Titanium-based implants are predominantly adopted in orthopedic and dental applications, yet their clinical performance is constrained by enduring challenges such as stress shielding, infection susceptibility, and inadequate osseointegration. To address these hurdles, recent advances have shifted titanium implant design from static biomimetic models toward dynamic, bioadaptive systems capable of actively modifying their architectural, mechanical, and surface properties after implantation. These systems can respond adaptively to specific pathophysiological microenvironments or be activated by external energy sources (e.g., light, ultrasound, electricity, or magnetism), thus providing on-demand biofunctionalities, such as antibacterial, pro-angiogenic, osteogenic, and immune/metabolic regulatory effects, ultimately mitigating complications (e.g., infection, inflammation) while accelerating osteoregeneration and osteointegration. In this Perspective, we propose a strategic framework to guide the design and development of next-generation titanium implants aligned with this trend. It highlights the emerging need to converge a deeper understanding of implant-biology interplays with synergistic advances from materials science (e.g., metamaterial and stimuli-responsive interface design) and cutting-edge bone biology (e.g., immunometabolic and neuro-osseous engineering). This interdisciplinary integration is aimed at enabling expedited, robust, bioadaptive regeneration regimens, particularly for challenging bone defects such as infected, osteoporotic, or diabetic conditions.
- Open Access
- Perspective
From Biomimetic to Bioadaptive: Next-Generation Titanium Implants for Functional Bone Regeneration
- Yuanyuan Liu 1,†,
- Dacheng Sang 2,3,4,†,
- Yuling Zhang 1,
- Chongbin Wei 5,
- Feifei Zhou 2,3,4,6,*,
- Zhaojun Jia 1,*
Author Information
Received: 20 Nov 2025 | Revised: 13 Jan 2026 | Accepted: 15 Jan 2026 | Published: 22 Jan 2026
Abstract
Graphical Abstract
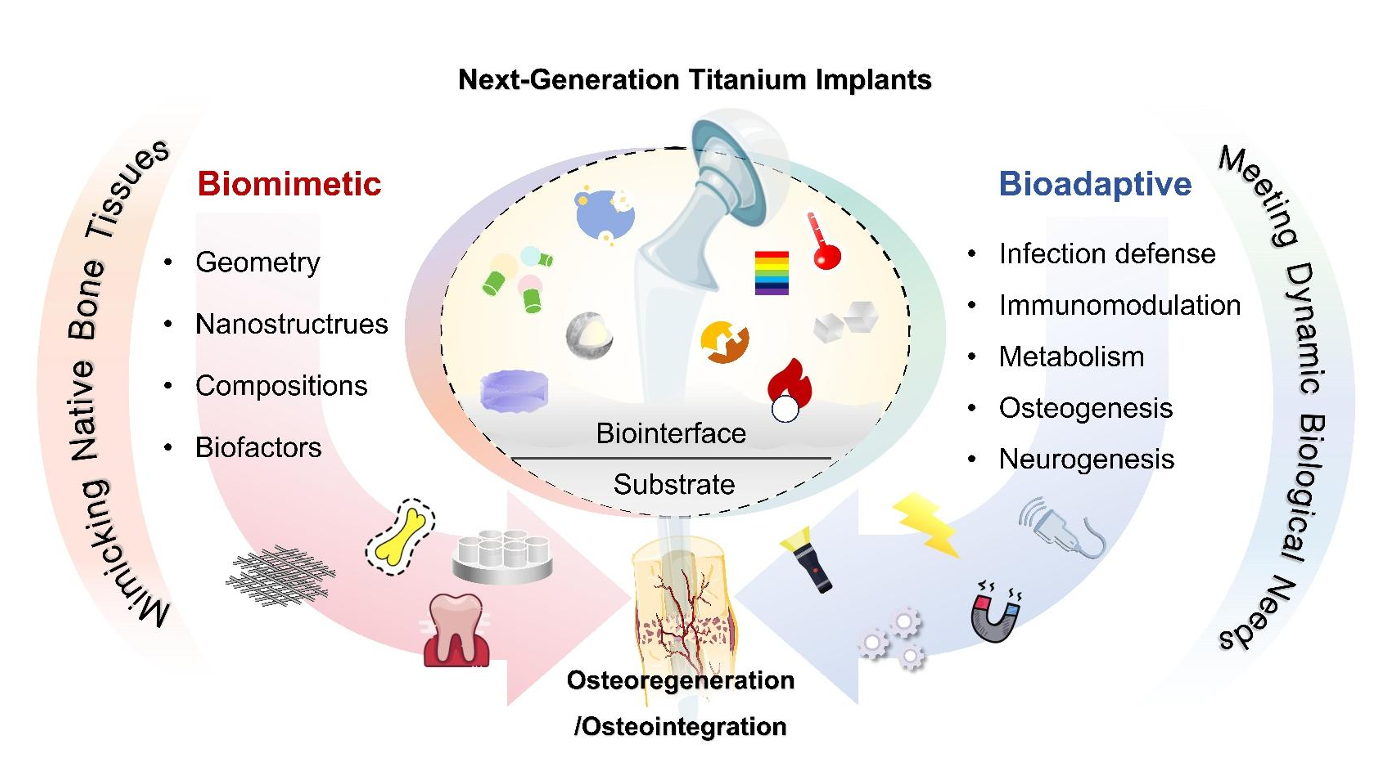
Keywords
bioadaptive implants and interfaces | stimuli-responsive designs | antibacterial functionalization | immune/metabolic engineering | osteoregeneration and osteointegration
References
- 1.
