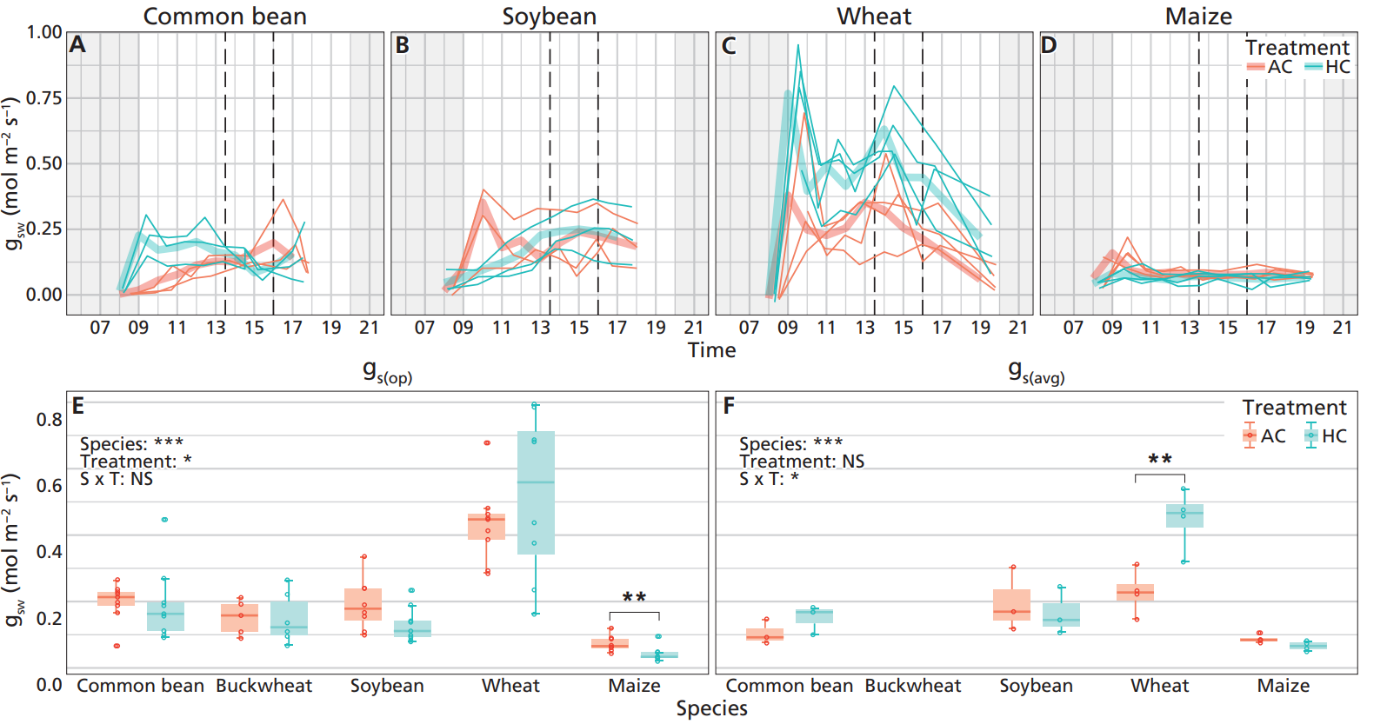
Understanding leaf trait responses to changing atmospheric CO2 concentrations ([CO2]) is essential for land- surface modelling. Eco-evolutionary optimality (EEO) theory predicts coordinated responses in leaf traits that maintain a balance of maximum carbon assimilation gain with minimal summed resource use costs. Although individual leaf trait responses to atmospheric [CO2] changes are relatively well understood, few experiments have documented coordination of responses across functional leaf trait categories related to photosynthesis, photosynthetic biochemistry, stomatal conductance (gs), and morphology. We examined leaf trait coordination in five common crop species, including buckwheat (C3), common bean (C3), maize (C4), soybean (C3), and wheat (C3). We specifically included traits relevant for testing EEO theory, including diurnal measurements of gs. Our results showed that among the four C3 species, gs and photosynthetic biochemistry traits were generally downregulated under elevated atmospheric [CO2], while photosynthetic rates increased, in line with P-model predictions. A principal component analysis (PCA) on the response ratios across C3 species revealed coordinated trait variation along two main axes: principal component (PC) 1 mainly described leaf economic traits and PC2 was mainly related to photosynthetic biochemistry and hydraulic capacity, indicating partial decoupling of leaf trait coordination. Intraspecific variation can arise from differences in photosynthetic pathways, as shown by the distinctive responses of the C4 species maize, with potential consequences for trait coordination. Our results provide support for EEO theory on predictions of leaf trait combinations and vegetation patterns under future climates.
- Open Access
- Article
Experimental Evidence for Coordinated Leaf Trait Responses to Elevated CO2 in Five Common Crop Species
- Astrid Odé 1,*,
- Paul L. Drake 2,
- Erik J. Veneklaas 2,3,
- Jan A. Lankhorst 1,
- Karin T. Rebel 1,
- Hugo J. de Boer 1
Author Information
Received: 19 Dec 2025 | Revised: 22 Mar 2026 | Accepted: 09 Apr 2026 | Published: 15 Apr 2026
Abstract
Graphical Abstract
Keywords
eco-evolutionary optimality | leaf trait coordination | vegetation modelling | photosynthetic capacity | stomatal conductance | leaf economic spectrum
References
- 1.
