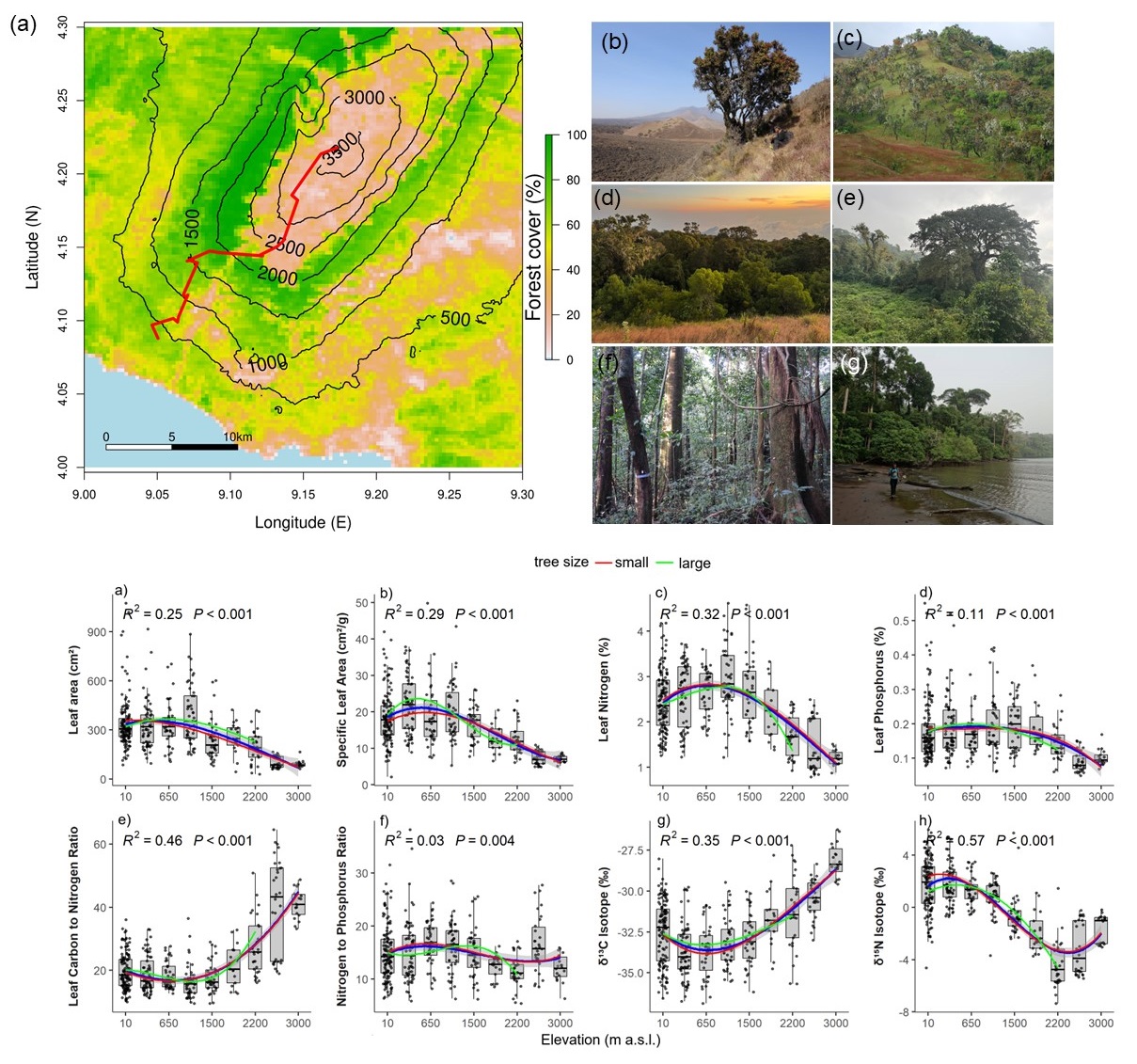
Understanding how tropical trees respond to complex environmental gradients is essential for predicting forest resilience under climate change. We examined variation in key leaf traits, including specific leaf area (SLA), foliar nitrogen (N) and phosphorus (P), C:N and N:P ratios, and stable isotope composition (δ13C, δ15N), in 160 tree species along a 3200-m elevational transect on Mount Cameroon. This gradient spans hyper-humid coastal rainforests to arid Afroalpine savannas, capturing sharp transitions in climate, soils, and forest structure. Leaf traits shifted nonlinearly with elevation, from acquisitive strategies at mid-elevations to conservative syndromes in lowlands and highlands. Mid-elevation forests (~1000–1500 m), characterized by moderate climate and canopy disturbance by elephants, supported nutrient-rich, high-SLA foliage. In contrast, high-elevation forests (>2000 m) exhibited low SLA, high C:N, and enriched δ13C, consistent with stress tolerance under cold, dry, and fire-prone conditions. Leaf N:P ratios indicated the strongest potential phosphorus limitation in hyper-humid lowlands, where extreme rainfall (>12,000 mm yr−1) likely promotes nutrient leaching and low soil P availability. Foliar δ15N declined markedly with elevation (from +5‰ to −5‰), consistent with shifts in nitrogen acquisition strategies and ecosystem N cycling, including increased reliance on mycorrhizal-mediated uptake associated with changes in dominant functional groups (e.g., Fabaceae vs. Ericaceae). A bimodal δ15N pattern, with enrichment in both lowland and upper montane forests, likely reflects contrasting nitrogen loss regimes associated with high rainfall and fire disturbance, respectively. Phylogenetically informed analyses revealed that climate, soils, forest structure, and evolutionary history jointly shaped trait–environment relationships, with strong phylogenetic signal in δ13C, C:N, and δ15N. Together, these results highlight the importance of integrating functional traits, isotopes, and phylogeny to predict tropical forest responses to global change.