Jia, Z.; Xu, X.; Zhu, D.; et al. Design, printing, and engineering of regenerative biomaterials for personalized bone healthcare. Prog. Mater. Sci. 2023, 134, 101072.
- 2.
Abd-Elaziem, W.; Darwish, M.A.; Hamada, A.; et al. Titanium-based alloys and composites for orthopedic implants applications: A comprehensive review. Mater. Des. 2024, 241, 112850.
- 3.
Williams, D.F. Biocompatibility pathways and mechanisms for bioactive materials: The bioactivity zone. Bioact. Mater. 2022, 10, 306–322.
- 4.
Huang, D.; Li, Z.; Li, G.; et al. Biomimetic structural design in 3D-printed scaffolds for bone tissue engineering. Mater. Today Bio 2025, 32, 101664.
- 5.
Li, Y.; Xiao, Y.; Liu, C. The horizon of materiobiology: A perspective on material-guided cell behaviors and tissue engineering. Chem. Rev. 2017, 117, 4376–4421.
- 6.
Cui, B.; Wen, M.; Cai, Z.; et al. Bio-inspired self-renewing implant surfaces with sequential biofunctional adaptation for infectious diabetic tissue repair. Adv. Funct. Mater. 2025, 35, 2418092.
- 7.
Smeets, R.; Stadlinger, B.; Schwarz, F.; et al. Impact of dental implant surface modifications on osseointegration. Biomed. Res. Int. 2016, 2016, 6285620.
- 8.
Parekh, R.B.; Shetty, O.; Tabassum, R. Surface modifications for endosseous dental implants. Int. J. Oral. Implant. Clin. Res. 2012, 3, 116–121.
- 9.
Espinoza-Montero, P.J.; Montero-Jiménez, M.; Fernández, L.; et al. In vitro wearing away of orthodontic brackets and wires in different conditions: A review. Heliyon 2022, 8, e10560.
- 10.
Mohseni, E.; Zalnezhad, E.; Bushroa, A.R. Comparative investigation on the adhesion of hydroxyapatite coating on Ti-6Al-4V implant: A review paper. Int. J. Adhes. Adhes. 2014, 48, 238–257.
- 11.
Lin, C.-W.; Ju, C.-P.; Chern Lin, J.-H. A comparison of the fatigue behavior of cast Ti-7.5Mo with c.p. titanium, Ti-6Al-4V and Ti-13Nb-13Zr alloys. Biomaterials 2005, 26, 2899–2907.
- 12.
Shen, X.; Shukla, P.; Subramaniyan, A.K.; et al. Residual stresses induced by laser shock peening in orthopaedic Ti-6Al-7Nb alloy. Opt. Laser Technol. 2020, 131, 106446.
- 13.
Vancleef, S.; Wesseling, M.; Duflou, J.R.; et al. Thin patient-specific clavicle fracture fixation plates can mechanically outperform commercial plates: An in silico approach. J. Orthop. Res. 2022, 40, 1695–1706.
- 14.
Perren, S.M.; Regazzoni, P.; Fernandez, A.A. How to choose between the implant materials steel and titanium in orthopedic trauma surgery: Part 2—Biological aspects. Acta Chir. Orthop. Traumatol. Cech. 2017, 84, 85–90.
