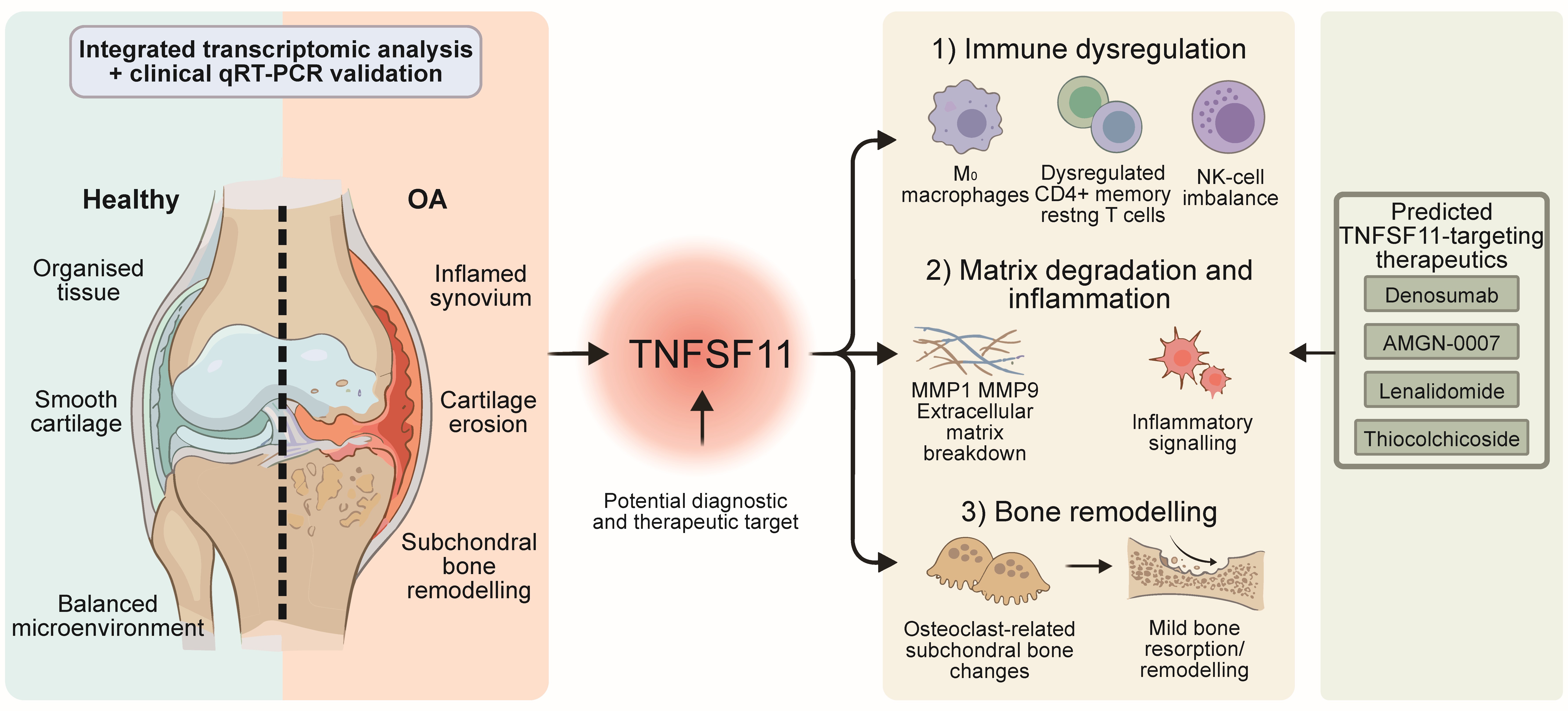
Background: Osteoarthritis (OA) is a chronic joint disease marked by inflammation of the synovium, subchondral bone sclerosis, and cartilage deterioration. Although its underlying molecular mechanisms remain incompletely elucidated, tumour necrosis factor superfamily member 11 (TNFSF11) has been closely linked to the pathophysiology of OA. Elucidating the specific role of TNFSF11 may facilitate the development of novel diagnostic strategies and therapeutic interventions for OA. Methods: This study examined TNFSF11’s function in OA using integrated bioinformatics. Transcriptome data of synovial tissues from OA patients and control (CON) subjects were retrieved from the Gene Expression Omnibus (GEO) database. The diagnostic value of TNFSF11 and differentially expressed genes (DEGs) was evaluated. TNFSF11-related genes were identified using weighted gene co-expression network analysis (WGCNA) and protein-protein interaction (PPI) analysis, results of which were confirmed by molecular docking. Immune cell infiltration and biological pathways were investigated using CIBERSORTx and functional enrichment analysis. TNFSF11-targeting medications were predicted using DrugBank. The causal associations between TNFSF11 and its target genes and OA were evaluated using two-sample Mendelian randomisation (MR) methodology. The expression of genes in clinical samples was verified using quantitative real-time polymerase chain reaction (qRT-PCR). Results: TNFSF11 was significantly upregulated in OA synovial tissues and demonstrated good diagnostic potential. A total of 372 TNFSF11-related genes were identified, among which 18 directly interacted with TNFSF11—findings that were validated by molecular docking. Functional enrichment was associated with immune response, cell adhesion, and matrix degradation pathways. TNFSF11 expression correlated with immune infiltration, particularly CD4 memory resting T cells, activated NK cells, and M0 macrophages. Four drugs (AMGN-0007, denosumab, lenalidomide, and thiocolchicoside) were predicted to target TNFSF11 with strong binding affinities. MR analysis revealed that CTSK, SPP1, and TBXAS1 were protective factors (all