- Open Access
- Article
Elevational Shifts in Tropical Tree Leaf Traits: Interactions between Soil, Climate, Light, and Phylogeny
- Jiří Doležal 1,2,*,
- Kirill Korznikov 1,
- Vaclav Bažant 3,
- Thinles Chondol 1,
- Adam Taylor Ruka 1,4,
- Jorge Gago 5
Author Information
Received: 04 Aug 2025 | Revised: 28 Jan 2026 | Accepted: 27 Feb 2026 | Published: 05 Mar 2026
Abstract
Graphical Abstract
Keywords
leaf functional traits | stable isotopes (δ13C | δ15N), tropical montane forests | elevational gradient | nitrogen cycling | mycorrhizal associations | phylogenetic trait conservatism | plant ecophysiology | trait–environment relationships | climate change adaptation
References
- 1.
Abatzoglou JT, Dobrowski SZ, Parks SA, & Hegewisch KC. (2018). TerraClimate, a high-resolution global dataset of monthly climate and climatic water balance from 1958 to 2015. Scientific Data, 5, 170191. https://doi.org/10.1038/sdata.2017.191
- 2.
Ågren GI. (2004). The C:N:P stoichiometry of autotrophs—theory and observations. Ecology Letters, 7, 185–191. https://doi.org/10.1111/j.1461-0248.2004.00567.x
- 3.
Asner GP, Brodrick PG, Philipson C, Vaughn NR, Martin RE, Knapp DE, Heckler J, Evans LJ, Jucker T, Goossens B, Stark DJ, Reynolds G, Ong R, Renneboog N, & Coomes DA. (2018). Mapped aboveground carbon stocks to advance forest conservation and recovery in Malaysian Borneo. Biological Conservation, 217, 289–310. https://doi.org/10.1016/j.biocon.2017.10.020
- 4.
Baraloto C, Paine CET, Poorter L, Beauchene J, Bonal D, Domenach AM, Hérault B, Patiño S, Roggy JC, & Chave J. (2010). Decoupled leaf and stem economics in rain forest trees. Ecology Letters, 13, 1338–1347. https://doi.org/10.1111/j.1461-0248.2010.01517.x
- 5.