- 15.
Disegi, J.A. Titanium alloys for fracture fixation implants. Injury 2000, 31, D14-D17.
- 16.
Massaad, E.; Fatima, N.; Kiapour, A.; et al. Polyetheretherketone versus titanium cages for posterior lumbar interbody fusion: Meta-analysis and review of the literature. Neurospine 2020, 17, 125–135.
- 17.
Yamanaka, K.; Mori, M.; Yamazaki, K.; et al. Analysis of the fracture mechanism of Ti-6Al-4V alloy rods that failed clinically after spinal instrumentation surgery. Spine 2015, 40, E767–E773.
- 18.
Wang, Y. Bioadaptability: An innovative concept for biomaterials. J. Mater. Sci. Technol. 2016, 32, 801–809.
- 19.
Xu, X.; Jia, Z.; Zheng, Y.; Wang, Y. Bioadaptability of biomaterials: Aiming at precision medicine. Matter 2021, 4, 2648–2650.
- 20.
Ma, J.; Zhou, Z.; Gao, M.; et al. Biosynthesis of bioadaptive materials: A review on developing materials available for tissue adaptation. J. Mater. Sci. Technol. 2016, 32, 810–814.
- 21.
Zheng, W.; Jin, Z.; Chen, X.; et al. Self-adaptive biomaterials for tissue repair: From design to application. Prog. Mater. Sci. 2026, 157, 101593.
- 22.
Zhou, J.; Yuan, F.; Jia, Z. Neutrophil polarization and immune regulation: Toward immune-bioadaptive dental implants. Regen. Med. Dent. 2025, 2, 19.
- 23.
Tian, Y.; Jiang, W.; Deng, L.; et al. A fractal-like hierarchical bionic scaffold for osseointegration. Adv. Funct. Mater. 2025, 35, 2415880.
- 24.
Bagheri, M.A.; Aubin, C.E.; Nault, M.L.; et al. Mechanical characterization of an origami-inspired super deformable metamaterial with high tunability for tissue engineering. Mater. Des. 2025, 251, 113701.
- 25.
Min, J.; Choi, K.Y.; Dreaden, E.C.; et al. Designer dual therapy nanolayered implant coatings eradicate biofilms and accelerate bone tissue repair. ACS Nano 2016, 10, 4441–4450.
- 26.
Yao, Q.; Ye, Z.; Sun, L.; et al. Bacterial infection microenvironment-responsive enzymatically degradable multilayer films for multifunctional antibacterial properties. J. Mater. Chem. B 2017, 5, 8532–8541.
- 27.
Wang, X.; Song, L.; Zhao, J.; et al. Bacterial adaptability of enzyme and pH dual-responsive surface for infection resistance. J. Mater. Chem. B 2018, 6, 7710–7718.
- 28.
Li, S.; Man, Z.; Zuo, K.; et al. Advancement in smart bone implants: The latest multifunctional strategies and synergistic mechanisms for tissue repair and regeneration. Bioact. Mater. 2025, 51, 333–382.
- 29.
Luo, L.; Zheng, W.; Li, J.; et al. 3D-printed titanium trabecular scaffolds with sustained release of hypoxia-induced exosomes for dual-mimetic bone regeneration. Adv. Sci. 2025, 12, e2500599.
- 30.
Han, L.; Che, S. An overview of materials with triply periodic minimal surfaces and related geometry: From biological structures to self-assembled systems. Adv. Mater. 2018, 30, 1705708.
- 31.
Liu, L.; Chen, H.; Zhang, A.; et al. Biomimetic gradient microporous scaffold with a triply periodic minimal surface enhances osseointegration by modulating macrophage polarization. Biomater. Res. 2025, 29, 0266.
- 32.
Karali, B.K.; Suthar, S.; Banerji, S.; et al. Asymmetric mechanical behavior and pre-osteoblast differentiation in 3D printed Ti6Al4V-based triply periodic minimal surface bone-analogues: The role of pore topology. Acta Biomater. 2025, 207, 633–652.
- 33.
Charbonnier, B.; Manassero, M.; Bourguignon, M.; et al. Custom-made macroporous bioceramic implants based on triply-periodic minimal surfaces for bone defects in load-bearing sites. Acta Biomater. 2020, 109, 254–266.
