In many cases, the presence of a shell of surface oxide detrimentally alters the properties of nanomaterials, rendering them ineffective for the targeted applications. This issue is yet to be addressed because the growth and removal of oxide(s) from the surface of nanomaterials is still poorly understood. In this article, we used vapor-phase-derived Ag nanocrystals with controlled sizes to elucidate the mechanisms of surface oxidation by exposing them to ambient atmosphere, aqueous environments, and ethylene glycol. Our experimental data suggested that the thickness of an oxide shell directly correlated with the extent of plasmon damping, and the optical properties of nanocrystals below 8 nm in size were more drastically affected by the presence of an oxide shell relative to larger counterparts. In addition, we found that aqueous environments with a relatively high pH gave rise to higher rates of oxidation. The time-dependent changes in optical properties were also validated using theoretical simulations based on the discrete dipole approximation. We concluded this article by discussing three different strategies for effectively removing the surface oxide, including decomposition through thermal annealing, wet-chemical reduction through the addition of a reducing agent, and decomposition and/or reduction by electron irradiation.
- Open Access
- Article
Understanding the Surface Oxidation of Ag Nanocrystals under Different Environmental Conditions
Author Information
Received: 03 Mar 2026 | Revised: 17 Mar 2026 | Accepted: 20 Mar 2026 | Published: 23 Mar 2026
Abstract
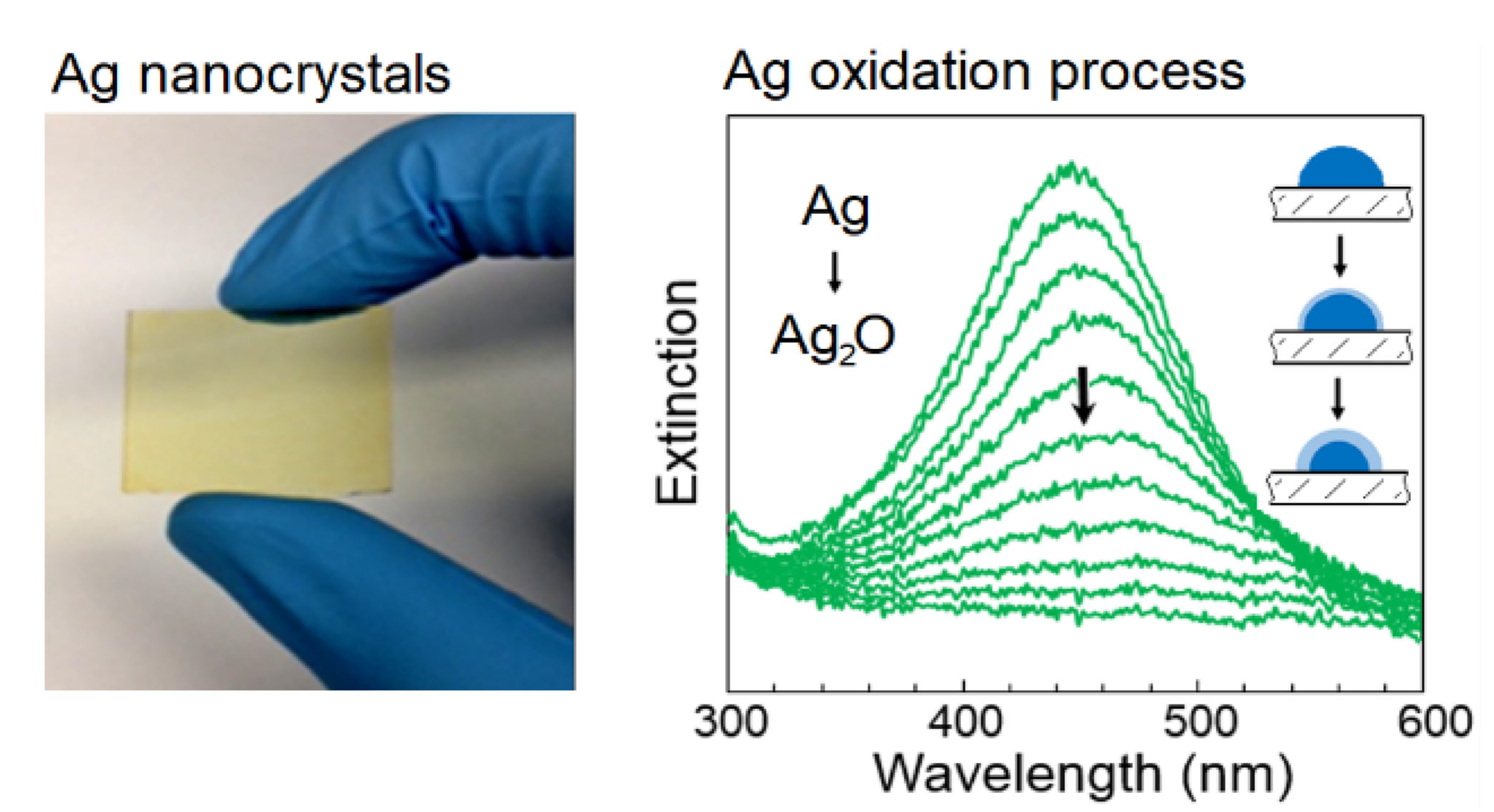
Graphical Abstract
Keywords
oxidation | silver | nanocrystal | plasmon | discrete dipole approximation
References
- 1.