p < 0.05), while FCGR1A, MMP1, MMP9, TNFRSF11A, and TNFSF11 were risk factors for OA (all p < 0.05). qRT-PCR validation confirmed significant upregulation of TNFSF11, TNFRSF11A, FCGR1A, MMP1, and MMP9 in OA samples (all p < 0.05), which was consistent with bioinformatics findings. Conclusions: TNFSF11 plays a critical role in OA pathogenesis by regulating target genes, immune cell infiltration, and inflammatory pathways. These findings offer novel insights into the function of TNFSF11 in OA and identify potential therapeutic targets for OA management.
- Open Access
- Article
TNFSF11 as a Key Regulator in Osteoarthritis Pathogenesis: Integrated Bioinformatics Analysis and Potential Therapeutic Targets
- Fumin Yuan 1,2,†,
- Xiwei Fan 1,2,3,4,5,†,
- Indira Prasadam 3,4,
- Antonia Rujia Sun 3,4,
- Xiaoxin Wu 1,2,5,
- Yanping Wang 6,*,
- Xinzhan Mao 1,2,5,*
Author Information
Received: 26 Jan 2026 | Revised: 02 Mar 2026 | Accepted: 06 Mar 2026 | Published: 19 Mar 2026
Abstract
Graphical Abstract
Keywords
osteoarthritis | TNFSF11 | WGCNA | PPI | immune infiltration | bioinformatics analysis | drug prediction
References
- 1.
Kulkarni, P.; Martson, A.; Vidya, R.; et al. Pathophysiological Landscape of Osteoarthritis. Adv. Clin. Chem. 2021, 100, 37–90. https://doi.org/10.1016/bs.acc.2020.04.002.
- 2.
Bannuru, R.R.; Osani, M.C.; Vaysbrot, E.E.; et al. OARSI Guidelines for the Non-Surgical Management of Knee, Hip, and Polyarticular Osteoarthritis. Osteoarthr. Cartil. 2019, 27, 1578–1589. https://doi.org/10.1016/j.joca.2019.06.011.
- 3.
Fisch, K.M.; Gamini, R.; Alvarez-Garcia, O.; et al. Identification of Transcription Factors Responsible for Dysregulated Networks in Human Osteoarthritis Cartilage by Global Gene Expression Analysis. Osteoarthr. Cartil. 2018, 26, 1531–1538. https://doi.org/10.1016/j.joca.2018.07.012.
- 4.
Mead, T.J.; Apte, S.S. ADAMTS Proteins in Human Disorders. Matrix Biol. 2018, 71–72, 225–239. https://doi.org/10.1016/j.matbio.2018.06.002.
- 5.
Milaras, C.; Lepetsos, P.; Dafou, D.; et al. Association of Matrix Metalloproteinase (MMP) Gene Polymorphisms with Knee Osteoarthritis: A Review of the Literature. Cureus 2021, 13, e18607. https://doi.org/10.7759/cureus.18607.