Ainsworth EA, Davey PA, Bernacchi CJ, Dermody OC, Heaton EA, Moore DJ, Morgan PB, Naidu SL, Ra HSY, Zhu XG, Curtis PS, & Long SP. (2002). A meta-analysis of elevated [CO2] effects on soybean (Glycine max) physiology, growth and yield. Global Change Biology, 8(8), 695–709. https://doi.org/10.1046/j.1365-2486.2002.00498.x
- 2.
Ainsworth EA, & Long SP. (2005). What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytologist, 165(2), 351–372. https://doi.org/10.1111/j.1469-8137.2004.01224.x
- 3.
Ainsworth EA, & Long SP. (2021). 30 years of free-air carbon dioxide enrichment (FACE): What have we learned about future crop productivity and its potential for adaptation? Global Change Biology, 27(1), 27–49. https://doi.org/10.1111/gcb.15375
- 4.
Ainsworth EA, & Rogers A. (2007). The response of photosynthesis and stomatal conductance to rising [CO2]: Mechanisms and environmental interactions. Plant, Cell and Environment, 30(3), 258–270. https://doi.org/10.1111/j.1365-3040.2007.01641.x
- 5.
Albert CH, Grassein F, Schurr FM, Vieilledent G, & Violle C. (2011). When and how should intraspecific variability be considered in trait-based plant ecology? Perspectives Plant Ecology, Evolution and Systematics, 13(3), 217–225. https://doi.org/10.1016/j.ppees.2011.04.003
- 6.
Anderegg LDL. (2023). Why can’t we predict traits from the environment? New Phytologist, 237(6), 1998–2004. https://doi.org/10.1111/nph.18586
- 7.
Augustine SP, & McCulloh KA. (2024). Physiological trait coordination and variability across and within three Pinus species. New Phytologist, 244(2), 451–463. https://doi.org/10.1111/nph.19859
- 8.
Blackman CJ, Aspinwall MJ, Resco de Dios V, Smith RA, & Tissue DT. (2016). Leaf photosynthetic, economics and hydraulic traits are decoupled among genotypes of a widespread species of eucalypt grown under ambient and elevated CO2. Functional Ecology, 30(9), 1491–1500. https://doi.org/10.1111/1365-2435.12661
- 9.
Bokshi AI, Thistlethwaite RJ, Chaplin ED, Kirii E, Trethowan RM, & Tan DKY. (2022). Physiological traits for evaluating heat-tolerance of Australian spring wheat cultivars at elevated CO2. Journal of Agronomy and Crop Science, 208(2), 178–196. https://doi.org/10.1111/jac.12584
- 10.
Borchers HW. (2011). pracma: Practical Numerical Math Functions. In CRAN: Contributed Packages. CRAN. https://doi.org/10.32614/CRAN.package.pracma
- 11.
Boyd RA, Gandin A, & Cousins AB. (2015). Temperature responses of C4 photosynthesis: Biochemical analysis of Rubisco, phosphoenolpyruvate carboxylase, and carbonic anhydrase in Setaria viridis. Plant Physiology, 169(3), 1850–1861. https://doi.org/10.1104/pp.15.00586
- 12.
Brodribb TJ, & Feild TS. (2010). Leaf hydraulic evolution led a surge in leaf photosynthetic capacity during early angiosperm diversification. Ecology Letters, 13(2), 175–183. https://doi.org/10.1111/j.1461-0248.2009.01410.x
- 13.
Brodribb TJ, Feild TS, & Jordan GJ. (2007). Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiology, 144(4), 1890–1898. https://doi.org/10.1104/pp.107.101352
- 14.
Buckley TN, & Diaz-Espejo A. (2015). Reporting estimates of maximum potential electron transport rate. New Phytologist, 205(1), 14–17. Blackwell Publishing Ltd. https://doi.org/10.1111/nph.13018
- 15.
Cardoso-Vilhena J, & Barnes J. (2001). Does nitrogen supply affect the response of wheat (Triticum aestivum cv. Hanno) to the combination of elevated CO2 and O3? Journal of Experimental Botany, 52(362), 1901–1911.
- 16.
Chavan SG, Duursma RA, Tausz M, & Ghannoum O. (2019). Elevated CO2 alleviates the negative impact of heat stress on wheat physiology but not on grain yield. Journal of Experimental Botany, 70(21), 6447–6459. https://doi.org/10.1093/jxb/erz386
- 17.
Conesa MA, Muir CD, Molins A, & Galmés J. (2020). Stomatal anatomy coordinates leaf size with Rubisco kinetics in the Balearic Limonium. AoB PLANTS, 12(1), plz050. https://doi.org/10.1093/aobpla/plz050
- 18.
Cui E, Weng E, Yan E, & Xia J. (2020). Robust leaf trait relationships across species under global environmental changes. Nature Communications, 11(1), 2999. https://doi.org/10.1038/s41467-020-16839-9
- 19.
de Boer HJ, Price CA, Wagner-Cremer F, Dekker SC, Franks PJ, & Veneklaas EJ. (2016). Optimal allocation of leaf epidermal area for gas exchange. New Phytologist, 210(4), 1219–1228. https://doi.org/10.1111/nph.13929
- 20.