Long, R.; Zhou, S.; Wiley, B.J.; Xiong, Y. Oxidative Etching for Controlled Synthesis of Metal Nanocrystals: Atomic Addition and Subtraction. Chem. Soc. Rev. 2014, 43, 6288–6310.
- 2.
Sun, X.; Kim, J.; Gilroy, K.D.; Liu, J.; König, T.A.F.; Qin, D. Gold-Based Cubic Nanoboxes with Well- Defined Openings at the Corners and Ultrathin Walls Less Than Two Nanometers Thick. ACS Nano 2016, 10, 8019–8025.
- 3.
Ling, D.; Lee, N.; Hyeon, T. Chemical synthesis and assembly of uniformly sized iron oxide nanoparticles for medical applications. Acc. Chem. Res. 2015, 48, 1276–1285.
- 4.
Prieto, P.; Nistor, V.; Nouneh, K.; Oyama, M.; Abd-Lefdil, M.; Díaz, R. XPS Study of Silver, Nickel and Bimetallic Silver–Nickel Nanoparticles Prepared by Seed-Mediated Growth. Appl. Surf. Sci. 2012, 258, 8807–8813.
- 5.
Ha, D.H.; Moreau, L.M.; Honrao, S.; Hennig, R.G.; Robinson, R.D. The Oxidation of Cobalt Nanoparticles into Kirkendall-Hollowed CoO and Co3O4: The Diffusion Mechanisms and Atomic Structural Transformations. J. Phys. Chem. C 2013, 117,14303–14312.
- 6.
Knight, M.W.; King, N.S.; Liu, L.; Everitt, H.O.; Nordlander, P.; Halas, N.J. Aluminum for Plasmonics. ACS Nano 2014, 8, 834–840.
- 7.
Jeong, S.; Woo, K.; Kim, D.; Lim, S.; Kim, J.S.; Shin, H.; Xia, Y.; Moon, J. Controlling the Thickness of the Surface Oxide Layer on Cu Nanoparticles for the Fabrication of Conductive Structures by Ink-Jet Printing. Adv. Funct. Mater. 2008, 18, 679–686.
- 8.
Yin, Y.; Li, Z.Y.; Zhong, Z.; Gates, B.; Xia, Y.; Venkateswaran, S. Synthesis and Characterization of Stable Aqueous Dispersions of Silver Nanoparticles Through the Tollens Process. J. Mater. Chem. 2002, 12, 522–527.
- 9.
Le Chatelier, H.; Über die Oxydation des Silbers. Z. Phys. Chemie 1887, 1, 516–518.
- 10.
Lewis, G.N.; Zersetzung von Silberoxyd durch Autokatalyse. Z. Phys. Chemie 1905, 52, 310–326.
- 11.
Han, Y.; Lupitskyy, R.; Chou, T.-M.; Stafford, C.M.; Du, H.; Sukhishvili, S. Effect of Oxidation on Surface-enhanced Raman Scattering Activity of Silver Nanoparticles: A Quantitative Correlation. Anal. Chem. 2011, 83, 5873–5880.