- 6.
Goldring, M.B.; Otero, M.; Tsuchimochi, K.; et al. Defining the Roles of Inflammatory and Anabolic Cytokines in Cartilage Metabolism. Ann. Rheum. Dis. 2008, 67, iii75–iii82. https://doi.org/10.1136/ard.2008.098764.
- 7.
Li, S.; Shi, Y.; Zhang, S.; et al. Corynoline Alleviates Osteoarthritis Development via the Nrf2/NF-κB Pathway. Oxid. Med. Cell. Longev. 2022, 2022, 2188145. https://doi.org/10.1155/2022/2188145.
- 8.
Fazio, A.; Di Martino, A.; Brunello, M.; et al. The Involvement of Signaling Pathways in the Pathogenesis of Osteoarthritis: An Update. J. Orthop. Transl. 2024, 47, 116–124. https://doi.org/10.1016/j.jot.2024.06.002.
- 9.
Sassi, N.; Laadhar, L.; Driss, M.; et al. The Role of the Notch Pathway in Healthy and Osteoarthritic Articular Cartilage: From Experimental Models to Ex Vivo Studies. Arthritis Res. Ther. 2011, 13, 208. https://doi.org/10.1186/ar3255.
- 10.
Karlsson, C.; Brantsing, C.; Egell, S.; et al. Notch1, Jagged1, and HES5 Are Abundantly Expressed in Osteoarthritis. Cells Tissues Organs 2008, 188, 287–298. https://doi.org/10.1159/000121610.
- 11.
Hu, X.; Li, J.; Fu, M.; et al. The JAK/STAT Signaling Pathway: From Bench to Clinic. Signal Transduct. Target. Ther. 2021, 6, 402. https://doi.org/10.1038/s41392-021-00791-1.
- 12.
Zhou, Q.; Ren, Q.; Jiao, L.; et al. The Potential Roles of JAK/STAT Signaling in the Progression of Osteoarthritis. Front. Endocrinol. 2022, 13, 1069057. https://doi.org/10.3389/fendo.2022.1069057.
- 13.
Lim, H.; Kim, H.P. Matrix Metalloproteinase-13 Expression in IL-1β-Treated Chondrocytes by Activation of the p38 MAPK/c-Fos/AP-1 and JAK/STAT Pathways. Arch. Pharmacal Res. 2011, 34, 109–117. https://doi.org/10.1007/s12272-011-0113-4.
- 14.
Meszaros, E.C.; Malemud, C.J. Phosphorylation of STAT Proteins by Recombinant Human IL-6 in Immortalized Human Chondrocyte Cell Lines, T/C28a2 and C28/I2. J. Inflamm. Res. 2017, 10, 143–150. https://doi.org/10.2147/jir.S93797.
- 15.
Xuan, F.; Yano, F.; Mori, D.; et al. Wnt/β-Catenin Signaling Contributes to Articular Cartilage Homeostasis through Lubricin Induction in the Superficial Zone. Arthritis Res. Ther. 2019, 21, 247. https://doi.org/10.1186/s13075-019-2041-5.
- 16.
Zhou, Y.; Wang, T.; Hamilton, J.L.; et al. Wnt/β-Catenin Signaling in Osteoarthritis and in Other Forms of Arthritis. Curr. Rheumatol. Rep. 2017, 19, 53. https://doi.org/10.1007/s11926-017-0679-z.
- 17.
Li, K.; Zhang, Y.; Zhang, Y.; et al. Tyrosine Kinase Fyn Promotes Osteoarthritis by Activating the β-Catenin Pathway. Ann. Rheum. Dis. 2018, 77, 935–943. https://doi.org/10.1136/annrheumdis-2017-212658.
- 18.
Cheng, P.; Wirka, R.C.; Kim, J.B.; et al. Smad3 Regulates Smooth Muscle Cell Fate and Mediates Adverse Remodeling and Calcification of the Atherosclerotic Plaque. Nat. Cardiovasc. Res. 2022, 1, 322–333. https://doi.org/10.1038/s44161-022-00042-8.
- 19.