- 34.
Pugliese, R.; Graziosi, S. Biomimetic scaffolds using triply periodic minimal surface-based porous structures for biomedical applications. SLAS Technol. 2023, 28, 165–182.
- 35.
Qin, Y.; Jing, Z.; Zou, D.; et al. A metamaterial scaffold beyond modulus limits: Enhanced osteogenesis and angiogenesis of critical bone defects. Nat. Commun. 2025, 16, 2180.
- 36.
Long, S.-Y.; Fu, Y.-J.; Zhang, Z.-M.; et al. Architecture mechanics mediated osteogenic progression in bone regeneration of artificial scaffolds. Sci. Adv. 2025, 11, eadv8804.
- 37.
Souza, J.C.M.; Sordi, M.B.; Kanazawa, M.; et al. Nano-scale modification of titanium implant surfaces to enhance osseointegration. Acta Biomater. 2019, 94, 112–131.
- 38.
Wang, R.; Li, J.; Bi, Q.; et al. Crystallographic plane-induced selective mineralization of nanohydroxyapatite on fibrous-grained titanium promotes osteointegration and biocorrosion resistance. Biomaterials 2025, 313, 122800.
- 39.
Che, Z.; Sheng, X.; Sun, Q.; et al. Deferoxamine functionalized alginate-based collagen composite material enhances the integration of metal implant and bone interface. Carbohydr. Polym. 2025, 349, 122944.
- 40.
Jing, Z.; Yuan, W.; Wang, J.; et al. Simvastatin/hydrogel-loaded 3D-printed titanium alloy scaffolds suppress osteosarcoma via TF/NOX2-associated ferroptosis while repairing bone defects. Bioact. Mater. 2024, 33, 223–241.
- 41.
Wang, C.; Ju, J.; Fu, C.; et al. Metabolically engineered extracellular vesicles released from a composite hydrogel delivery system regulate the microenvironment for periprosthetic osteolysis treatment. J. Extracell. Vesicles 2025, 14, e70098.
- 42.
Zheng, W.; Tian, S.; Huo, J.; et al. Mechanically adaptive Mg-Ti composites guided by single-cell insights accelerate load-bearing bone regeneration via dual modulation of osteogenesis and osteoclastogenesis. Bioact. Mater. 2026, 57, 54–72.
- 43.
Jiang, P.; Zhang, Y.; Hu, R.; et al. Advanced surface engineering of titanium materials for biomedical applications: From static modification to dynamic responsive regulation. Bioact. Mater. 2023, 27, 15–57.
- 44.
Batool, S.A.; Ghazanfar, E.; Ahmed, H.; et al. Improved physicochemical properties of structurally modified titanium coated with zein-mesoporous bioactive glass nanoparticles-Commiphora wightii for orthopaedic applications. Int. J. Biol. Macromol. 2025, 305, 140870.
- 45.
Liu, Y.; Wen, M.; Hao, J.; et al. Engineering metallic implants with self-catalytic degradable bionanozyme coating and ion-releasing biointerface for bioadaptive anti-infective and osteogenic functionality. ACS Appl. Mater. Interface 2026, 18, 3578–3598.
- 46.
Zhang, J.; Ren, K.; Qiu, J.; et al. Nanotexture and crystal phase regulation for synergistic enhancement in re-endothelialization on medical pure titanium surface. Mater. Today Bio 2024, 25, 100968.
- 47.
Zhang, Q.; Pan, R.-L.; Wang, H.; et al. Nanoporous titanium implant surface accelerates osteogenesis via the piezo1/acetyl-CoA/β-catenin pathway. Nano Lett. 2024, 24, 8257–8267.
- 48.
Fialho, L.; Alves, C.F.A.; Carvalho, S. A decade of progress on MAO-treated tantalum surfaces: Advances and contributions for biomedical applications. Nanomaterials 2022, 12, 2319.
- 49.
Zhang, T.; Zhou, W.; Yang, W.; et al. Vancomycin-encapsulated hydrogel loaded microarc-oxidized 3D-printed porous Ti6Al4V implant for infected bone defects: Reconstruction, anti-infection, and osseointegration. Bioact. Mater. 2024, 42, 18–31.
- 50.
Feng, F.; Wu, Y.; Xin, H.; et al. Surface characteristics and biocompatibility of ultrafine-grain Ti after sandblasting and acid etching for dental implants. ACS Biomater. Sci. Eng. 2019, 5, 5107–5115.
