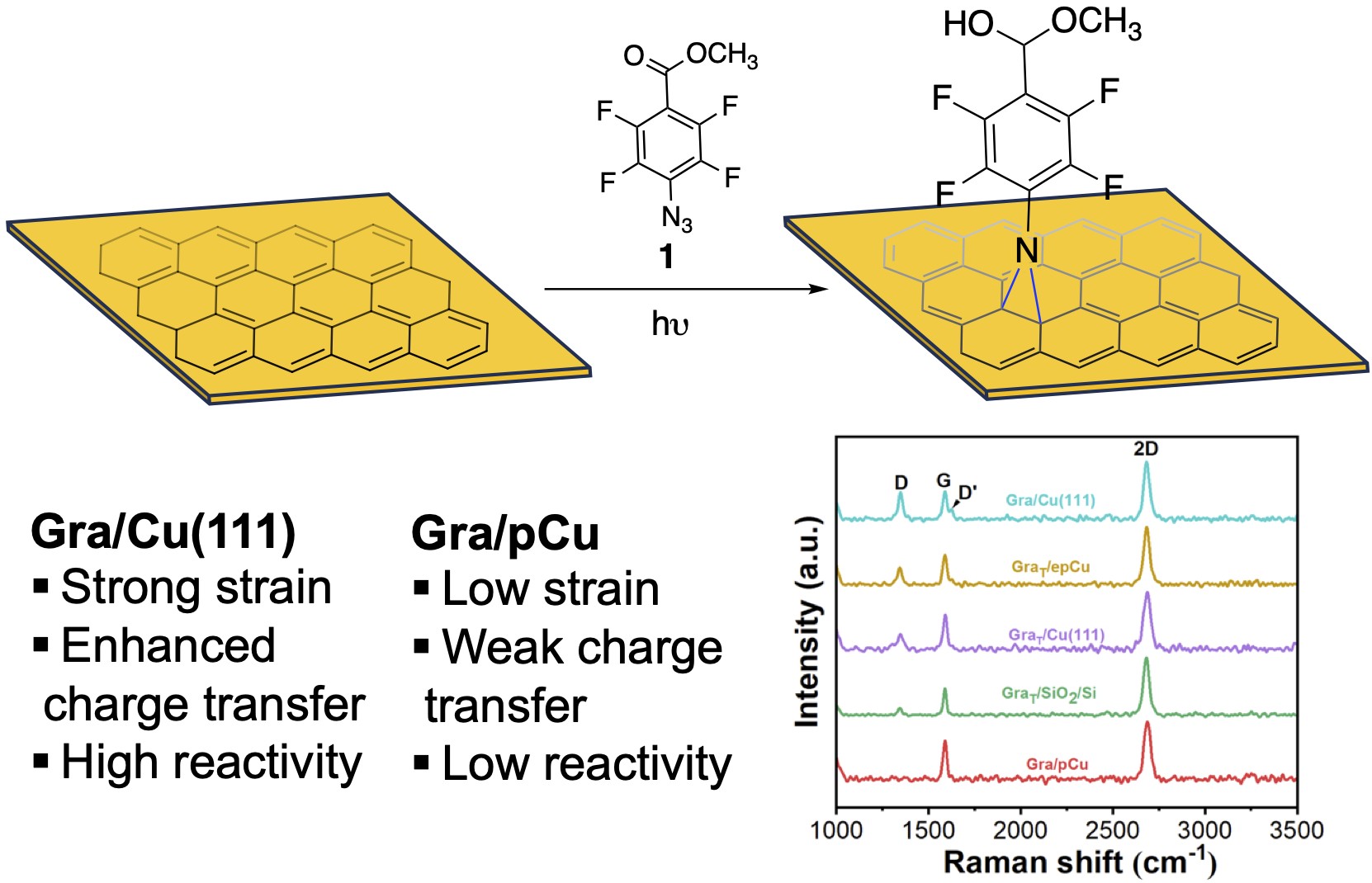
Effective methods for the covalent functionalization of pristine graphene are limited due to its low chemical reactivity. We developed a covalent chemistry to functionalize graphene using perfluorophenyl azide (PFPA) and demonstrated that the reactivity of graphene can be enhanced by metal substrates. In this work, we investigated the impact of crystalline lattice and morphology of Cu on the reactivity of graphene with PFPA. Graphene grown on single crystalline Cu(111) (Gra/Cu(111)) exhibited the highest degree of functionalization, as evidenced by the largest increase in the D-band intensity obtained from Raman spectroscopy and Raman mapping, along with a low crystallite domain size LD and a high defect density nD. In contrast, graphene grown on electropolished polycrystalline Cu (Gra/pCu) showed minimal reactivity toward PFPA reaction. Further Raman analyses demonstrate that Cu(111) induces stronger charge transfer and higher strain in graphene compared to pCu, primarily due to superior lattice matching between graphene and Cu(111), leading to higher reactivity of Gra/Cu(111) toward electron-deficient perfluorophenyl nitrene. Upon transfer of graphene grown on Cu(111) to other substrates, including Cu(111), electropolished pCu, and silicon wafer, the reactivity decreased, and the reactivity of transferred graphene correlates mainly with surface roughness. The decreased reactivity is attributed to weakened graphene–substrate charge interactions after transfer. These findings establish substrate engineering as an effective and non-invasive method for tuning the chemical reactivity of graphene without the need for harsh activation conditions.
- Open Access
- Article
Reactivity of Perfluorophenyl Azide with Graphene Supported on Cu: Effect of Substrate Crystallinity
- Jia Tu,
- Mingdi Yan *