Deans RM, Brodribb TJ, Busch FA, & Farquhar GD. (2020). Optimization can provide the fundamental link between leaf photosynthesis, gas exchange and water relations. Nature Plants, 6(9), 1116–1125. https://doi.org/10.1038/s41477-020-00760-6
- 21.
Dong N, Prentice IC, Wright IJ, Evans BJ, Togashi HF, Caddy-Retalic S, McInerney FA, Sparrow B, Leitch E, & Lowe AJ. (2020). Components of leaf-trait variation along environmental gradients. New Phytologist, 228(1), 82–94. https://doi.org/10.1111/nph.16558
- 22.
Dong N, Prentice IC, Wright IJ, Wang H, Atkin OK, Bloomfield KJ, Domingues TF, Gleason SM, Maire V, Onoda Y, Poorter H, & Smith NG. (2022). Leaf nitrogen from the perspective of optimal plant function. Journal of Ecology, 110(11), 2585–2602. https://doi.org/10.1111/1365-2745.13967
- 23.
Drake PL, Froend RH, & Franks PJ. (2013). Smaller, faster stomata: Scaling of stomatal size, rate of response, and stomatal conductance. Journal of Experimental Botany, 64(2), 495–505. https://doi.org/10.1093/jxb/ers347
- 24.
Duursma RA. (2015). Plantecophys—An R package for analysing and modelling leaf gas exchange data. PLoS ONE, 10(11), e0143346. https://doi.org/10.1371/journal.pone.0143346
- 25.
Fan Y, Asao S, Furbank RT, von Caemmerer S, Day DA, Tcherkez G, Sage TL, Sage RF, & Atkin OK. (2022). The crucial roles of mitochondria in supporting C4 photosynthesis. New Phytologist, 233(3), 1083–1096. https://doi.org/10.1111/nph.17818
- 26.
Fan Y, Noble DWA, Medlyn BE, Monson RK, Sage RF, Smith NG, Ainsworth EA, Busch FA, Danila FR, Ermakova M, Friesen P, Furbank RT, Gan SH, Ghannoum O, … & Way DA. (2025). Environmental factors have a greater influence on photosynthetic capacity in C4 plants than biochemical subtypes or growth forms. New Phytologist, 248(3), 1205–1224. https://doi.org/10.1111/nph.70525
- 27.
Farquhar GD, von Caemmerer S, & Berry JA. (1980). A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta, 149(1), 78–90. https://doi.org/10.1007/BF00386231
- 28.
*Farquhar GD, O’Leary MH, & Berry JA. (1982). On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Australian Journal of Plant Physiology, 9(2), 121–137. https://doi.org/10.1071/PP9820121
- 29.
*Farquhar GD, & Richards RA. (1984). Isotopic composition of plant carbon correlates with water-use efficiency of wheat genotypes. Australian Journal of Plant Physiology, 11(6), 539–552. https://doi.org/10.1071/PP9840539
- 30.
*Feng X. (1999). Trends in intrinsic water-use efficiency of naural trees for the past 100-200 years: A response to atmospheric CO2 concentration. Geochimica et Cosmochimica Acta, 63(13–14), 1891–1903. https://doi.org/10.1016/S0016-7037(99)00088-5
- 31.
Ferris R, Sabatti M, Miglietta F, Mills RF, & Taylor G. (2001). Leaf area is stimulated in Populus by free air CO2 enrichment (POPFACE), through increased cell expansion and production. Plant, Cell and Environment, 24(3), 305–315. https://doi.org/10.1046/j.1365-3040.2001.00684.x
- 32.
Fox J, Weisberg S, & Price B. (2001). car: Companion to Applied Regression. In CRAN: Contributed Packages. CRAN. https://doi.org/10.32614/CRAN.package.car
- 33.
Franklin O, Harrison SP, Dewar R, Farrior CE, Brännström Å, Dieckmann U, Pietsch S, Falster D, Cramer W, Loreau M, Wang H, Mäkelä A, Rebel KT, Meron E, … & Colin I. (2020). Organizing principles for vegetation dynamics. Nature Plants, 6(5), 444–453. https://doi.org/10.1038/s41477-020-0655-x
- 34.
Franks PJ, & Beerling DJ. (2009). Maximum leaf conductance driven by CO2 effects on stomatal size and density over geologic time. Proceedings of the National Academy of Sciences of the United States of America, 106(25), 10343–10347. https://doi.org/10.1073/pnas.0904209106
- 35.
Franks PJ, Drake PL, & Beerling DJ. (2009). Plasticity in maximum stomatal conductance constrained by negative correlation between stomatal size and density: An analysis using Eucalyptus globulus. Plant, Cell and Environment, 32(12), 1737–1748. https://doi.org/10.1111/j.1365-3040.2009.002031.x
- 36.
Franks PJ, & Farquhar GD. (2001). The Effect of Exogenous Abscisic Acid on Stomatal Development, Stomatal Mechanics, and Leaf Gas Exchange in Tradescantia virginiana. Plant Physiology, 125(2), 935–942.