Bi R, Fu W, & Fu X. (2024). Phosphorus dynamics in volcanic soils of Weizhou Island, China: implications for environmental and agricultural applications. Environmental Geochemistry and Health, 46, 458. https://doi.org/10.1007/s10653-024-02238-2
- 6.
Cable S, & Cheek M. (1998). The Plants of Mt Cameroon: A Conservation Checklist. Royal Botanic Gardens, Kew.
- 7.
Cavender-Bares J, Kozak KH, Fine PVA, & Kembel SW. (2009). The merging of community ecology and phylogenetic biology. Ecology Letters, 12, 693–715. https://doi.org/10.1111/j.1461-0248.2009.01314.x
- 8.
Cernusak LA, Winter K, & Turner BL. (2010). Leaf nitrogen to phosphorus ratios of tropical trees: experimental assessment of physiological and environmental controls. New Phytologist, 185(3), 770–779. https://doi.org/10.1111/j.1469-8137.2009.03106.x
- 9.
Cheek M, Cable S, Hepper FN, Ndam N, & Watts J. (1996). Mapping plant biodiversity on Mount Cameroon. In The Biodiversity of African Plants (van der Maesen LJG, van der Burgt XM, & van Medenbach de Rooy JM, Eds.; pp. 110–120). Springer. https://doi.org/10.1007/978-94-009-0285-5_16
- 10.
Cornwell WK, & Ackerly DD. (2009). Community assembly and shifts in plant trait distributions across an environmental gradient in coastal California. Ecological Monographs, 79, 109–126. https://doi.org/10.1890/07-1134.1
- 11.
Craine JM, Brookshire ENJ, Cramer MD, Hasselquist NJ, Koba K, Marin-Spiotta E, & Wang L. (2015). Ecological interpretations of nitrogen isotope ratios of terrestrial plants and soils. Plant and Soil, 396, 1–26. https://doi.org/10.1007/s11104-015-2542-1
- 12.
Da Silveira L, Sternberg L, Mulkey SS, & Wright SJ. (1989). Ecological interpretation of leaf carbon isotope ratios: influence of respired carbon dioxide. Ecology, 70, 1317–1324. https://doi.org/10.2307/1938191
- 13.
Delabye S, Maicher V, Sáfián S, Doležal J, Altman J, Janeček Š, Kobe IN, Murkwe M, Šebek P, & Tropek R. (2021). Butterfly and moth communities differ in their response to habitat structure in rainforests of Mount Cameroon. Biotropica, 53(2), 567–580. https://doi.org/10.1111/btp.12900
- 14.
Díaz S, Kattge J, Cornelissen JHC, Wright IJ, Lavorel S, Dray S, Reu B, Kleyer M, Wirth C, Colin Prentice I, Garnier E, Bönisch G, Westoby M, Poorter H, & Gorné LD. (2016). The global spectrum of plant form and function. Nature, 529, 167–171. https://doi.org/10.1038/s41597-022-01774-9
- 15.
Doležal J, Dančák M, Kučera J, Majeský Ľ, Altman J, Řeháková K, Čapková K, Vondrák J, Fibich P, & Liancourt P. (2022). Fire, climate and biotic interactions shape diversity patterns along an Afrotropical elevation gradient. Journal of Biogeography, 49, 1248–1259. https://doi.org/10.1111/jbi.14378
- 16.
Doležal J, Korznikov K, Altman J, Fibich P, Řeháková K, Lltonga EN, Majeský Ľ, & Dančák M. (2023). Ecological niches of epiphyllous bryophytes along an Afrotropical elevation gradient. Oikos, 2023, e09772. http://dx.doi.org/10.1111/oik.09772
- 17.
Doležal, J. (2026). Data from: Elevational Shifts in Tropical Tree Leaf Traits: Interactions between Soil, Climate, Light, and Phylogeny. In Plant Ecophysiology (Vol. 2, Number 1). Zenodo. https://doi.org/10.5281/zenodo.1
- 18.
