- Open Access
- Editorial
Epigenetic Mechanisms of Psychedelics in Addiction: Emerging Evidence and Therapeutic Potential
- Antonio Inserra 1,2,3,*,
- Francesca Zoratto 1,
- Mauro Pettorruso 2,4,5,
- Giovanni Martinotti 2,4,5,6
Author Information
Received: 10 Mar 2026 | Accepted: 30 Mar 2026 | Published: 17 Apr 2026
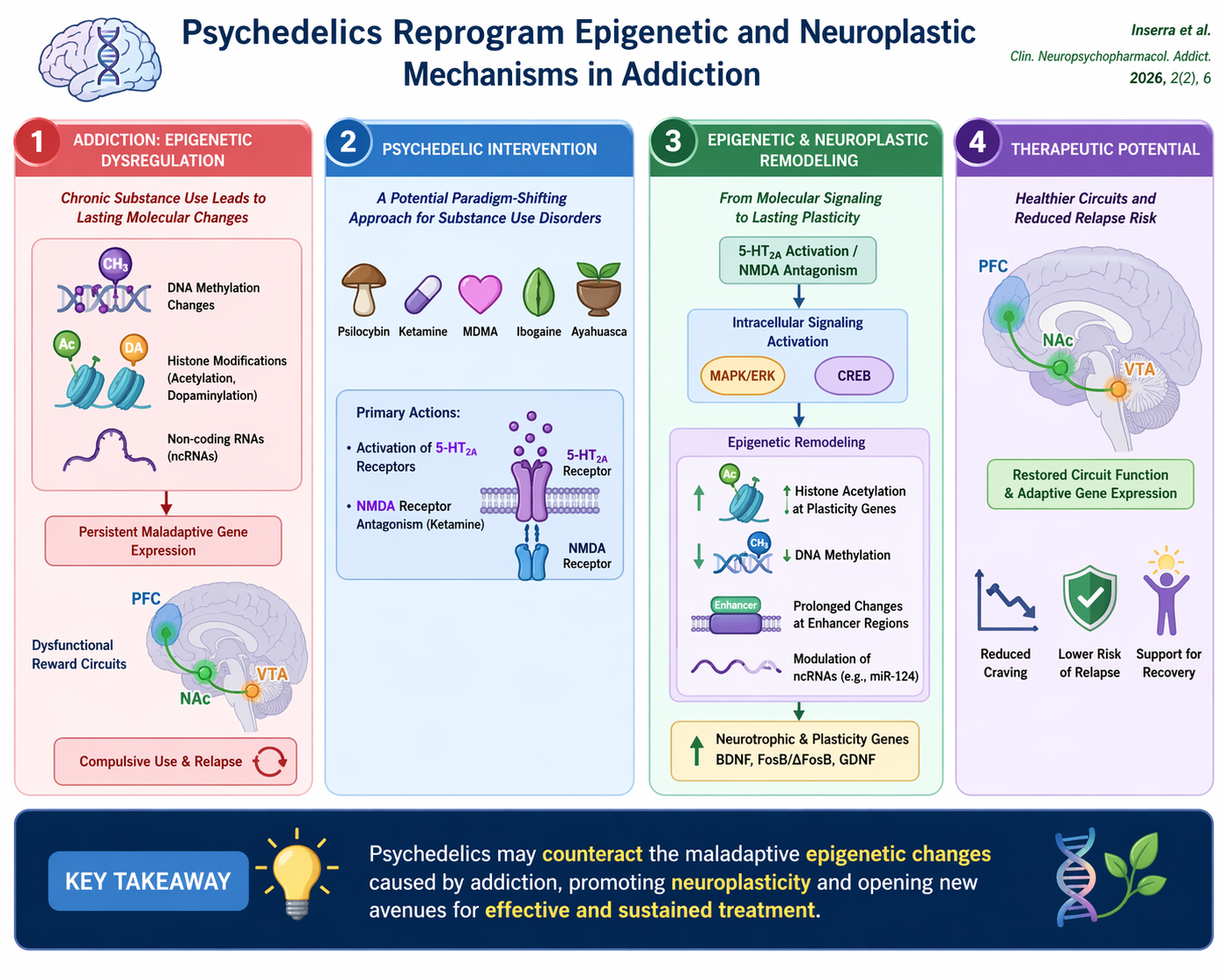
Graphical Abstract
References
- 1.
Hamilton, P.J.; Nestler, E.J. Epigenetics and addiction. Curr. Opin. Neurobiol. 2019, 59, 128–136. https://doi.org/10.1016/j.conb.2019.05.005.
- 2.
Koijam, A.S.; Singh, K.D.; Nameirakpam, B.S.; et al. Drug addiction and treatment: An epigenetic perspective. Biomed. Pharmacother. 2024, 170, 115951.
- 3.
Lepack, A.E.; Werner, C.T.; Stewart, A.F.; et al. Dopaminylation of histone H3 in ventral tegmental area regulates cocaine seeking. Science 2020, 368, 197–201. https://doi.org/10.1126/science.aaw8806.
- 4.
Köck, P.; Froelich, K.; Walter, M.; et al. A systematic literature review of clinical trials and therapeutic applications of ibogaine. J. Subst. Abuse Treat. 2022, 138, 108717. https://doi.org/10.1016/j.jsat.2021.108717.
- 5.
Heinsbroek, J.A.; Giannotti, G.; Bonilla, J.; et al. Tabernanthalog reduces motivation for heroin and alcohol in a polydrug use model. Psychedelic Med. 2023, 1, 111–119.