- 51.
Yang, W.E.; Huang, H.H. TiO2 nanonetwork on rough Ti enhanced osteogenesis in vitro and in vivo. J. Dent. Res. 2021, 100, 1186–1193.
- 52.
Gao, H.; Yu, M.-J.; Chen, X.; et al. Enhanced mechanical, antibacterial, and osteogenic performance of zinc-doped micro-nano porous layer on Ti6Al4V: Realized by ion exchange and induction heating. Surf. Coat. Technol. 2025, 497, 131763.
- 53.
Chen, L.; Guo, Z.; Duan, G.; et al. The multifaceted biomimetic titanium implant promotes bone integration with sequential antibacterial and immune modulation properties. Bioact. Mater. 2025, 51, 494–511.
- 54.
Chen, Y.; Zhou, L.; Guan, M.; et al. Multifunctionally disordered TiO2 nanoneedles prevent periprosthetic infection and enhance osteointegration by killing bacteria and modulating the osteoimmune microenvironment. Theranostics 2024, 14, 6016–6035.
- 55.
Sun, M.; Xu, C.; Wu, R.; et al. Surface-engineered orthopedic implant to break the antimicrobial-osteogenic paradox at biointerfaces in the “race for the surface”. Adv. Mater. 2025, 37, 2506113.
- 56.
Lei, Y.; Hai, J.; Zhang, X.; et al. Preparation of porous TC4 bone scaffold VEGF-Si/HA-TiO2 nanotube composite coating: Crystal structure improvement and bio sustained release promoting angiogenesis and osteogenic regeneration. J. Drug Deliv. Sci. Technol. 2025, 114, 107540.
- 57.
Xie, H.; Zhang, L.; Chen, J.; et al. A dual-catalysis mode for ROS regulation to accelerate biointegration of implants in infected diabetic wound. Adv. Funct. Mater. 2025, 35, 2423015.
- 58.
Bao, S.; Yu, D.; Tang, Z.; et al. Conformationally regulated “nanozyme-like” cerium oxide with multiple free radical scavenging activities for osteoimmunology modulation and vascularized osseointegration. Bioact. Mater. 2024, 34, 64–79.
- 59.
Li, J.; Wen, J.; Li, B.; et al. Valence state manipulation of cerium oxide nanoparticles on a titanium surface for modulating cell fate and bone formation. Adv. Sci. 2018, 5, 1700678.
- 60.
Han, J.; Ma, Q.; An, Y.; et al. The current status of stimuli-responsive nanotechnologies on orthopedic titanium implant surfaces. J. Nanobiotechnol. 2023, 21, 277.
- 61.
Jayasree, A.; Ivanovski, S.; Gulati, K. On or Off: Triggered therapies from anodized nano-engineered titanium implants. J. Control. Release 2021, 333, 521–535.
- 62.
Yu, Z.; Wang, Z.; Chen, Y.; et al. Programmed surface platform orchestrates anti-bacterial ability and time-sequential bone healing for implant-associated infection. Biomaterials 2025, 313, 122772.
- 63.
Böhner, D.A.; Mörl, K.; Beck-Sickinger, A.G. Bio-inspired biomaterial coating for enzyme responsive release of antimicrobial peptides. Mater. Today Bio 2025, 33, 101958.
- 64.
Wang, Q.; Gao, Y.; Chen, Y.; et al. Synergistic enhancement of antibacterial and osteo-immunomodulatory activities of titanium implants via dual-responsive multifunctional surfaces. Adv. Healthc. Mater. 2025, 14, 2404260.
- 65.
Liu, J.; Han, X.; Zhang, T.; et al. Reactive oxygen species (ROS) scavenging biomaterials for anti-inflammatory diseases: From mechanism to therapy. J. Hematol. Oncol. 2023, 16, 116.
- 66.
Yang, Y.; He, X.; Zhao, Z.; Yi, J. Macrophage-centric biomaterials for bone regeneration in diabetes mellitus: Contemporary advancements, challenges, and future trajectories. Cureus 2024, 16, e66621.
- 67.
Zhu, Y.; Liang, H.; Liu, X.; et al. Regulation of macrophage polarization through surface topography design to facilitate implant-to-bone osteointegration. Sci. Adv. 2021, 7, eabf6654.
- 68.