Egli M, Dahms D, & Norton K. (2014). Soil formation rates on silicate parent material in alpine environments: Different approaches–different results? Geoderma, 213, 320–333. https://doi.org/10.1016/j.geoderma.2013.08.016
- 19.
Fan R, Morozumi T, Maximov TC, & Sugimoto A. (2018). Effect of floods on the δ13C values in plant leaves: a study of willows in northeastern Siberia. PeerJ 6, e5374. https://doi.org/10.7717/peerj.5374
- 20.
Farquhar GD, Ehleringer JR, & Hubick KT. (1989). Carbon isotope discrimination and photosynthesis. Annual Review of Plant Physiology and Plant Molecular Biology, 40, 503–537. https://doi.org/10.1146/annurev.pp.40.060189.002443
- 21.
Ferenc M, Sedláček O, Tropek R, Albrecht T, Altman J, Dančák M, & Hořák D. (2018). Something is missing at the bottom: Importance of coastal rainforests for conservation of trees, birds and butterflies in the Mount Cameroon area. African Journal of Ecology, 56, 679–683. https://doi.org/10.1111/aje.12506
- 22.
Flexas J, Bota J, Loreto F, Cornic G. & Sharkey T.D. (2004), Diffusive and Metabolic Limitations to Photosynthesis under Drought and Salinity in C3 Plants. Plant Biology, 6, 269–279. https://doi.org/10.1055/s-2004-820867
- 23.
Flexas J, Zhang Y, Gulías J, Xiong D, Carriquí M, Baraza E, Du T, Lei Z, Meng H, Dou H, Ribas-Carbo M, Xiang D, & Xu W. (2022). Leaf physiological traits of plants from the Qinghai–Tibet Plateau and other arid sites in China: Identifying susceptible species and well-adapted extremophiles. Journal of Plant Physiology, 272, 153689. https://doi.org/10.1016/j.jplph.2022.153689
- 24.
Fogel ML, Wooller MJ, Cheeseman J, Smallwood BJ, Roberts Q, Romero I, & Meyers MJ. (2008). Unusually negative nitrogen isotopic compositions (δ15N) of mangroves and lichens in an oligotrophic, microbially influenced ecosystem. Biogeosciences, 5, 1693–1704. https://doi.org/10.5194/bg-5-1693-2008
- 25.
Fyllas NM, Bentley LP, Shenkin A, Asner GP, Atkin OK, Díaz S, Enquist BJ, Farfan-Rios W, Gloor E, Guerrieri R. (2017). Solar radiation and functional traits explain the decline of forest primary productivity along a tropical elevation gradient. Ecology Letters, 20, 730–740. https://doi.org/10.1111/ele.12771
- 26.
Garnier E, Navas ML, & Grigulis K. (2015). Plant Functional Diversity: Organism Traits, Community Structure, and Ecosystem Properties. Oxford University Press. https://doi.org/10.1093/acprof:oso/9780198757368.001.0001
- 27.
Girardin CA, Malhi Y, Feeley KJ, Rapp JM, Silman MR, Meir P, & Aragão LEOC. (2014). Seasonality of above-ground net primary productivity along an Andean altitudinal transect in Peru. Journal of Tropical Ecology, 30, 1–17. https://doi.org/10.1017/S0266467414000443
- 28.
Girardin CA, Malhi Y, Doughty CE, Metcalfe DB, Meir P, del Aguila‐Pasquel J, & Rowland L. (2016). Seasonal trends of Amazonian rainforest phenology, net primary productivity, and carbon allocation. Global Biogeochemical Cycles, 30, 700–715. https://doi.org/10.1002/2015GB005270
- 29.
Givnish TJ. (2002). Adaptive significance of evergreen vs deciduous leaves: solving the triple paradox. Silva Fennica, 36, 703–743. https://doi.org/10.14214/sf.535
- 30.
Goldsmith GR, Bentley LP, Shenkin A, Salinas N, Blonder B, Martin RE, & Malhi Y. (2017). Variation in leaf wettability traits along a tropical montane elevation gradient. New Phytologist, 214, 989–1001. https://doi.org/10.1111/nph.14121
- 31.