Lafyatis, R. Transforming Growth Factor β—At the Centre of Systemic Sclerosis. Nat. Rev. Rheumatol. 2014, 10, 706–719. https://doi.org/10.1038/nrrheum.2014.137.
- 20.
Pickup, M.; Novitskiy, S.; Moses, H.L. The Roles of TGFβ in the Tumour Microenvironment. Nat. Rev. Cancer 2013, 13, 788–799. https://doi.org/10.1038/nrc3603.
- 21.
Liu-Bryan, R.; Terkeltaub, R. Emerging Regulators of the Inflammatory Process in Osteoarthritis. Nat. Rev. Rheumatol. 2015, 11, 35–44. https://doi.org/10.1038/nrrheum.2014.162.
- 22.
Chen, Z.; Zhong, H.; Wei, J.; et al. Inhibition of Nrf2/HO-1 Signaling Leads to Increased Activation of the NLRP3 Inflammasome in Osteoarthritis. Arthritis Res. Ther. 2019, 21, 300. https://doi.org/10.1186/s13075-019-2085-6.
- 23.
Tudorachi, N.B.; Totu, E.E.; Fifere, A.; et al. The Implication of Reactive Oxygen Species and Antioxidants in Knee Osteoarthritis. Antioxidants 2021, 10, 985. https://doi.org/10.3390/antiox10060985.
- 24.
Lepetsos, P.; Papavassiliou, K.A.; Papavassiliou, A.G. Redox and NF-κB Signaling in Osteoarthritis. Free Radical Biol. Med. 2019, 132, 90–100. https://doi.org/10.1016/j.freeradbiomed.2018.09.025.
- 25.
Bolduc, J.A.; Collins, J.A.; Loeser, R.F. Reactive Oxygen Species, Aging and Articular Cartilage Homeostasis. Free Radic. Biol. Med. 2019, 132, 73–82. https://doi.org/10.1016/j.freeradbiomed.2018.08.038.
- 26.
Li, J.; Jiang, M.; Yu, Z.; et al. Artemisinin Relieves Osteoarthritis by Activating Mitochondrial Autophagy through Reducing TNFSF11 Expression and Inhibiting PI3K/AKT/mTOR Signaling in Cartilage. Cell. Mol. Biol. Lett. 2022, 27, 62. https://doi.org/10.1186/s11658-022-00365-1.
- 27.
Sun, Z.; Liu, Q.; Lv, Z.; et al. Targeting Macrophagic SHP2 for Ameliorating Osteoarthritis via TLR Signaling. Acta Pharm. Sin. B 2022, 12, 3073–3084. https://doi.org/10.1016/j.apsb.2022.02.010.
- 28.
Kloppenburg, M.; Berenbaum, F. Osteoarthritis Year in Review 2019: Epidemiology and Therapy. Osteoarthritis Cartilage 2020, 28, 242–248. https://doi.org/10.1016/j.joca.2020.01.002.
- 29.
Zeng, X.Z.; He, L.G.; Wang, S.; et al. Aconine Inhibits RANKL-Induced Osteoclast Differentiation in RAW264.7 Cells by Suppressing NF-κB and NFATc1 Activation and DC-STAMP Expression. Acta Pharmacol. Sin. 2016, 37, 255–263. https://doi.org/10.1038/aps.2015.85.
- 30.
Bonnet, N.; Bourgoin, L.; Biver, E.; et al. RANKL Inhibition Improves Muscle Strength and Insulin Sensitivity and Restores Bone Mass. J. Clin. Invest. 2019, 129, 3214–3223. https://doi.org/10.1172/jci125915.
- 31.
Yan, M.; Su, J.; Li, Y. Rheumatoid Arthritis-Associated Bone Erosions: Evolving Insights and Promising Therapeutic Strategies. BioSci. Trends 2020, 14, 342–348. https://doi.org/10.5582/bst.2020.03253.
- 32.
Yue, Z.; Niu, X.; Yuan, Z.; et al. RSPO2 and RANKL Signal through LGR4 to Regulate Osteoclastic Premetastatic Niche Formation and Bone Metastasis. J. Clin. Invest. 2022, 132, e144579. https://doi.org/10.1172/jci144579.
- 33.
