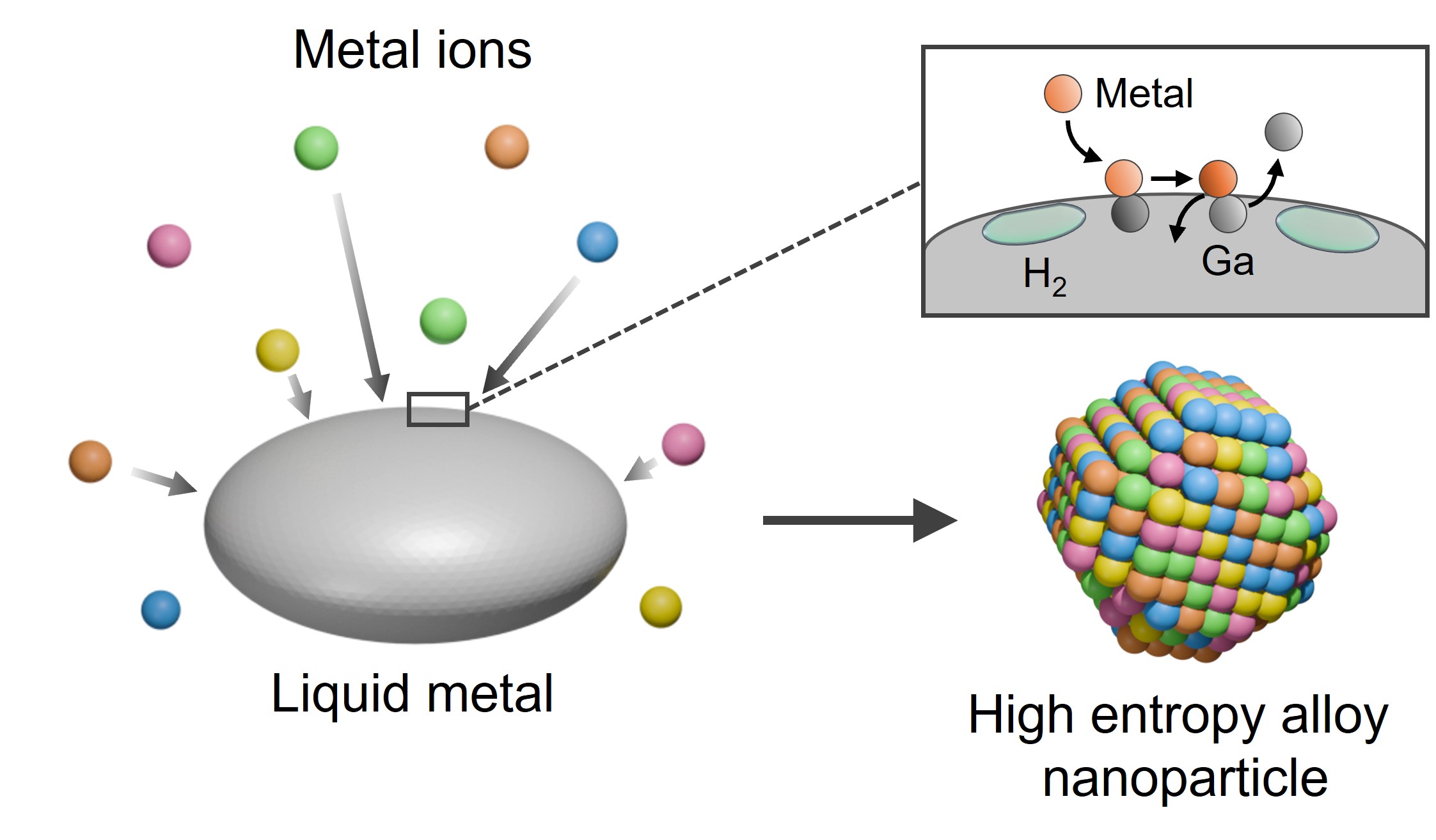
High-entropy alloy nanoparticles have emerged as a promising material platform for catalysis, sensing, energy storage, and biomedical applications, but their synthesis is often constrained by high temperature or energy inputs, as well as the intrinsic immiscibility of the constituent elements. A recent study in Nature introduces an isothermal solidification approach, in which metals are rapidly reduced and mixed in a sacrificial liquid metal, enabling the formation of nanoparticles containing up to 20 elements with controlled structure and morphology.