Xu, H.; Tan, X.; Gerhard, E.; et al. Metabotissugenic citrate biomaterials orchestrate bone regeneration via citrate-mediated signaling pathways. Sci. Adv. 2025, 11, eady2862.
- 69.
Yuan, Y.; Xu, Y.; Mao, Y.; et al. Three birds, one stone: An osteo-microenvironment stage-regulative scaffold for bone defect repair through modulating early osteo-immunomodulation, middle neovascularization, and later osteogenesis. Adv. Sci. 2024, 11, e2306428.
- 70.
Hu, K.; Shang, Z.; Yang, X.; et al. Macrophage polarization and the regulation of bone immunity in bone homeostasis. J. Inflamm. Res. 2023, 16, 3563–3580.
- 71.
Lee, R.S.B.; Hamlet, S.M.; Moon, H.-J.; et al. Re-establishment of macrophage homeostasis by titanium surface modification in type II diabetes promotes osseous healing. Biomaterials 2021, 267, 120464.
- 72.
Shi, X.; Xu, C.; Chen, Z.; et al. Biomaterial-mediated macrophage polarization remodeling and sequential regulation: A potential strategy in bone infections treatment. Bone Res. 2025, 13, 96.
- 73.
Wang, Z.; Niu, Y.; Tian, X.; et al. Switching on and off macrophages by a “bridge-burning” coating improves bone-implant integration under osteoporosis. Adv. Funct. Mater. 2021, 31, 2007408.
- 74.
Wang, Y.-N.; Yu, L.; Wang, T.; et al. Apolipoprotein E facilitates titanium implant osseointegration by regulating osteogenesis-lipogenesis balance. Int. J. Biol. Macromol. 2023, 236, 123998.
- 75.
Collins, M.T.; Marcucci, G.; Anders, H.-J.; et al. Skeletal and extraskeletal disorders of biomineralization. Nat. Rev. Endocrinol. 2022, 18, 473–489.
- 76.
Guo, S.; Zhang, M.; Zhang, K.; et al. ROS-responsive titanium implant coatings enhance osteoporotic osseointegration via microenvironment remodeling and mitochondrial repair. Adv. Funct. Mater. 2025, e24289. https://doi.org/10.1002/adfm.202524289
- 77.
Chen, M.; Wang, D.; Li, M.; et al. Nanocatalytic biofunctional MOF coating on titanium implants promotes osteoporotic bone regeneration through cooperative pro-osteoblastogenesis MSC reprogramming. ACS Nano 2022, 16, 15397–15412.
- 78.
Ding, P.; Gao, C.; Gao, Y.; et al. Osteocytes regulate senescence of bone and bone marrow. eLife 2022, 11, e814802022, 11, e81480.
- 79.
Ahmed, W.; Zhai, Z.; Gao, C. Adaptive antibacterial biomaterial surfaces and their applications. Mater. Today Bio 2019, 2, 100017.
- 80.
Liu, Z.; Yi, Y.; Wang, S.; et al. Bio-inspired self-adaptive nanocomposite array: From non-antibiotic antibacterial actions to cell proliferation. ACS Nano 2022, 16, 16549–16562.
- 81.
Wang, L.; Zhang, C.; Zhao, J.; et al. Biomimetic targeting nanoadjuvants for sonodynamic and chronological multi-immunotherapy against holistic biofilm-related infections. Adv. Mater. 2024, 36, 2308110.
- 82.
Yang, D.; Ding, M.; Song, Y.; et al. Nanotherapeutics with immunoregulatory functions for the treatment of bacterial infection. Biomater. Res. 2025, 27, 73.
- 83.
Yue, X.; Sun, X.; Li, Z.; et al. Biomimetic piezoelectric periosteum-bone integrated implant promotes bone defect repair by remodeling osteogenic microenvironment. Adv. Funct. Mater. 2025, 35, 2423492.
- 84.
Wang, Z.; Tang, Y.; Wang, P.; et al. Dynamical integration of antimicrobial, anti-inflammatory, and pro-osteogenic activities on polyetheretherketone via a porous N-halamine polymeric coating. Adv. Funct. Mater. 2023, 33, 2307286.
- 85.
Wu, Y.; Cai, Z.; Zhang, Y.; et al. Nanocatalytic magnesium osteoimplants with biodegradable self-adaptive interfaces for therapeutic repair of infected bone defects. Bioact. Mater. 2026, 58, 632–649.