Author Information
Received: 01 Mar 2026 | Revised: 14 Mar 2026 | Accepted: 16 Mar 2026 | Published: 18 Mar 2026
Abstract
Graphical Abstract
Keywords
graphene | covalent functionalization | perfluorophenyl azide | substrate effect | Cu(111) | polycrystalline Cu
References
- 1.
Liu, J.; Tang, J.; Gooding, J.J. Strategies for chemical modification of graphene and applications of chemically modified graphene. J. Mater. Chem. 2012, 22, 12435–12452.
- 2.
Park, J.; Yan, M. Covalent functionalization of graphene with reactive intermediates. Acc. Chem. Res. 2013, 46, 181–189.
- 3.
Stergiou, A.; Cantón-Vitoria, R.; Psarrou, M.N.; Economopoulos, S.P.; Tagmatarchis, N. Functionalized graphene and targeted applications–Highlighting the road from chemistry to applications. Prog. Mater Sci. 2020, 114, 100683.
- 4.
Chua, C.K.; Pumera, M. Covalent chemistry on graphene. Chem. Soc. Rev. 2013, 42, 3222–3233.
- 5.
Yan, M.; Cai, S.X.; Keana, J.F.W. Photochemical and Thermal Reactions of C60 with N-Succinimidyl 4-Azido-2,3,5,6-tetrafluorobenzoate: A New Method for Functionalization of C60. J. Org. Chem. 2002, 59, 5951–5954.
- 6.
Liu, F.; Du, W.; Liang, Q.; Wang, Y.; Zhang, J.; Zhao, J.; Zhu, S. Synthesis of 4-aziridino[C60]fullerene-1,8-naphthalimide (C60-NI dyads) and their photophysical properties. Tetrahedron 2010, 66, 5467–5471.
- 7.
Khalilov, M.; Tulyabaev, A.R.; Akhmetov, A.R.; Tuktarov, A.R. Synthesis and 13C NMR features of N-substituted aziridino[60]fullerenes. Russ. Chem. Bull. 2016, 64, 2725–2730.
- 8.
Gross, A.J.; Yu, S.S.; Downard, A.J. Two-component mixed and patterned films on carbon surfaces through the photografting of arylazides. Langmuir 2010, 26, 7285–7292.
- 9.
Tanaka, M.; Sawaguchi, T.; Sato, Y.; Yoshioka, K.; Niwa, O. Surface modification of GC and HOPG with diazonium, amine, azide, and olefin derivatives. Langmuir 2011, 27, 170–178.