- Open Access
- Commentary
Kinetic Trapping of High-Entropy Alloys via Fast Reduction and Mixing in a Liquid Metal
Author Information
Received: 16 Mar 2026 | Revised: 28 Mar 2026 | Accepted: 30 Mar 2026 | Published: 31 Mar 2026
Abstract
Graphical Abstract
Keywords
high-entropy alloy | isothermal solidification | liquid metal | nanoparticle synthesis | in-situ transmission electron microscopy
References
- 1.
George, E.P.; Raabe, D.; Ritchie, R.O. High-Entropy Alloys. Nat. Rev. Mater. 2019, 4, 515–534.
- 2.
Yeh, J.W.; Chen, S.K.; Lin, S.J.; Gan, J.Y.; Chin, T.S.; Shun, T.T.; Tsau, C.H.; Chang, S.Y. Nanostructured High-Entropy Alloys with Multiple Principal Elements: Novel Alloy Design Concepts and Outcomes. Adv. Eng. Mater. 2004, 6, 299–303.
- 3.
Yao, Y.G.; Huang, Z.N.; Xie, P.F.; Lacey, S.D.; Jacob, R.J.; Xie, H.; Chen, F.J.; Nie, A.M.; Pu, T.C.; Rehwoldt, M.; et al. Carbothermal Shock Synthesis of High-Entropy-Alloy Nanoparticles. Science 2018, 359, 1489–1494.
- 4.
Glasscott, M.W.; Pendergast, A.D.; Goines, S.; Bishop, A.R.; Hoang, A.T.; Renault, C.; Dick, J.E. Electrosynthesis of High-Entropy Metallic Glass Nanoparticles for Designer, Multi-Functional Electrocatalysis. Nat. Commun. 2019, 10, 2650.
- 5.
Wang, B.; Wang, C.; Yu, X.W.; Cao, Y.; Gao, L.F.; Wu, C.P.; Yao, Y.F.; Lin, Z.Q.; Zou, Z.G. General Synthesis of High-Entropy Alloy and Ceramic Nanoparticles in Nanoseconds. Nat. Synth. 2022, 1, 138–146.
- 6.
McCormick, C.R.; Schaak, R.E. Simultaneous Multication Exchange Pathway to High-Entropy Metal Sulfide Nanoparticles. J. Am. Chem. Soc. 2021, 143, 1017–1023.
- 7.
Liu, Y.H.; Hsieh, C.J.; Hsu, L.C.; Lin, K.H.; Hsiao, Y.C.; Chi, C.C.; Lin, J.T.; Chang, C.W.; Lin, S.C.; Wu, C.Y.; et al. Toward Controllable and Predictable Synthesis of High-Entropy Alloy Nanocrystals. Sci. Adv. 2023, 9, eadf9931.
- 8.
Kang, Y.Q.; Cretu, O.; Kikkawa, J.; Kimoto, K.; Nara, H.; Nugraha, A.S.; Kawamoto, H.; Eguchi, M.; Liao, T.; Sun, Z.Q.; et al. Mesoporous Multimetallic Nanospheres with Exposed Highly Entropic Alloy Sites. Nat. Commun. 2023, 14, 2023.
- 9.
Wang, M.; Lin, Y.L. Gallium-Based Liquid Metals as Reaction Media for Nanomaterials Synthesis. Nanoscale 2024, 16, 6915–6933.
- 10.
Zhang, Q.B.; Gallant, M.C.; Chen, Y.; Song, Z.G.; Liu, Y.; Zheng, Q.; Chen, L.F.; Bustillo, K.C.; Huang, Y.; Persson, K.A.; et al. Isothermal Solidification for High-Entropy Alloy Synthesis. Nature 2025, 646, 323–330.
- 11.
Gan, T.S.; Shang, W.H.; Handschuh-Wang, S.; Zhang, Y.K.; Zhou, X.C. Liquid Metal Nanoreactor Enables Living Galvanic Replacement Reaction. Chem. Mater. 2024, 36, 3042–3053.
- 12.
Cao, G.H.; Liang, J.J.; Guo, Z.L.; Yang, K.A.; Wang, G.; Wang, H.L.; Wan, X.H.; Li, Z.Y.; Bai, Y.J.; Zhang, Y.L.; et al. Liquid Metal for High-Entropy Alloy Nanoparticles Synthesis. Nature 2023, 619, 73–77.
- 13.
Kubik, M.; Wang, S.; Camargo, P.H.C. Chemically Interpretable Machine Learning for Predicting HER Activity in Au-Based Alloys. Mater. Interfaces 2025, 2, 406–417.

This work is licensed under a Creative Commons Attribution 4.0 International License.