Chen, Z.; Wang, W.; Zhang, Y.; et al. Identification of Four-Gene Signature to Diagnose Osteoarthritis through Bioinformatics and Machine Learning Methods. Cytokine 2023, 169, 156300. https://doi.org/10.1016/j.cyto.2023.156300.
- 34.
Kovács, B.; Vajda, E.; Nagy, E.E. Regulatory Effects and Interactions of the Wnt and OPG-RANKL-RANK Signaling at the Bone-Cartilage Interface in Osteoarthritis. Int. J. Mol. Sci. 2019, 20, 4653. https://doi.org/10.3390/ijms20184653.
- 35.
Danks, L.; Komatsu, N.; Guerrini, M.M.; et al. RANKL Expressed on Synovial Fibroblasts Is Primarily Responsible for Bone Erosions during Joint Inflammation. Ann. Rheum. Dis. 2016, 75, 1187–1195. https://doi.org/10.1136/annrheumdis-2014-207137.
- 36.
Parkin, J.; Cohen, B. An Overview of the Immune System. Lancet 2001, 357, 1777–1789. https://doi.org/10.1016/s0140-6736(00)04904-7.
- 37.
Luo, P.; Yuan, Q.; Wan, X.; et al. Effects of Immune Cells and Cytokines on Different Cells in OA. J. Inflamm. Res. 2023, 16, 2329–2343. https://doi.org/10.2147/jir.S413578.
- 38.
Goldring, M.B.; Otero, M. Inflammation in Osteoarthritis. Curr. Opin. Rheumatol. 2011, 23, 471–478. https://doi.org/10.1097/BOR.0b013e328349c2b1.
- 39.
Yan, Y.; Tao, H.; He, J.; et al. The HDOCK Server for Integrated Protein-Protein Docking. Nat. Protoc. 2020, 15, 1829–1852. https://doi.org/10.1038/s41596-020-0312-x.
- 40.
Lu, J.; Hu, D.; Zhang, Y.; et al. Current Comprehensive Understanding of Denosumab (The RANKL Neutralizing Antibody) in the Treatment of Bone Metastasis of Malignant Tumors, Including Pharmacological Mechanism and Clinical Trials. Front. Oncol. 2023, 13, 1133828. https://doi.org/10.3389/fonc.2023.1133828.
- 41.
Lacey, D.L.; Boyle, W.J.; Simonet, W.S.; et al. Bench to Bedside: Elucidation of the OPG-RANK-RANKL Pathway and the Development of Denosumab. Nat. Rev. Drug Discov. 2012, 11, 401–419. https://doi.org/10.1038/nrd3705.
- 42.
Kostenuik, P.J.; Nguyen, H.Q.; McCabe, J.; et al. Denosumab, a Fully Human Monoclonal Antibody to RANKL, Inhibits Bone Resorption and Increases BMD in Knock-In Mice That Express Chimeric (Murine/Human) RANKL. J. Bone Miner. Res. 2009, 24, 182–195. https://doi.org/10.1359/jbmr.081112.
- 43.
Dougall, W.C.; Holen, I.; González Suárez, E. Targeting RANKL in Metastasis. BoneKEy Rep. 2014, 3, 519. https://doi.org/10.1038/bonekey.2014.14.
- 44.
Chiu, Y.G.; Ritchlin, C.T. Denosumab: Targeting the RANKL Pathway to Treat Rheumatoid Arthritis. Expert Opin. Biol. Ther. 2017, 17, 119–128. https://doi.org/10.1080/14712598.2017.1263614.
- 45.
Bone, H.G.; Chapurlat, R.; Brandi, M.L.; et al. The Effect of Three or Six Years of Denosumab Exposure in Women with Postmenopausal Osteoporosis: Results from the FREEDOM Extension. J. Clin. Endocrinol. Metab. 2013, 98, 4483–4492. https://doi.org/10.1210/jc.2013-1597.
- 46.
Huang, J.G.; Xia, C.; Zheng, X.P.; et al. 17β-Estradiol Promotes Cell Proliferation in Rat Osteoarthritis Model Chondrocytes via PI3K/Akt Pathway. Cell. Mol. Biol. Lett. 2011, 16, 564–575. https://doi.org/10.2478/s11658-011-0023-y.
- 47.