- 37.
Franks PJ, Leitch IJ, Ruszala EM, Hetherington AM, & Beerling DJ. (2012). Physiological framework for adaptation of stomata to CO2 from glacial to future concentrations. Philosophical Transactions of the Royal Society B: Biological Sciences, 367(1588), 537–546. https://doi.org/10.1098/rstb.2011.0270
- 38.
Franks PJ, Royer DL, Beerling DJ, Van De Water PK, Cantrill DJ, Barbour MM, & Berry JA. (2014). New constraints on atmospheric CO2 concentration for the Phanerozoic. Geophysical Research Letters, 41(13), 4685–4694. https://doi.org/10.1002/2014GL060457
- 39.
Ghannoum O, Al-Salman YM, & Cano FJ. (2025). Opportunities for improving intrinsic water use efficiency in C4 plants under climate change. New Phytologist, 248(6), 2656–2673. https://doi.org/10.1111/nph.70660
- 40.
Ghannoum O, Von Caemmerer S, Ziska LH, & Conroy JP. (2000). The growth response of C4 plants to rising atmospheric CO2 partial pressure: A reassessment. Plant, Cell and Environment, 23(9), 931–942. https://doi.org/10.1046/j.1365-3040.2000.00609.x
- 41.
Givnish TJ, Pires JC, Graham SW, McPherson MA, Prince LM, Patterson TB, Rai HS, Roalson EH, Evans TM, Hahn WJ, Millam RC, Meerow AW, Molvray M, Kores PJ, … & Sytsma KJ. (2005). Repeated evolution of net venation and fleshy fruits among monocots in shaded habitats confirms a priori predictions: Evidence from an ndhF phylogeny. Proceedings of the Royal Society B: Biological Sciences, 272(1571), 1481–1490. https://doi.org/10.1098/rspb.2005.3067
- 42.
Grossiord C, Buckley TN, Cernusak LA, Novick KA, Poulter B, Siegwolf RTW, Sperry JS, & McDowell NG. (2020). Plant responses to rising vapor pressure deficit. New Phytologist, 226(6), 1550–1566. https://doi.org/10.1111/nph.16485
- 43.
Han Y, Wang J, Zhang Y, & Wang S. (2023). Effects of Regulated Deficit Irrigation and Elevated CO2 Concentration on the Photosynthetic Parameters and Stomatal Morphology of Two Maize Cultivars. Journal of Plant Growth Regulation, 42(5), 2884–2892. https://doi.org/10.1007/s00344-022-10754-7
- 44.
Harrison SP, Cramer W, Franklin O, Prentice IC, Wang H, Brännström Å, de Boer H, Dieckmann U, Joshi J, Keenan TF, Lavergne A, Manzoni S, Mengoli G, Morfopoulos C, … & Wright IJ. (2021). Eco-evolutionary optimality as a means to improve vegetation and land-surface models. New Phytologist, 231(6), 2125–2141. https://doi.org/10.1111/nph.17558
- 45.
Henn JJ, Buzzard V, Enquist BJ, Halbritter AH, Klanderud K, Maitner BS, Michaletz ST, Pötsch C, Seltzer L, Telford RJ, Yang Y, Zhang L, & Vandvik V. (2018). Intraspecific trait variation and phenotypic plasticity mediate alpine plant species response to climate change. Frontiers in Plant Science, 9, 1548. https://doi.org/10.3389/fpls.2018.01548
- 46.
Hincke AJC, Broere T, Kürschner WM, Donders TH, & Wagner-Cremer F. (2016). Multi-year leaf-level response to sub-ambient and elevated experimental CO2 in Betula nana. PLoS ONE, 11(6), e0157400. https://doi.org/10.1371/journal.pone.0157400
- 47.
Huntingford C, Zelazowski P, Galbraith D, Mercado LM, Sitch S, Fisher R, Lomas M, Walker AP, Jones CD, Booth BBB, Malhi Y, Hemming D, Kay G, Good P, … & Cox PM. (2013). Simulated resilience of tropical rainforests to CO2-induced climate change. Nature Geoscience, 6(4), 268–273. https://doi.org/10.1038/ngeo1741
- 48.
IPCC. (2023). Climate Change 2023: Synthesis Report. In Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change (Lee H, Romero J. Eds.; 184p). IPCC. https://doi.org/10.59327/IPCC/AR6-9789291691647
- 49.
Joshi J, Stocker BD, Hofhansl F, Zhou S, Dieckmann U, & Prentice IC. (2022). Towards a unified theory of plant photosynthesis and hydraulics. Nature Plants, 8(11), 1304–1316. https://doi.org/10.1038/s41477-022-01244-5
- 50.
Kassambara A, & Mundt F. (2016). factoextra: Extract and Visualize the Results of Multivariate Data Analyses. In CRAN: Contributed Packages. CRAN. https://doi.org/10.32614/CRAN.package.factoextra
- 51.
Klein T. (2014). The variability of stomatal sensitivity to leaf water potential across tree species indicates a continuum between isohydric and anisohydric behaviours. Functional Ecology, 28(6), 1313–1320. https://doi.org/10.1111/1365-2435.12289
- 52.