- 10.
Choi, J.; Kim, K.-j.; Kim, B.; Lee, H.; Kim, S. Covalent functionalization of epitaxial graphene by azidotrimethylsilane. J. Phys. Chem. C 2009, 113, 9433–9435.
- 11.
Strom, T.A.; Dillon, E.P.; Hamilton, C.E.; Barron, A.R. Nitrene addition to exfoliated graphene: A one-step route to highly functionalized graphene. Chem. Commun. 2010, 46, 4097–4099.
- 12.
Vadukumpully, S.; Gupta, J.; Zhang, Y.; Xu, G.Q.; Valiyaveettil, S. Functionalization of surfactant wrapped graphene nanosheets with alkylazides for enhanced dispersibility. Nanoscale 2011, 3, 303–308.
- 13.
Poe, R.; Schnapp, K.; Young, M.J.; Grayzar, J.; Platz, M.S. Chemistry and kinetics of singlet pentafluorophenylnitrene. J. Am. Chem. Soc. 1992, 114, 5054–5067.
- 14.
Kotzyba-Hibert, F.; Kapfer, I.; Goeldner, M. Recent trends in photoaffinity labeling. Angew. Chem. Int. Ed. 1995, 34, 1296–1312.
- 15.
Denis, P.A.; Iribarne, F. Monolayer and bilayer graphene functionalized with nitrene radicals. J. Phys. Chem. C 2011, 115, 195–203.
- 16.
Bräse, S.; Gil, C.; Knepper, K.; Zimmermann, V. Organic azides: An exploding diversity of a unique class of compounds. Angew. Chem. Int. Ed. 2005, 44, 5188–5240.
- 17.
Wang, X.; Liu, L.H.; Ramstrom, O.; Yan, M. Engineering nanomaterial surfaces for biomedical applications. Exp. Biol. Med. 2009, 234, 1128–1139.
- 18.
Liu, L.H.; Yan, M. Perfluorophenyl azides: New applications in surface functionalization and nanomaterial synthesis. Acc. Chem. Res. 2010, 43, 1434–1443.
- 19.
Liu, L.-H.; Yan, M. Functionalization of pristine graphene with perfluorophenyl azides. J. Mater. Chem. 2011, 21, 3273–3276.
- 20.
Nagel, T.; Jurkiewicz, L.; Hauke, F.; Hirsch, A. Laser-Initiated Covalent Functionalization of Graphene Using Perfluorophenylazides with Local Addend-Binding Control. Phys. Status Solidi Basic Res. 2025, 263, 2500348.
- 21.
Yang, X.; Chen, F.; Kim, M.A.; Liu, H.; Wolf, L.M.; Yan, M. On the Reactivity Enhancement of Graphene by Metallic Substrates towards Aryl Nitrene Cycloadditions. Chem. Eur. J. 2021, 27, 7887–7896.
- 22.
Park, J.; Yang, X.; Wickramasinghe, D.; Sundhoro, M.; Orbey, N.; Chow, K.F.; Yan, M. Functionalization of pristine graphene for the synthesis of covalent graphene-polyaniline nanocomposite. RSC Adv. 2020, 10, 26486–26493.
- 23.
Park, J.; Jin, T.; Liu, C.; Li, G.; Yan, M. Three-dimensional graphene–TiO2 nanocomposite photocatalyst synthesized by covalent attachment. ACS Omega 2016, 1, 351–356.
- 24.
Liu, L.-H.; Yan, M. Simple method for the covalent immobilization of graphene. Nano Lett. 2009, 9, 3375–3378.
- 25.
Liu, L.-H.; Zorn, G.; Castner, D.G.; Solanki, R.; Lerner, M.M.; Yan, M. A simple and scalable route to wafer-size patterned graphene. J. Mater. Chem. 2010, 20, 5041–5046.
- 26.
Park, J.; Jayawardena, H.S.N.; Chen, X.; Jayawardana, K.W.; Sundhoro, M.; Ada, E.; Yan, M. A general method for the fabrication of graphene–nanoparticle hybrid material. Chem. Commun. 2015, 51, 2882–2885.