Abramoff, B.; Caldera, F.E. Osteoarthritis: Pathology, Diagnosis, and Treatment Options. Med. Clin. North Am. 2020, 104, 293–311. https://doi.org/10.1016/j.mcna.2019.10.007.
- 48.
Peat, G.; Thomas, M.J. Osteoarthritis Year in Review 2020: Epidemiology & Therapy. Osteoarthr. Cartil. 2021, 29, 180–189. https://doi.org/10.1016/j.joca.2020.10.007.
- 49.
Li, J.; Jiang, M.; Xiong, C.; et al. KLF4, Negatively Regulated by miR-7, Suppresses Osteoarthritis Development via Activating TGF-β1 Signaling. Int. Immunopharmacol. 2022, 102, 108416. https://doi.org/10.1016/j.intimp.2021.108416.
- 50.
Mandl, L.A. Osteoarthritis Year in Review 2018: Clinical. Osteoarthr. Cartil. 2019, 27, 359–364. https://doi.org/10.1016/j.joca.2018.11.001.
- 51.
Yu, D.; Xu, J.; Liu, F.; et al. Subchondral Bone Changes and the Impacts on Joint Pain and Articular Cartilage Degeneration in Osteoarthritis. Clin. Exp. Rheumatol. 2016, 34, 929–934.
- 52.
Nagy, E.; Vajda, E.; Vari, C.; et al. Meloxicam Ameliorates the Cartilage and Subchondral Bone Deterioration in Monoiodoacetate-Induced Rat Osteoarthritis. PeerJ 2017, 5, e3185. https://doi.org/10.7717/peerj.3185.
- 53.
Shi, J.; Schmitt-Talbot, E.; DiMattia, D.A.; et al. The Differential Effects of IL-1 and TNF-Alpha on Proinflammatory Cytokine and Matrix Metalloproteinase Expression in Human Chondrosarcoma Cells. Inflamm. Res. 2004, 53, 377–389. https://doi.org/10.1007/s00011-004-1271-3.
- 54.
Anderson, D.M.; Maraskovsky, E.; Billingsley, W.L.; et al. A Homologue of the TNF Receptor and Its Ligand Enhance T-Cell Growth and Dendritic-Cell Function. Nature 1997, 390, 175–179. https://doi.org/10.1038/36593.
- 55.
Li, Q.; Liu, J.; Liu, W.; et al. LOX-1 Regulates P. gingivalis-Induced Monocyte Migration and Adhesion to Human Umbilical Vein Endothelial Cells. Front. Cell Dev. Biol. 2020, 8, 596. https://doi.org/10.3389/fcell.2020.00596.
- 56.
Manning, J.E.; Lewis, J.W.; Marsh, L.J.; et al. Insights into Leukocyte Trafficking in Inflammatory Arthritis—Imaging the Joint. Front. Cell Dev. Biol. 2021, 9, 635102. https://doi.org/10.3389/fcell.2021.635102.
- 57.
Pezhman, L.; Tahrani, A.; Chimen, M. Dysregulation of Leukocyte Trafficking in Type 2 Diabetes: Mechanisms and Potential Therapeutic Avenues. Front. Cell Dev. Biol. 2021, 9, 624184. https://doi.org/10.3389/fcell.2021.624184.
- 58.
Law, Y.Y.; Lin, Y.M.; Liu, S.C.; et al. Visfatin Increases ICAM-1 Expression and Monocyte Adhesion in Human Osteoarthritis Synovial Fibroblasts by Reducing miR-320a Expression. Aging 2020, 12, 18635–18648. https://doi.org/10.18632/aging.103889.
- 59.
Xiao, S.; Tang, Y.; Lin, Y.; et al. Tracking Osteoarthritis Progress through Cationic Nanoprobe-Enhanced Photoacoustic Imaging of Cartilage. Acta Biomater. 2020, 109, 153–162. https://doi.org/10.1016/j.actbio.2020.04.001.
- 60.
Han, D.; Fang, Y.; Tan, X.; et al. The Emerging Role of Fibroblast-like Synoviocytes-Mediated Synovitis in Osteoarthritis: An Update. J. Cell. Mol. Med. 2020, 24, 9518–9532. https://doi.org/10.1111/jcmm.15669.
- 61.