Lammertsma EI, De Boer HJ, Dekker SC, Dilcher DL, Lotter AF, & Wagner-Cremer F. (2011). Global CO2 rise leads to reduced maximum stomatal conductance in Florida vegetation. Proceedings of the National Academy of Sciences of the United States of America, 108(10), 4035–4040. https://doi.org/10.1073/pnas.1100371108
- 53.
Laughlin DC. (2014). The intrinsic dimensionality of plant traits and its relevance to community assembly. Journal of Ecology, 102(1), 186–193. https://doi.org/10.1111/1365-2745.12187
- 54.
Leakey ADB. (2009). Rising atmospheric carbon dioxide concentration and the future of C4 crops for food and fuel. Proceedings of the Royal Society B: Biological Sciences 276(1666), 2333–2343. https://doi.org/10.1098/rspb.2008.1517
- 55.
Leakey ADB, Ainsworth EA, Bernacchi CJ, Rogers A, Long SP, & Ort DR. (2009). Elevated CO2 effects on plant carbon, nitrogen, and water relations: Six important lessons from FACE. Journal of Experimental Botany, 60(10), 2859–2876. https://doi.org/10.1093/jxb/erp096
- 56.
Lee TD, Barrott SH, & Reich PB. (2011). Photosynthetic responses of 13 grassland species across 11 years of free-air CO2 enrichment is modest, consistent and independent of N supply. Global Change Biology, 17(9), 2893–2904. https://doi.org/10.1111/j.1365-2486.2011.02435.x
- 57.
Lenth RV. (2017). emmeans: Estimated Marginal Means, aka Least-Squares Means. In CRAN: Contributed Packages. CRAN. https://doi.org/10.32614/CRAN.package.emmeans
- 58.
Li F, He C, Chang Z, Ma C, Yu J, Liu L, Zhang Y, & Hao L. (2023). Effects of elevated carbon dioxide on plant growth and leaf photosynthesis of annual ryegrass along a phosphorus deficiency gradient. Frontiers in Plant Science, 14. https://doi.org/10.3389/fpls.2023.1271262
- 59.
Li F, Kang S, & Zhang J. (2004). Interactive effects of elevated CO2, nitrogen and drought on leaf area, stomatal conductance, and evapotranspiration of wheat. Agricultural Water Management, 67(3), 221–233. https://doi.org/10.1016/j.agwat.2004.01.005
- 60.
Li L, McCormack ML, Ma C, Kong D, Zhang Q, Chen X, Zeng H, Niinemets Ü, & Guo D. (2015). Leaf economics and hydraulic traits are decoupled in five species-rich tropical-subtropical forests. Ecology Letters, 18(9), 899–906. https://doi.org/10.1111/ele.12466
- 61.
Long SP, Ainsworth EA, Rogers A, & Ort DR. (2004). Rising Atmospheric Carbon Dioxide: Plants FACE the future. Annual Review of Plant Biology, 55(1), 591–628. https://doi.org/10.1146/annurev.arplant.55.031903.141610
- 62.
Luo X, Keenan TF, Chen JM, Croft H, Colin Prentice I, Smith NG, Walker AP, Wang H, Wang R, Xu C, & Zhang Y. (2021). Global variation in the fraction of leaf nitrogen allocated to photosynthesis. Nature Communications, 12(1), 4866. https://doi.org/10.1038/s41467-021-25163-9
- 63.
Maire V, Gross N, Hill D, Martin R, Wirth C, Wright IJ, & Soussana JF. (2013). Disentangling Coordination among Functional Traits Using an Individual-Centred Model: Impact on Plant Performance at Intra- and Inter-Specific Levels. PLoS ONE, 8(10), e77372. https://doi.org/10.1371/journal.pone.0077372
- 64.
Maire V, Martre P, Kattge J, Gastal F, Esser G, Fontaine S, & Soussana JF. (2012). The coordination of leaf photosynthesis links C and N fluxes in C3 plant species. PLoS ONE, 7(6), e38345. https://doi.org/10.1371/journal.pone.0038345
- 65.
Maréchaux I, Bartlett MK, Sack L, Baraloto C, Engel J, Joetzjer E, & Chave J. (2015). Drought tolerance as predicted by leaf water potential at turgor loss point varies strongly across species within an Amazonian forest. Functional Ecology, 29(10), 1268–1277. https://doi.org/10.1111/1365-2435.12452
- 66.
Matthews JSA, Vialet-Chabrand SRM, & Lawson T. (2017). Diurnal variation in gas exchange: The balance between carbon fixation and water loss. Plant Physiology, 174(2), 614–623. https://doi.org/10.1104/pp.17.00152
- 67.
McElwain JC, & Steinthorsdottir M. (2017). Paleoecology, ploidy, paleoatmospheric composition, and developmental biology: A review of the multiple uses of fossil stomata. Plant Physiology 174(2), 650–664. https://doi.org/10.1104/pp.17.00204
- 68.
Mcelwain JC, Yiotis C, & Lawson T. (2016). Using modern plant trait relationships between observed and theoretical maximum stomatal conductance and vein density to examine patterns of plant macroevolution. New Phytologist, 209(1), 94–103. https://doi.org/10.1111/nph.13579
- 69.