Güsewell S. (2004). N:P ratios in terrestrial plants: variation and functional significance. New Phytologist, 164, 243–266. https://doi.org/10.1111/j.1469-8137.2004.01192.x
- 32.
Hall JB. (1973). Vegetational zones on the southern slopes of Mount Cameroon. Vegetatio, 27(1), 49–69. https://doi.org/10.1007/BF02389340
- 33.
Helsen K, Viana JL, Lin TY, Kuo LY, & Zelený D. (2023). Functional-trait contrasts between terrestrial and epiphytic ferns in Taiwanese subtropical cloud forests. Journal of Vegetation Science, 34, e13220. https://doi.org/10.1111/jvs.13220
- 34.
Hobbie EA, & Högberg P. (2012). Nitrogen isotopes link mycorrhizal fungi and plants to nitrogen dynamics. New Phytologist, 196, 367–382. https://doi.org/10.1111/j.1469-8137.2012.04300.x
- 35.
Högberg P. (1997). Tansley Review No. 95: 15N natural abundance in soil–plant systems. New Phytologist, 137, 179–203. https://doi.org/10.1046/j.1469-8137.1997.00808.x
- 36.
Hogue BA, & Inglett PW. (2012). Nutrient release from combustion residues of two contrasting herbaceous vegetation types. Science of the Total Environment, 431, 9–19. https://doi.org/10.1016/j.scitotenv.2012.04.074
- 37.
Holtum JAM, & Winter K. (2005). Carbon isotope composition of canopy leaves in a tropical forest in Panama throughout a seasonal cycle. Trees, 19, 545–551. https://doi.org/10.1007/s00468-005-0413-8
- 38.
Homeier J, Seeler T, Pierick K, & Leuschner C. (2021). Leaf trait variation in species-rich tropical Andean forests. Scientific Reports, 11, 9993. https://doi.org/10.1038/s41598-021-89190-8
- 39.
Hořák D, Ferenc M, Sedláček O, Motombi FN, Svoboda M, Altman J, Albrecht T, Djomo Nana E, Janeček Š, Dančák M, Majeský Ľ, Lltonga EN, & Doležal J. (2019). Forest structure determines spatial changes in avian community along an elevational gradient in tropical Africa. Journal of Biogeography, 46, 2466–2478. https://doi.org/10.1111/jbi.13688
- 40.
Islam MA, & Macdonald SE. (2004). Ecophysiological adaptations of black spruce (Picea mariana) and tamarack (Larix laricina) seedlings to flooding. Trees—Structure and Function, 18, 35–42. https://doi.org/10.7939/R36H4CQ0C
- 41.
Kitayama K, & Aiba SI. (2002). Ecosystem structure and productivity of tropical rain forests along altitudinal gradients with contrasting soil phosphorus pools on Mount Kinabalu, Borneo. Journal of Ecology, 90, 37–51. https://doi.org/10.1046/j.0022-0477.2001.00634.x
- 42.
Kitajima K, & Poorter L. (2010). Tissue‐level leaf toughness, but not lamina thickness, predicts sapling leaf lifespan and shade tolerance of tropical tree species. New Phytologist, 186(3), 708–721. https://doi.org/10.1111/j.1469-8137.2010.03212.x
- 43.
Koerselman W, & Meuleman AFM. (1996). The vegetation N:P ratio: a new tool to detect the nature of nutrient limitation. Journal of Applied Ecology, 33, 1441–1450. https://doi.org/10.2307/2404783
- 44.
Körner C, Farquhar GD, & Roksandic Z. (1988). A global survey of carbon isotope discrimination in plants from high altitude. Oecologia, 74(4), 623–632. https://doi.org/10.1007/BF00380063
- 45.
Körner C. (2007). The use of ‘altitude’ in ecological research. Trends in Ecology & Evolution, 22(11), 569–574. https://doi.org/10.1016/j.tree.2007.09.006
- 46.