- 12.
L’vov, BV. Kinetics and Mechanism of Thermal Decomposition of Silver Oxide. Thermochimica Acta 1999, 333, 13–19.
- 13.
Xia, Y.; Xiong, Y.; Lim, B.; Skrabalak, S.E. Shape-controlled Synthesis of Metal Nanocrystals: Simple Chemistry Meets Complex Physics? Angew. Chem. Intl. Ed. 2009, 48, 60–103.
- 14.
Mayer, K.M.; Hafner, J.H. Localized Surface Plasmon Resonance Sensors. Chem. Rev. 2011, 111, 3828–3857.
- 15.
Sachan, R.; Ramos, V.; Malasi, A.; Yadavali, S.; Bartley, B.; Garcia, H.; Duscher, G.; Kalyanaraman, R. Oxidation-resistant Silver Nanostructures for Ultrastable Plasmonic Applications. Adv. Mater. 2013, 25, 2045–2050.
- 16.
Gilroy, K.D.; Puibasset, J.; Vara, M.; Xia, Y. On the Thermodynamics and Experimental Control of Twinning in Metal Nanocrystals. Angew. Chem. Int. Ed. 2017, 129, 8773–8777.
- 17.
Draine, B.T.; Flatau, P.J. Discrete-dipole Approximation for Scattering Calculations. J. Opt. Soc. Am. 1994, 11, 1491–1499.
- 18.
Graedel, T. Corrosion Mechanisms for Silver Exposed to the Atmosphere. J. Electrochem. Soc. 1992, 139, 1963–1970.
- 19.
Medasani, B.; Park, Y.H.; Vasiliev, I. Theoretical Study of the Surface Energy, Stress, and Lattice Contraction of Silver Nanoparticles. Phys. Rev. B 2007, 75, 235436.
- 20.
Gilroy, K.D.; Ruditskiy, A.; Peng, H.C.; Qin, D.; Xia, Y. Bimetallic Nanocrystals: Syntheses, Properties, and Applications. Chem. Rev. 2016, 116, 10414–10472.
- 21.
Cortie, M.B.; McDonagh, A.M. Synthesis and Optical Properties of Hybrid and Alloy Plasmonic Nanoparticles. Chem. Rev. 2011, 111, 3713–3735.
- 22.
Blaber, M.G.; Henry, A.I.; Bingham, J.M.; Schatz, G.C.; Van Duyne, R.P. LSPR Imaging of Silver Triangular Nanoprisms: Correlating Scattering with Structure Using Electrodynamics for Plasmon Lifetime Analysis. J. Phys. Chem. C 2012, 116, 393–403.
- 23.
Kreibig, U.; Vollmer, M. Optical Properties of Metal Clusters; Springer: Berlin/Heidelberg, Germany, 1995, p. 79.
- 24.
Hartland, G.V. Optical Studies of Dynamics in Noble Metal Nanostructures. Chem. Rev. 2014, 111, 3858–3887.
- 25.
Gilroy, K.D.; Xia, Y. Dimerization of Colloidal Particles Through Controlled Aggregation for Enhanced Properties and Applications, Chem. Asian J. 2016, 11, 2341–2351.
- 26.
Vodyankina, O.V.; Kurina, L.N.; Izatulina, G.A. Surface Interaction of Ethylene Glycol with Silver. React. Kinet. Catal. Lett. 1998, 64, 103–108.
- 27.
Sani, E.; Dell’Oro, A. Optical Constants of Ethylene Glycol Over an Extremely Wide Spectral Range. Opt. Mater. 2015, 37, 36–41.
- 28.
Li, C.; Shuford, K.L.; Park, Q.H.; Cai, W.; Li, Y.; Lee, E.J.; Cho, S.O.; High-Yield Synthesis of Single-Crystalline Gold Nano-octahedra. Angew. Chem. 2007, 119, 3328–3332.
- 29.
Jiu, J.; Wang, J.; Sugahara, T.; Nagao, S.; Nogi, M.; Koga, H.; Suganuma, K.; Hara, M.; Nakazawa, E.; Uchida, H. The Effect of Light and Humidity on the Stability of Silver Nanowire Transparent Electrodes. RSC Adv. 2015, 5, 27657−27664.