Medlyn BE, Duursma RA, Eamus D, Ellsworth DS, Prentice IC, Barton CVM, Crous KY, De Angelis P, Freeman M, & Wingate L. (2011). Reconciling the optimal and empirical approaches to modelling stomatal conductance. Global Change Biology, 17(6), 2134–2144. https://doi.org/10.1111/j.1365-2486.2010.02375.x
- 70.
Mengoli G, Agustí-Panareda A, Boussetta S, Harrison SP, Trotta C, & Prentice IC. (2022). Ecosystem Photosynthesis in Land-Surface Models: A First-Principles Approach Incorporating Acclimation. Journal of Advances in Modeling Earth Systems, 14(1), e2021MS002767. https://doi.org/10.1029/2021MS002767
- 71.
Muir CD. (2019). Is Amphistomy an Adaptation to High Light? Optimality Models of Stomatal Traits along Light Gradients. Integrative and Comparative Biology, 59(3), 571–584. https://doi.org/10.1093/icb/icz085
- 72.
Murray M, SohWK, Yiotis C, Batke S, Parnell AC, Spicer RA, Lawson T, Caballero R, Wright IJ, Purcell C, & McElwain JC. (2019). Convergence in maximum stomatal conductance of C3 woody angiosperms in natural ecosystems across bioclimatic zones. Frontiers in Plant Science, 10, 558. https://doi.org/10.3389/fpls.2019.00558
- 73.
Murray M, Soh WK, Yiotis C, Spicer RA, Lawson T, & McElwain JC. (2020). Consistent relationship between field-measured stomatal conductance and theoretical maximum stomatal conductance in C3 woody angiosperms in four major biomes. International Journal of Plant Sciences, 181(1), 142–154. https://doi.org/10.1086/706260
- 74.
Nick Smith’s Ecophysiology Lab, McNellis R, & Keenan T. (2024). SmithEcophysLab/optimal_vcmax_R: Optimal Vcmax version 3.2.1. Zenodo. https://doi.org/10.5281/zenodo.14186765
- 75.
Nimmo V, Violle C, Entz M, Rolhauser AG, & Isaac ME. (2023). Changes in crop trait plasticity with domestication history: Management practices matter. Ecology and Evolution, 13(11), e10690. https://doi.org/10.1002/ece3.10690
- 76.
Norby RJ, Delucia EH, Gielen B, Calfapietra C, Giardina CP, King JS, Ledford J, Mccarthy HR, Moore DJP, Ceulemans R, De Angelis P, Finzi AC, Karnosky DF, Kubiske ME, … & Oren R. (2005). Forest response to elevated CO2 is conserved across a broad range of productivity. Proceedings of the National Academy of Sciences of the United States of America, 102(50), 18052–18056. https://doi.org/10.1073/pnas.0509478102
- 77.
Odé A, Smith NG, Rebel KT, & de Boer HJ. (2025). Temporal constraints on leaf-level trait plasticity for next-generation land surface models. Annals of Botany, 136(2), 263–274. https://doi.org/10.1093/aob/mcaf045
- 78.
Perkowski EA, Ezekannagha E, & Smith NG. (2025). Nitrogen demand, availability, and acquisition strategy control plant responses to elevated CO2. Journal of Experimental Botany, 76(10), 2908–2923. https://doi.org/10.1093/jxb/eraf118
- 79.
Poorter H, Knopf O, Wright IJ, Temme AA, Hogewoning SW, Graf A, Cernusak LA, & Pons TL. (2022). A meta-analysis of responses of C3 plants to atmospheric CO2: dose–response curves for 85 traits ranging from the molecular to the whole-plant level. New Phytologist 233(4), 1560–1596. https://doi.org/10.1111/nph.17802
- 80.
Poorter H, Niinemets Ü, Poorter L, Wright IJ, & Villar R. (2009). Causes and consequences of variation in leaf mass per area (LMA): A meta-analysis. New Phytologist 182(3), 565–588. https://doi.org/10.1111/j.1469-8137.2009.02830.x
- 81.
Poorter H, Pons TL, & Reichgelt T. (2025). Stomatal Density and Index Are More Responsive to Light Intensity than to [CO2]: A Meta-Analysis and Implications for Paleo-CO2 Reconstruction. Plant Ecophysiology, 1, 1. https://doi.org/10.53941/plantecophys.2025.100001
- 82.
Poorter H, Van Berkel Y, Baxter R, Den Hertog J, Dijkstra P, Gifford RM, Griffin KL, Roumet C, Roy J, & Wong SC. (1997). The effect of elevated CO2 on the chemical composition and construction costs of leaves of 27 C3 species. Plant, Cell and Environment, 20(4), 472–482. https://doi.org/10.1046/j.1365-3040.1997.d01-84.x
- 83.
Prentice IC, & Cowling SA. (2013). Dynamic Global Vegetation Models. In Encyclopedia of Biodiversity (2nd ed., pp. 670–689). Elsevier Inc. https://doi.org/10.1016/B978-0-12-384719-5.00412-3
- 84.
Prentice IC, Dong N, Gleason SM, Maire V, & Wright IJ. (2014). Balancing the costs of carbon gain and water transport: Testing a new theoretical framework for plant functional ecology. Ecology Letters, 17(1), 82–91. https://doi.org/10.1111/ele.12211
- 85.