Körner C. (2021). Alpine Plant Life: Functional Plant Ecology of High Mountain Ecosystems. Springer Nature. https://doi.org/10.1007/978-3-030-59538-8
- 47.
Kreuzwieser J, & Rennenberg H. (2014). Molecular and physiological responses of trees to waterlogging stress. Plant, Cell & Environment, 37, 2245–2259. https://doi.org/10.1111/pce.12310
- 48.
Laughlin DC, & Messier J. (2015). Fitness of multidimensional phenotypes in dynamic adaptive landscapes. Trends in Ecology & Evolution, 30, 487–496. https://doi.org/10.1016/j.tree.2015.06.003
- 49.
Laughlin DC. (2014). The intrinsic dimensionality of plant traits and its relevance to community assembly. Journal of Ecology, 102, 186–193. https://doi.org/10.1111/1365-2745.12187
- 50.
Maicher V, Delabye S, Murkwe M, Doležal J, Altman J, Kobe IN, Desmist J, Fokam EB, Pyrcz T, & Tropek R. (2020). Effects of disturbances by forest elephants on diversity of trees and insects in tropical rainforests on Mount Cameroon. Scientific Reports, 10, 21618. https://doi.org/10.1038/s41598-020-78659-7
- 51.
Markesteijn L, & Poorter L. (2009). Seedling root morphology and biomass allocation of 62 tropical tree species in relation to drought- and shade-tolerance. Journal of Ecology, 97, 311–325. https://doi.org/10.1111/j.1365-2745.2008.01466.x
- 52.
Markesteijn L, Poorter L, Bongers F, Paz H, & Sack L. (2011). Hydraulics and life history of tropical dry forest tree species: coordination of species’ drought and shade tolerance. New Phytologist, 191(2), 480–495. https://doi.org/10.1111/j.1469-8137.2011.03708.x
- 53.
Martinelli LA, Almeida S, Brown IF, Moreira MZ, Victoria RL, Sternberg LSL, Ferreira CAC, & Thomas WW. (1998). Stable carbon isotope ratio of tree leaves, boles and fine litter in a tropical forest in Rondônia, Brazil. Oecologia, 114, 170–179. https://doi.org/10.1007/s004420050433
- 54.
Martinelli LA, Piccolo MC, Townsend AR, Vitousek PM, Cuevas E, McDowell W, Robertson GP, Santos OC, & Treseder K. (1999). Nitrogen stable isotope composition of leaves and soil: tropical versus temperate forests. Biogeochemistry, 46, 45–65. https://doi.org/10.1023/A:1006100128782
- 55.
McCullagh P, & Nelder JA. (1989). Generalized linear models (2nd ed.). Chapman and Hall. https://doi.org/10.1201/9780203753736
- 56.
Méndez-Alonzo R, Paz H, Zuluaga RC, Rosell JA, & Olson ME. (2012). Coordinated evolution of leaf and stem economics in tropical dry forest trees. Ecology, 93, 2397–2406. https://doi.org/10.1890/11-1213.1
- 57.
Morgan JAW, Bending GD, & White PJ. (2005). Biological costs and benefits to plant–microbe interactions in the rhizosphere. Journal of Experimental Botany, 56, 1729–1739. https://doi.org/10.1093/jxb/eri205
- 58.
Moser G, Hertel D, & Leuschner C. (2007). Altitudinal change in LAI and stand leaf biomass in tropical montane forests: a transect study in Ecuador and a pan-tropical meta-analysis. Ecosystems, 10, 924–935. https://doi.org/10.1007/s10021-007-9063-6
- 59.
Oliveras I, Bentley L, Fyllas NM, Gvozdevaite A, Shenkin AF, Peprah T, Morandi P, Peixoto KS, Boakye M, Adu-Bredu S, Schwantes Marimon B, Marimon Junior BH, Salinas N, Martin R, …Malhi Y. (2020). The influence of taxonomy and environment on leaf trait variation along tropical abiotic gradients. Frontiers in Forests and Global Change, 3, 18. https://doi.org/10.3389/ffgc.2020.00018
- 60.