- 86.
Cui, C.; Zhao, Y.; Bai, Z.; et al. The effect of antibacterial-osteogenic surface modification on the osseointegration of titanium implants: A static and dynamic strategy. ACS Biomater. Sci. Eng. 2024, 10, 4093–4113.
- 87.
Beyrami, H.; Golshan, M.; Zardehi-Tabriz, A.; et al. Smart coatings: Fundamentals, preparation approaches, and applications. Adv. Mater. Technol. 2025, 10, e00574.
- 88.
Tripathi, S.; Raheem, A.; Dash, M.; et al. Surface engineering of orthopedic implants for better clinical adoption. J. Mater. Chem. B 2024, 12, 11302–11335.
- 89.
Martínez Campos, E.; Santos-Coquillat, A.; Mingo, B.; et al. Albumin loaded PEO coatings on Ti—potential as drug eluting systems. Surf. Coat. Technol. 2015, 283, 44–51.
- 90.
Fu, J.; Wang, C.; Liu, X.; et al. Smart responsive materials for antibacterial therapy: Progress, opportunities, and challenges. Prog. Mater. Sci. 2026, 155, 101532.
- 91.
Filipov, E.; Delibaltov, D.; Stefanov, R.; et al. Surface functionalization of 3D printed poly-ε-caprolactone by ultrashort laser mirostructuring and ZnO nanolayer deposition. J. Phys. Conf. Ser. 2024, 2710, 012018.
- 92.
Thakur, S.; Chauhan, S.R. Development of a critical edge-based adaptive toolpath strategy to improve geometrical accuracy of incrementally formed titanium implants. J. Manuf. Process. 2024, 110, 114–125.
- 93.
Thejane, K.; du Preez, W.; Booysen, G. Implementing digital twinning in an additive manufacturing process chain. MATEC Web Conf. 2023, 388, 10001.
- 94.
Holzapfel, B.M.; Reichert, J.C.; Schantz, J.-T.; et al. How smart do biomaterials need to be? A translational science and clinical point of view. Adv. Drug Deliv. Rev. 2013, 65, 581–603.
- 95.
Zhang, K.; Liang, W.; Chen, X.-B.; et al. Smart materials strategy for vascular challenges targeting in-stent restenosis: A critical review. Regener. Biomater. 2025, 12, rbaf020.
- 96.
Prakash, A.; Malviya, R.; Sridhar, S.B.; et al. 4D printing in dynamic and adaptive bone implants: Progress in bone tissue engineering. Bioprinting 2024, 44, e00373.
- 97.
Guo, Y.; Mi, J.; Ye, C.; et al. A practical guide to promote informatics-driven efficient biotopographic material development. Bioact. Mater. 2022, 8, 515–528.
- 98.
Malachi, I.O.; Olawumi, A.O.; Oladapo, B.I. AI-driven optimization of spinal implant design using parametric modelling. Colloids Surf. B 2025, 253, 114753.
- 99.
Zaheer, M.U.; Razzaq, M.H.; Aycan, M.F.; et al. AI-assisted design and evaluation of SLM-Ti64 implants for enhanced bone regeneration. Adv. Healthc. Mater. 2025, 14, e03154.
- 100.
Alanazi, B.N.; Ahmed, H.A.; Alharbi, N.S.; et al. Exploring 4D printing of smart materials for regenerative medicine applications. RSC Adv. 2025, 15, 32155–32171.
- 101.
Wu, Y.; Guo, J.; Chen, Z.; et al. Deciphering the skeletal interoceptive circuitry to control bone homeostasis. BMEMat 2025, 3, e12138.
- 102.
Fan, J.; Ding, Z.; Cai, Y.; et al. Revolutionizing bone regeneration: Vascularized bone tissue engineering with advanced 3D printing technology. Aggregate 2025, 6, e731.
- 103.
Zhao, X.; Yao, M.; Wang, Y.; et al. Neuroregulation during bone formation and regeneration: Mechanisms and strategies. ACS Appl. Mater. Interfaces 2025, 17, 7223–7250.
- 104.
Sun, X.; Zhong, R.; Wu, C.; et al. 3D printed titanium scaffolds with bi-directional gradient QK-functionalized surface. Adv. Mater. 2025, 37, 2406421.

This work is licensed under a Creative Commons Attribution 4.0 International License.