Liu, J.F.; Hou, S.M.; Tsai, C.H.; et al. CCN4 Induces Vascular Cell Adhesion Molecule-1 Expression in Human Synovial Fibroblasts and Promotes Monocyte Adhesion. Biochim. Biophys. Acta Mol. Cell Res. 2013, 1833, 966–975. https://doi.org/10.1016/j.bbamcr.2012.12.023.
- 62.
Yang, C.R.; Shih, K.S.; Liou, J.P.; et al. Denbinobin Upregulates miR-146a Expression and Attenuates IL-1β-Induced Upregulation of ICAM-1 and VCAM-1 Expressions in Osteoarthritis Fibroblast-like Synoviocytes. J. Mol. Med. 2014, 92, 1147–1158. https://doi.org/10.1007/s00109-014-1192-8.
- 63.
Bertuglia, A.; Lacourt, M.; Girard, C.; et al. Osteoclasts Are Recruited to the Subchondral Bone in Naturally Occurring Post-Traumatic Equine Carpal Osteoarthritis and May Contribute to Cartilage Degradation. Osteoarthr. Cartil. 2016, 24, 555–566. https://doi.org/10.1016/j.joca.2015.10.008.
- 64.
Yan, J.Y.; Tian, F.M.; Wang, W.Y.; et al. Parathyroid Hormone (1–34) Prevents Cartilage Degradation and Preserves Subchondral Bone Micro-Architecture in Guinea Pigs with Spontaneous Osteoarthritis. Osteoarthr. Cartil. 2014, 22, 1869–1877. https://doi.org/10.1016/j.joca.2014.07.013.
- 65.
Tan, W.; Zhang, W.; Strasner, A.; et al. Tumour-Infiltrating Regulatory T Cells Stimulate Mammary Cancer Metastasis through RANKL-RANK Signalling. Nature 2011, 470, 548–553. https://doi.org/10.1038/nature09707.
- 66.
Titanji, K. Beyond Antibodies: B Cells and the OPG/RANK-RANKL Pathway in Health, Non-HIV Disease and HIV-Induced Bone Loss. Front. Immunol. 2017, 8, 1851. https://doi.org/10.3389/fimmu.2017.01851.
- 67.
Camara, A.; Cordeiro, O.G.; Alloush, F.; et al. Lymph Node Mesenchymal and Endothelial Stromal Cells Cooperate via the RANK-RANKL Cytokine Axis to Shape the Sinusoidal Macrophage Niche. Immunity 2019, 50, 1467–1481.e6. https://doi.org/10.1016/j.immuni.2019.05.008.
- 68.
Prieto-Potin, I.; Largo, R.; Roman-Blas, J.A.; et al. Characterization of Multinucleated Giant Cells in Synovium and Subchondral Bone in Knee Osteoarthritis and Rheumatoid Arthritis. BMC Musculoskelet. Disord. 2015, 16, 226. https://doi.org/10.1186/s12891-015-0664-5.
- 69.
Wojdasiewicz, P.; Poniatowski, Ł.A.; Szukiewicz, D. The Role of Inflammatory and Anti-Inflammatory Cytokines in the Pathogenesis of Osteoarthritis. Mediat. Inflamm. 2014, 2014, 561459. https://doi.org/10.1155/2014/561459.
- 70.
Klein-Wieringa, I.R.; de Lange-Brokaar, B.J.; Yusuf, E.; et al. Inflammatory Cells in Patients with Endstage Knee Osteoarthritis: A Comparison between the Synovium and the Infrapatellar Fat Pad. J. Rheumatol. 2016, 43, 771–778. https://doi.org/10.3899/jrheum.151068.
- 71.
Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; et al. Role of Proinflammatory Cytokines in the Pathophysiology of Osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. https://doi.org/10.1038/nrrheum.2010.196.
- 72.
Xu, L.; Hu, Y.J.; Peng, Y.; et al. Early Zoledronate Treatment Inhibits Subchondral Bone Microstructural Changes in Skeletally-Mature, ACL-Transected Canine Knees. Bone 2023, 167, 116638. https://doi.org/10.1016/j.bone.2022.116638.
- 73.