- 27.
Kong, N.; Park, J.; Yang, X.; Ramström, O.; Yan, M. Carbohydrate functionalization of few-layer graphene through microwave-assisted reaction of perfluorophenyl azide. ACS Appl. Bio Mater. 2018, 2, 284–291.
- 28.
Liu, L.-H.; Lerner, M.M.; Yan, M. Derivitization of pristine graphene with well-defined chemical functionalities. Nano Lett. 2010, 10, 3754–3756.
- 29.
Tu, J.; Yan, M. Enhancing the Chemical Reactivity of Graphene through Substrate Engineering. Small 2024, 2024, e2408116.
- 30.
Criado, A.; Melchionna, M.; Marchesan, S.; Prato, M. The Covalent Functionalization of Graphene on Substrates. Angew. Chem. Int. Ed. 2015, 54, 10734–10750.
- 31.
Yang, X.; Chen, F.; Kim, M.A.; Liu, H.; Wolf, L.M.; Yan, M. Using metal substrates to enhance the reactivity of graphene towards Diels-Alder reactions. Phys. Chem. Chem. Phys. 2022, 24, 20082–20093.
- 32.
Tu, J.; Zhou, W.; Wolf, L.M.; Yan, M. Inverse-Electron-Demand Diels-Alder Reaction of Tropone with Graphene Supported on Cu(111). Small 2025, 21, e03669.
- 33.
Zhang, X.; Luo, D.; Zhang, H.; Hwang, D.Y.; Park, S.O.; Li, B.-W.; Biswal, M.; Jiang, Y.; Huang, Y.; Kwak, S.K. Effect of copper substrate surface orientation on the reductive functionalization of graphene. Chem. Mater. 2019, 31, 8639–8648.
- 34.
Plsek, J.; Kovaricek, P.; Vales, V.; Kalbac, M. Tuning the Reactivity of Graphene by Surface Phase Orientation. Chem. Eur. J. 2017, 23, 1839–1845.
- 35.
Li, B.W.; Luo, D.; Zhu, L.; Zhang, X.; Jin, S.; Huang, M.; Ding, F.; Ruoff, R.S. Orientation-Dependent Strain Relaxation and Chemical Functionalization of Graphene on a Cu(111) Foil. Adv. Mater. 2018, 30, 1706504.
- 36.
Perera, H.A.; Raviranga, N.G.H.; Ramstrom, O.; Yan, M. Trehalose-Functionalized Magnetic Affinity Probe Provides Biochemical Evidence of Nanoparticle Internalization in Mycobacteria. ACS Infect. Dis. 2025, 11, 2847–2858.
- 37.
Tu, J.; Zhou, W.; Kiani, A.; Wolf, L.M.; Yan, M. Chemical Vapor Deposition of Monolayer Graphene on Centimeter-Sized Cu(111) for Nanoelectronics Applications. ACS Appl. Nano Mater. 2025, 8, 4926–4939.
- 38.
Yang, X.; Yan, M. Removing contaminants from transferred CVD graphene. Nano Res. 2020, 13, 599–610.
- 39.
Ferrari, A.C.; Basko, D.M. Raman spectroscopy as a versatile tool for studying the properties of graphene. Nat. Nanotechnol. 2013, 8, 235–246.
- 40.
Cancado, L.G.; Jorio, A.; Ferreira, E.H.; Stavale, F.; Achete, C.A.; Capaz, R.B.; Moutinho, M.V.; Lombardo, A.; Kulmala, T.S.; Ferrari, A.C. Quantifying defects in graphene via Raman spectroscopy at different excitation energies. Nano Lett. 2011, 11, 3190–3196.
- 41.
Lucchese, M.M.; Stavale, F.; Ferreira, E.M.; Vilani, C.; Moutinho, M.d.O.; Capaz, R.B.; Achete, C.A.; Jorio, A. Quantifying ion-induced defects and Raman relaxation length in graphene. Carbon 2010, 48, 1592–1597.