Ordoñez JC, Van Bodegom PM, Witte JPM, Wright IJ, Reich PB, & Aerts R. (2009). A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Global Ecology and Biogeography, 18, 137–149. https://doi.org/10.1111/j.1466-8238.2008.00441.x
- 61.
Payton RW. (1993). Final Project Report R4600: Ecology, Altitudinal Zonation and Conservation of Tropical Rain Forests of Mount Cameroon. Soil Survey and Land Research Centre.
- 62.
Plavcová L, Jandová V, Altman J, Liancourt P, Korznikov K, & Doležal J. (2024). Variations in wood anatomy in Afrotropical trees with a particular emphasis on radial and axial parenchyma. Annals of Botany, 134, https://doi.org/151–162. 10.1093/aob/mcae049
- 63.
Poorter L. (2009). Leaf traits show different relationships with shade tolerance in moist versus dry tropical forests. New Phytologist, 181(4), 890–900. https://doi.org/10.1111/j.1469-8137.2008.02715.x
- 64.
Porder S, & Hilley GE. (2011). Linking chronosequences with the rest of the world: predicting soil phosphorus content in denuding landscapes. Biogeochemistry, 102(1), 153–166. https://doi.org/10.1007/s10533-010-9428-3
- 65.
Porder S, & Ramachandran S. (2013). The phosphorus concentration of common rocks—A potential driver of ecosystem P status. Plant and Soil, 367, 41–55. https://doi.org/10.1007/s11104-012-1490-2
- 66.
Pringle EG, Adams RI, Broadbent E, Busby PE, Donatti CI, Kurten EL, Renton K, & Dirzo R. (2011). Distinct leaf-trait syndromes of evergreen and deciduous trees in a seasonally dry tropical forest. Biotropica, 43, 299–308. https://doi.org/10.1111/j.1744-7429.2010.00697.x
- 67.
Proctor J, Edwards ID, Payton RW, & Nagy L. (2007). Zonation of forest vegetation and soils of Mount Cameroon, West Africa. Plant Ecology, 192, 251–269. https://doi.org/10.1007/s11258-007-9326-5
- 68.
Quesada CA, Lloyd J, Schwarz M, Patiño S, Baker TR, Czimczik C, & Paiva R. (2010). Variations in chemical and physical properties of Amazon forest soils in relation to their genesis. Biogeosciences, 7(5), 1515–1541. https://doi.org/10.5194/bg-7-1515-2010
- 69.
Řeháková K, Čapková K, Altman J, Dančák M, Majeský Ľ, & Doležal J. (2022). Contrasting patterns of soil chemistry and vegetation cover determine diversity changes of soil phototrophs along an Afrotropical elevation gradient. Ecosystems, 25, 1020–1036. https://doi.org/10.1007/s10021-021-00698-6
- 70.
Reich PB. (2014). The world‐wide ‘fast–slow’plant economics spectrum: a traits manifesto. Journal of Ecology, 102(2), 275–301. https://doi.org/10.1111/1365-2745.12211
- 71.
Santiago L, Silvera K, Andrade J, & Dawson T. (2017). Functional strategies of tropical dry forest plants in relation to growth form and isotopic composition. Environmental Research Letters, 12, 1088–9326. https://doi.org/10.1088/1748-9326/aa8959
- 72.
Schreeg L, Santiago L, Wright SJ, & Turner BL. (2014). Stem, root, and older leaf N: P ratios are more responsive indicators of soil nutrient availability than new foliage. Ecology, 95, 2062–2068. https://doi.org/10.1890/13-1671.1
- 73.
Soethe N, Lehmann J, & Engels C. (2008). Nutrient availability at different altitudes in a tropical montane forest in Ecuador. Journal of Tropical Ecology, 24, 397–406. https://doi.org/10.1017/S026646740800504X
- 74.