Prentice IC, Liang X, Medlyn BE, & Wang YP. (2015). Reliable, robust and realistic: The three R’s of next-generation land-surface modelling. Atmospheric Chemistry and Physics, 15(10), 5987–6005. https://doi.org/10.5194/acp-15-5987-2015
- 86.
Pritchard SG, Rogers HH, Prior SA, & Peterson CM. (1999). Elevated CO2 and plant structure: A review. Global Change Biology, 5(7), 807–837. https://doi.org/10.1046/j.1365-2486.1999.00268.x
- 87.
Purcell C, Batke SP, Yiotis C, Caballero R, Soh WK, Murray M, & McElwain JC. (2018). Increasing stomatal conductance in response to rising atmospheric CO2. Annals of Botany, 121(6), 1137–1149. https://doi.org/10.1093/aob/mcx208
- 88.
Reich PB. (2014). The world-wide “fast-slow” plant economics spectrum: A traits manifesto. Journal of Ecology, 102(2), 275–301. https://doi.org/10.1111/1365-2745.12211
- 89.
Sack L, Scoffoni C, John GP, Poorter H, Mason CM, Mendez-Alonzo R, & Donovan LA. (2013). How do leaf veins influence the worldwide leaf economic spectrum? Review and synthesis. Journal of Experimental Botany, 64(13), 4053–4080. https://doi.org/10.1093/jxb/ert316
- 90.
Sage RF, Sharkey TD, & Seemann JR. (1989). Acclimation of Photosynthesis to Elevated CO2 in Five C3 Species1. Plant Physiology, 89(2), 590–596. https://academic.oup.com/plphys/article/89/2/590/6083463
- 91.
Salim M, Chen Y, Ye H, Nguyen HT, Solaiman ZM, & Siddique KHM. (2022). Screening of soybean genotypes based on root morphology and shoot traits using the semi-hydroponic phenotyping platform and rhizobox technique. Agronomy, 12(1), 56. https://doi.org/10.3390/agronomy12010056
- 92.
Sanchez-Martinez P, Ackerly DD, Martínez-Vilalta J, Mencuccini M, Dexter KG, & Dawson TE. (2024). A framework to study and predict functional trait syndromes using phylogenetic and environmental data. Methods in Ecology and Evolution, 15(4), 666–681. https://doi.org/10.1111/2041-210X.14304
- 93.
Sanchez-Martinez P, Martínez-Vilalta J, Dexter KG, Segovia RA, & Mencuccini M. (2020). Adaptation and coordinated evolution of plant hydraulic traits. Ecology Letters, 23(11), 1599–1610. https://doi.org/10.1111/ele.13584
- 94.
Schimel D, Stephens BB, & Fisher JB. (2015). Effect of increasing CO2 on the terrestrial carbon cycle. Proceedings of the National Academy of Sciences of the United States of America, 112(2), 436–441. https://doi.org/10.1073/pnas.1407302112
- 95.
Schneider CA, Rasband WS, & Eliceiri KW. (2012). NIH Image to ImageJ: 25 years of image analysis. Nature Methods, 9(7), 671–675. https://doi.org/10.1038/nmeth.2089
- 96.
Shao J, Li G, Li Y, & Zhou X. (2022). Intraspecific responses of plant productivity and crop yield to experimental warming: A global synthesis. Science of the Total Environment, 840, 156685. https://doi.org/10.1016/j.scitotenv.2022.156685
- 97.
Siefert A, Violle C, Chalmandrier L, Albert CH, Taudiere A, Fajardo A, Aarssen LW, Baraloto C, Carlucci MB, Cianciaruso MV, de L Dantas V, de Bello F, Duarte LDS, Fonseca CR, … & Wardle DA. (2015). A global meta-analysis of the relative extent of intraspecific trait variation in plant communities. Ecology Letters, 18(12), 1406–1419. https://doi.org/10.1111/ele.12508
- 98.
Smith NG, & Dukes JS. (2013). Plant respiration and photosynthesis in global-scale models: Incorporating acclimation to temperature and CO2. Global Change Biology 19(1), 45–63. https://doi.org/10.1111/j.1365-2486.2012.02797.x
- 99.
Smith NG, & Keenan TF. (2020). Mechanisms underlying leaf photosynthetic acclimation to warming and elevated CO2 as inferred from least-cost optimality theory. Global Change Biology, 26(9), 5202–5216. https://doi.org/10.1111/gcb.15212
- 100.
Smith NG, Keenan TF, Colin Prentice I, Wang H, Wright IJ, Niinemets Ü, Crous KY, Domingues TF, Guerrieri R, Yoko Ishida F, Kattge J, Kruger EL, Maire V, Rogers A, … & Zhou SX. (2019). Global photosynthetic capacity is optimized to the environment. Ecology Letters, 22(3), 506–517. https://doi.org/10.1111/ele.13210
- 101.
Sonawane BV, Sharwood RE, Whitney S, & Ghannoum O. (2018). Shade compromises the photosynthetic efficiency of NADP-ME less than that of PEP-CK and NAD-ME C4 grasses. Journal of Experimental Botany, 69(12), 3053–3068. https://doi.org/10.1093/jxb/ery129
- 102.