- 42.
Lee, J.E.; Ahn, G.; Shim, J.; Lee, Y.S.; Ryu, S. Optical separation of mechanical strain from charge doping in graphene. Nat. Commun. 2012, 3, 1024.
- 43.
Wang, Q.H.; Jin, Z.; Kim, K.K.; Hilmer, A.J.; Paulus, G.L.; Shih, C.J.; Ham, M.H.; Sanchez-Yamagishi, J.D.; Watanabe, K.; Taniguchi, T.; et al. Understanding and controlling the substrate effect on graphene electron-transfer chemistry via reactivity imprint lithography. Nat. Chem. 2012, 4, 724–732.
- 44.
Wu, Q.; Wu, Y.; Hao, Y.; Geng, J.; Charlton, M.; Chen, S.; Ren, Y.; Ji, H.; Li, H.; Boukhvalov, D.W.; et al. Selective surface functionalization at regions of high local curvature in graphene. Chem. Commun. 2013, 49, 677–679.
- 45.
Deng, S.; Rhee, D.; Lee, W.K.; Che, S.; Keisham, B.; Berry, V.; Odom, T.W. Graphene Wrinkles Enable Spatially Defined Chemistry. Nano Lett. 2019, 19, 5640–5646.
- 46.
Giovannetti, G.; Khomyakov, P.A.; Brocks, G.; Karpan, V.V.; van den Brink, J.; Kelly, P.J. Doping graphene with metal contacts. Phys. Rev. Lett. 2008, 101, 026803.
- 47.
Kong, L.; Bjelkevig, C.; Gaddam, S.; Zhou, M.; Lee, Y.H.; Han, G.H.; Jeong, H.K.; Wu, N.; Zhang, Z.; Xiao, J. Graphene/substrate charge transfer characterized by inverse photoelectron spectroscopy. J. Phys. Chem. C 2010, 114, 21618–21624.
- 48.
Khomyakov, P.; Giovannetti, G.; Rusu, P.; Brocks, G.V.; Van den Brink, J.; Kelly, P.J. First-principles study of the interaction and charge transfer between graphene and metals. Phys. Rev. B. 2009, 79, 195425.
- 49.
Chen, C.; Avila, J.; Asensio, M.C. Chemical and electronic structure imaging of graphene on Cu: A NanoARPES study. J. Phys. Condens. Matter 2017, 29, 183001.
- 50.
Zhou, L.; Liao, L.; Wang, J.; Yu, J.; Li, D.; Xie, Q.; Liu, Z.; Yang, Y.; Guo, X.; Liu, Z. Substrate-Induced Graphene Chemistry for 2D Superlattices with Tunable Periodicities. Adv. Mater. 2016, 28, 2148–2154.
- 51.
Cullen, W.G.; Yamamoto, M.; Burson, K.M.; Chen, J.-H.; Jang, C.; Li, L.; Fuhrer, M.S.; Williams, E.D. High-fidelity conformation of graphene to SiO 2 topographic features. Phys. Rev. Lett. 2010, 105, 215504.
- 52.
Li, T.; Zhang, Z. Substrate-regulated morphology of graphene. J. Phys. D Appl. Phys. 2010, 43, 075303.
- 53.
Zhang, Y.; Heiranian, M.; Janicek, B.; Budrikis, Z.; Zapperi, S.; Huang, P.Y.; Johnson, H.T.; Aluru, N.R.; Lyding, J.W.; Mason, N. Strain modulation of graphene by nanoscale substrate curvatures: A molecular view. Nano Lett. 2018, 18, 2098–2104.
- 54.
Negrito, M.; Elinski, M.B.; Hawthorne, N.; Pedley, M.P.; Han, M.; Sheldon, M.; Espinosa-Marzal, R.M.; Batteas, J.D. Using Patterned Self-Assembled Monolayers to Tune Graphene–Substrate Interactions. Langmuir 2021, 37, 9996–10005.

This work is licensed under a Creative Commons Attribution 4.0 International License.