Swenson NG, & Enquist BJ. (2009). Opposing assembly mechanisms in a Neotropical dry forest: Implications for phylogenetic and functional community ecology. Ecology, 90, 2161–2170. https://doi.org/10.1890/08-1025.1
- 75.
ter Braak CJF, & Šmilauer P. (2012). Canoco Reference Manual and User’s Guide: Software for Ordination (Version 5.0). Microcomputer Power.
- 76.
Townsend AR, Cleveland CC, Asner GP, & Bustamante MMC. (2007). Controls over foliar N:P ratios in tropical rain forests. Ecology, 88, 107–118. https://doi.org/10.1890/0012-9658(2007)88[107:COFNRI]2.0.CO;2
- 77.
Turner BL, Bielnicka AW, & Kim PJ. (2024). Stable soil phosphorus transformations following long-term litter manipulation in a lowland tropical forest. Soil Advances, 1, 100002. https://doi.org/10.1016/j.soilad.2024.100002
- 78.
Vargas G, Brodribb TJ, Dupuy JM, González-M R, Hulshof CM, Medvigy D, Allerton TA, Pizano C, Salgado-Negret B, Schwartz NB (2021). Beyond leaf habit: generalities in plant function across 97 tropical dry forest tree species. New Phytologist 232, 148–161. https://doi.org/10.1111/nph.17584
- 79.
Veldhuis MP, Hulshof A, Fokkema W, Berg MP, & Olff H. (2016). Understanding nutrient dynamics in an African savanna: Local biotic interactions outweigh a major regional rainfall gradient. Journal of Ecology, 104, 913–923. https://doi.org/10.1111/1365-2745.12569
- 80.
Vitousek PM, & Howarth RW (1991). Nitrogen limitation on land and in the sea—How can it occur? Biogeochemistry, 13, 87–115. https://doi.org/10.1007/BF00002772
- 81.
Vitousek PM (2004). Nutrient Cycling and Limitation: Hawai‘i as a Model System. Princeton University Press, Princeton, NJ, USA.
- 82.
Vitousek PM, & Sanford RL. (1986). Nutrient cycling in moist tropical forest. Annual Review of Ecology and Systematics, 17, 137–167. https://doi.org/10.1146/annurev.es.17.110186.001033
- 83.
Vitousek PM, Porder S, Houlton BZ, & Chadwick OA. (2010). Terrestrial phosphorus limitation: Mechanisms, implications, and nitrogen–phosphorus interactions. Ecological Applications, 20, 5–15. https://doi.org/10.1890/08-0127.1
- 84.
Vu JCV, & Yelenosky G. (1992). Photosnythetic responses of rough lemon and sour orange to soil flooding, chilling, and short-term temperature fluctuations during growth. Environmental and Experimental Botany, 32, 471–477. https://doi.org/10.1016/0098-8472(92)90060-F
- 85.
Walker TW & Syers JK (1976). The fate of phosphorus during pedogenesis. Geoderma 15: 1–19. https://doi.org/10.1016/0016-7061(76)90066-5
- 86.
Waring EF, & Maricle BR. (2012). Photosynthetic variation and carbon isotope discrimination in invasive wetland grasses in response to flooding. Environmental and Experimental Botany, 77: 77–86. https://doi.org/10.1016/j.envexpbot.2011.10.013
- 87.
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F, & Villar R. (2004). The worldwide leaf economics spectrum. Nature, 428, 821–827. https://doi.org/10.1038/nature02403
- 88.
Wright IJ, Reich PB, & Westoby M. (2001). Strategy shifts in leaf physiology, structure and nutrient content between species of high‐and low‐rainfall and high‐and low‐nutrient habitats. Functional Ecology, 15(4), 423–434. https://doi.org/10.1046/j.0269-8463.2001.00542.x
- 89.
Yang Y, Siegwolf RTW, & Körner C. (2015). Species-specific and environment-induced variation of δ13C and δ15N in alpine plants. Frontiers in Plant Science, 6, 423. https://doi.org/10.3389/fpls.2015.00423

This work is licensed under a Creative Commons Attribution 4.0 International License.