Stocker BD, Wang H, Smith NG, Harrison SP, Keenan TF, Sandoval D, Davis T, & Prentice IC. (2020). P-model v1.0: An optimality-based light use efficiency model for simulating ecosystem gross primary production. Geoscientific Model Development, 13(3), 1545–1581. https://doi.org/10.5194/gmd-13-1545-2020
- 103.
Taylor SH, Hulme SP, Rees M, Ripley BS, Ian Woodward F, & Osborne CP. (2010). Ecophysiological traits in C3 and C4 grasses: A phylogenetically controlled screening experiment. New Phytologist, 185(3), 780–791. https://doi.org/10.1111/j.1469-8137.2009.03102.x
- 104.
Tricker PJ, Trewin H, Kull O, Clarkson GJJ, Eensalu E, Tallis MJ, Colella A, Doncaster CP, Sabatti M, & Taylor G. (2005). Stomatal conductance and not stomatal density determines the long-term reduction in leaf transpiration of poplar in elevated CO2. Oecologia, 143(4), 652–660. https://doi.org/10.1007/s00442-005-0025-4
- 105.
Von Caemmerer S. (2021). Updating the steady-state model of C4 photosynthesis. Journal of Experimental Botany 72(17), 6003–6017. Oxford University Press. https://doi.org/10.1093/jxb/erab266
- 106.
Wang H, Prentice IC, Keenan TF, Davis TW, Wright IJ, Cornwell WK, Evans BJ, & Peng C. (2017). Towards a universal model for carbon dioxide uptake by plants/704/47/704/158/851 letter. Nature Plants, 3(9), 734–741. https://doi.org/10.1038/s41477-017-0006-8
- 107.
Wang H, Prentice IC, Wright IJ, Warton DI, Qiao S, Xu X, Zhou J, Kikuzawa K, & Stenseth NChr. (2023). Leaf economics fundamentals explained by optimality principles. Science Advances, 9(3). https://doi.org/10.1126/sciadv.add5667
- 108.
Wang S, Zhang Y, Ju W, Chen JM, Ciais P, Cescatti A, Sardans J, Janssens IA, Wu M, Berry JA, Campbell E, Fernández-Martínez M, Alkama R, Sitch S, … & Peñuelas J. (2020). Recent global decline of CO2 fertilization effects on vegetation photosynthesis. Science, 370(6522), 1295–1300. https://doi.org/10.1126/science.abb7772
- 109.
Wei Z, Abdelhakim LOA, Fang L, Peng X, Liu J, & Liu F. (2022). Elevated CO2 effect on the response of stomatal control and water use efficiency in amaranth and maize plants to progressive drought stress. Agricultural Water Management, 266, 107609. https://doi.org/10.1016/j.agwat.2022.107609
- 110.
Wong SC, Cowan IR, & Farquhar GD. (1979). Stomatal conductance correlates with photosynthetic capacity. Nature, 282(5737), 424–426. https://doi.org/10.1038/282424a0
- 111.
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F, Cavender-Bares J, Chapin T, Cornelissen JHC, Diemer M, Flexas J, Garnier E, Groom PK, Gulias J, … & Villar R. (2004). The worldwide leaf economics spectrum. Nature, 428(6985), 821–827. https://doi.org/10.1038/nature02403
- 112.
Xie J, Wang Z, & Li Y. (2022). Stomatal opening ratio mediates trait coordinating network adaptation to environmental gradients. New Phytologist, 235(3), 907–922. https://doi.org/10.1111/nph.18189
- 113.
Xiong D, Douthe C, & Flexas J. (2018). Differential coordination of stomatal conductance, mesophyll conductance, and leaf hydraulic conductance in response to changing light across species. Plant Cell and Environment, 41(2), 436–450. https://doi.org/10.1111/pce.13111
- 114.
Xu H, Wang H, Prentice IC, Harrison SP, & Wright IJ. (2021a). Coordination of plant hydraulic and photosynthetic traits: confronting optimality theory with field measurements. New Phytologist, 232(3), 1286–1296. https://doi.org/10.1111/nph.17656
- 115.
Xu H, Wang H, Prentice IC, Harrison SP, & Wright IJ. (2021b). Plant hydraulics coordinated with photosynthetic traits and climate. bioRxiv, https://doi.org/10.1101/2021.03.02.433324
- 116.
Xu Z, Jiang Y, Jia B, & Zhou G. (2016). Elevated-CO2 response of stomata and its dependence on environmental factors. Frontiers in Plant Science 7, 657. https://doi.org/10.3389/fpls.2016.00657
- 117.
Zheng Y, Li F, Hao L, Yu J, Guo L, Zhou H, Ma C, Zhang X, & Xu M. (2019). Elevated CO2 concentration induces photosynthetic down-regulation with changes in leaf structure, non-structural carbohydrates and nitrogen content of soybean. BMC Plant Biology, 19(1), 255. https://doi.org/10.1186/s12870-019-1788-9
- 118.
Zhou J, Cieraad E, & van Bodegom PM. (2022). Global analysis of trait–trait relationships within and between species. New Phytologist, 233(4), 1643–1656. https://doi.org/10.1111/nph.17879

This work is licensed under a Creative Commons Attribution 4.0 International License.