Ziemian, S.N.; Witkowski, A.M.; Wright, T.M.; et al. Early Inhibition of Subchondral Bone Remodeling Slows Load-Induced Posttraumatic Osteoarthritis Development in Mice. J. Bone Miner. Res. 2021, 36, 2027–2038. https://doi.org/10.1002/jbmr.4397.
- 74.
Neogi, T.; Li, S.; Peloquin, C.; et al. Effect of Bisphosphonates on Knee Replacement Surgery. Ann. Rheum. Dis. 2018, 77, 92–97. https://doi.org/10.1136/annrheumdis-2017-211811.
- 75.
Laslett, L.L.; Kingsbury, S.R.; Hensor, E.M.; et al. Effect of Bisphosphonate Use in Patients with Symptomatic and Radiographic Knee Osteoarthritis: Data from the Osteoarthritis Initiative. Ann. Rheum. Dis. 2014, 73, 824–830. https://doi.org/10.1136/annrheumdis-2012-202989.
- 76.
Kadri, A.; Funck-Brentano, T.; Lin, H.; et al. Inhibition of Bone Resorption Blunts Osteoarthritis in Mice with High Bone Remodelling. Ann. Rheum. Dis. 2010, 69, 1533–1538. https://doi.org/10.1136/ard.2009.124586.
- 77.
Bihlet, A.R.; Byrjalsen, I.; Andersen, J.R.; et al. Symptomatic and Structural Benefit of Cathepsin K Inhibition by MIV-711 in a Subgroup with Unilateral Pain: Post-Hoc Analysis of a Randomised Phase 2a Clinical Trial. Clin. Exp. Rheumatol. 2022, 40, 1034–1037. https://doi.org/10.55563/clinexprheumatol/1kvgte.
- 78.
Conaghan, P.G.; Bowes, M.A.; Kingsbury, S.R.; et al. Disease-Modifying Effects of a Novel Cathepsin K Inhibitor in Osteoarthritis: A Randomized Controlled Trial. Ann. Intern. Med. 2020, 172, 86–95. https://doi.org/10.7326/m19-0675.
- 79.
Lindström, E.; Rizoska, B.; Tunblad, K.; et al. The Selective Cathepsin K Inhibitor MIV-711 Attenuates Joint Pathology in Experimental Animal Models of Osteoarthritis. J. Transl. Med. 2018, 16, 56. https://doi.org/10.1186/s12967-018-1425-7.
- 80.
Body, J.J.; Greipp, P.; Coleman, R.E.; et al. A Phase I Study of AMGN-0007, a Recombinant Osteoprotegerin Construct, in Patients with Multiple Myeloma or Breast Carcinoma Related Bone Metastases. Cancer 2003, 97, 887–892. https://doi.org/10.1002/cncr.11138.
- 81.
Di Lorenzo, L. Denosumab in Elderly Osteoporotic Patients. A Narrative Review. Clin. Ter. 2023, 174, 545–549. https://doi.org/10.7417/ct.2023.5023.
- 82.
Wang, Y.; Xu, J.; Zhang, X.; et al. TNF-α-Induced LRG1 Promotes Angiogenesis and Mesenchymal Stem Cell Migration in the Subchondral Bone during Osteoarthritis. Cell Death Dis. 2017, 8, e2715. https://doi.org/10.1038/cddis.2017.129.
- 83.
Weisel, K.; Kanz, L. Lenalidomide. Recent Results Cancer Res. 2014, 201, 347–357. https://doi.org/10.1007/978-3-642-54490-3_21.
- 84.
Reuter, S.; Gupta, S.C.; Phromnoi, K.; et al. Thiocolchicoside Suppresses Osteoclastogenesis Induced by RANKL and Cancer Cells through Inhibition of Inflammatory Pathways: A New Use for an Old Drug. Br. J. Pharmacol. 2012, 165, 2127–2139. https://doi.org/10.1111/j.1476-5381.2011.01702.x.
- 85.
Yan, Y.; Zhang, D.; Zhou, P.; et al. HDOCK: A Web Server for Protein-Protein and Protein-DNA/RNA Docking Based on a Hybrid Strategy. Nucleic Acids Res. 2017, 45, w365–w373. https://doi.org/10.1093/nar/gkx407.

This work is licensed under a Creative Commons Attribution 4.0 International License.