- 30.
Levard, C.; Hotze, E.M.; Colman, B.P.; Dale, A.L.; Truong, L.; Yang, X.Y.; Bone, A.J.; Brown, G.E.; Tanguay, R.L.; Di Giulio, R.T.; et al. Sulfidation of Silver Nanoparticles: Natural Antidote to their Toxicity. Environ. Sci. Technol. 2013, 47, 13440−13448.
- 31.
Atwater, J.E.; Holtsnider, J.T.; Wheeler, R.R., Jr. Microwave Regenerable Air Purification Device; National Technical Information Service: Springfield, VA, USA, 1996.
- 32.
Hoflund, G.B.; Hazos, Z.F. Surface Characterization Study of Ag, AgO, and Ag2O Using X-ray Photoelectron Spectroscopy and Electron Energy-loss Spectroscopy. Phys. Rev. B 2000, 62, 11126.
- 33.
Weaver, J.F.; Hoflund, G.B. Surface Characterization Study of the Thermal Decomposition of Ag2O. Chem. Mater. 1994, 6, 1693–1699.
- 34.
Weaver, J.F.; Hoflund, G.B. Surface Characterization Study of the Thermal Decomposition of AgO. J. Phys. Chem. 1994, 98, 8519–8524.
- 35.
Matikainen, A.; Nuutinen, T.; Itkonen, T.; Heinilehto, S.; Puustinen, J.; Hiltunen, J.; Lappalainen, J.; Karioja, P.; Vahimaa, P. Atmospheric Oxidation and Carbon Contamination of Silver and its Effect on Surface-enhanced Raman Spectroscopy (SERS). Sci. Rep. 2016, 6, 37192.
- 36.
Bates, C.W.; Wertheim, G.K.; Buchanan, D.N.E. Nature of the 3.8 eV Plasmon in X-ray Photoemission from Silver. Phys. Lett. A. 1979, 72, 178–180.
- 37.
Kim, M.J.; Cho, Y.S.; Park, S.H.; Huh, Y.D. Facile Synthesis and Fine Morphological Tuning of Ag2O. Cryst. Growth Des. 2012, 12, 4180−4185.
- 38.
Wang, X.; Wu, H.F.; Kuang, Q.; Huang, R.B.; Xie, Z.X.; Zheng, L.S. Shape-dependent Antibacterial Activities of Ag2O Polyhedral Particles. Langmuir 2010, 26, 2774–2778.
- 39.
Kim, M.J.; Kim, S.; Park, H.; Huh, Y.D. Morphological Evolution of Ag2O Microstructures from Cubes to Octapods and their Antibacterial Activities. Bull. Korean Chem. Soc. 2011, 32, 3793–3795.
- 40.
Holewinski, A.; Idrobo, J.C.; Linic, S. High-performance Ag–Co Alloy Catalysts for Electrochemical Oxygen Reduction. Nature Chem. 2014, 6, 828–834.
- 41.
Mayer, S.T.; Muller, R.H. Nucleation of Silver (I) Oxide Investigated by Spectroscopic Ellipsometry. J Electrochem. Soc. 2005, 152, A179–A185.
- 42.
Kadlec, O.; Dubinin, M.M. Investigation of Kinetics of Thermal Decomposition of Solids. Communication 2. Thermal Decomposition of Silver Carbonate. Izv. AN. SSSR. Otd. Khim. Nauk. 1961, 3, 390–396. (In Russian)
- 43.
Bratsch, S.G.J. Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K. Phys. Chem. Ref. Data 1989, 18, 1–21.
- 44.
Johnson, P.B.; Christy, R.W. Optical Constants of the Noble Metals. Phys. Rev. B 1972, 6, 4370–4379.
- 45.
Gao, X.Y.; Wang, S.Y.; Li, J.; Zheng, Y.X.; Zhang, R.J.; Zhou, P.; Yang, Y.M.; Chen, L.Y. Study of Structure and Optical Properties of Silver Oxide Films by Ellipsometry, XRD and XPS Methods. Thin Solid Films 2004, 455–456, 438–442.

This work is licensed under a Creative Commons Attribution 4.0 International License.