- 6.
Cameron, L.P.; Tombari, R.J.; Lu, J.; et al. A non-hallucinogenic psychedelic analogue with therapeutic potential. Nature 2021, 589, 474–479.
- 7.
Havel, V.; Kruegel, A.C.; Bechand, B.; et al. Oxa-Iboga alkaloids lack cardiac risk and disrupt opioid use in animal models. Nat. Commun. 2024, 15, 8118. https://doi.org/10.1038/s41467-024-51856-y.
- 8.
Bogenschutz, M.P.; Ross, S.; Bhatt, S.; et al. Percentage of Heavy Drinking Days Following Psilocybin-Assisted Psychotherapy vs Placebo in the Treatment of Adult Patients With Alcohol Use Disorder: A Randomized Clinical Trial. JAMA Psychiatry 2022, 79, 953–962. https://doi.org/10.1001/jamapsychiatry.2022.2096.
- 9.
Johnson, M.W.; Garcia-Romeu, A.; Cosimano, M.P.; et al. Pilot study of the 5-HT2AR agonist psilocybin in the treatment of tobacco addiction. J. Psychopharmacol. 2014, 28, 983–992.
- 10.
Johnson, M.W.; Garcia-Romeu, A.; Griffiths, R.R. Long-term follow-up of psilocybin-facilitated smoking cessation. Am. J. Drug Alcohol Abus. 2017, 43, 55–60.
- 11.
Luquiens, A.; Belahda, D.; Graux, C.; et al. Psilocybin in alcohol use disorder and comorbid depressive symptoms: Results from a feasibility randomized clinical trial. Addiction 2025. https://doi.org/10.1111/add.70152.
- 12.
Grabski, M.; McAndrew, A.; Lawn, W.; et al. Adjunctive Ketamine With Relapse Prevention-Based Psychological Therapy in the Treatment of Alcohol Use Disorder. Am. J. Psychiatry 2022, 179, 152–162. https://doi.org/10.1176/appi.ajp.2021.21030277.
- 13.
Dakwar, E.; Nunes Edward, V.; Hart Carl, L.; et al. A Single Ketamine Infusion Combined With Mindfulness-Based Behavioral Modification to Treat Cocaine Dependence: A Randomized Clinical Trial. Am. J. Psychiatry 2019, 176, 923–930. https://doi.org/10.1176/appi.ajp.2019.18101123.
- 14.
Janssen-Aguilar, R.; Meshkat, S.; Demchenko, I.; et al. Role of ketamine in the treatment of substance use disorders: A systematic review. J. Subst. Use Addict. Treat. 2025, 175, 209705. https://doi.org/10.1016/j.josat.2025.209705.
- 15.
Martinotti, G.; Chiappini, S.; Pettorruso, M.; et al. Therapeutic Potentials of Ketamine and Esketamine in Obsessive–Compulsive Disorder (OCD), Substance Use Disorders (SUD) and Eating Disorders (ED): A Review of the Current Literature. Brain Sci. 2021, 11, 856. https://doi.org/10.3390/brainsci11070856.
- 16.
Sessa, B.; Higbed, L.; O’Brien, S.; et al. First study of safety and tolerability of 3,4-methylenedioxymethamphetamine-assisted psychotherapy in patients with alcohol use disorder. J. Psychopharmacol. 2021, 35, 375–383. https://doi.org/10.1177/0269881121991792.
- 17.
Thurgur, H.; Sessa, B.; Higbed, L.; et al. MDMA-assisted psychotherapy for AUD: Bayesian analysis of WHO drinking risk level and exploratory analysis of drinking behavior and psychosocial functioning at 3 months follow-up. Alcohol. Alcohol. 2025, 60, agaf031. https://doi.org/10.1093/alcalc/agaf031.
- 18.
Richard, J.; Garcia-Romeu, A. Psychedelics in the Treatment of Substance Use Disorders and Addictive Behaviors: A Scoping Review. Curr. Addict. Rep. 2025, 12, 15. https://doi.org/10.1007/s40429-025-00629-8.
- 19.
Agin-Liebes, G.; Haas, T.F.; Lancelotta, R.; et al. Naturalistic use of mescaline is associated with self-reported psychiatric improvements and enduring positive life changes. ACS Pharmacol. Transl. Sci. 2021, 4, 543–552.
- 20.
Uthaug, M.V.; Davis, A.K.; Haas, T.F.; et al. The epidemiology of mescaline use: Pattern of use, motivations for consumption, and perceived consequences, benefits, and acute and enduring subjective effects. J. Psychopharmacol. 2022, 36, 309–320.
- 21.
Blum, K.; Futterman, S.F.L.; Pascarosa, P. Peyote, a potential ethnopharmacologic agent for alcoholism and other drug dependencies: Possible biochemical rationale. Clin. Toxicol. 1977, 11, 459–472.
- 22.
Ruffell, S.G.D.; Netzband, N.; Tsang, W.; et al. Ceremonial Ayahuasca in Amazonian Retreats—Mental Health and Epigenetic Outcomes From a Six-Month Naturalistic Study. Front. Psychiatry 2021, 12, 687615. https://doi.org/10.3389/fpsyt.2021.687615.
- 23.
Inserra, A. Hypothesis: The Psychedelic Ayahuasca Heals Traumatic Memories via a Sigma 1 Receptor-Mediated Epigenetic-Mnemonic Process. Front. Pharmacol. 2018, 9, 330. https://doi.org/10.3389/fphar.2018.00330.
- 24.
Inserra, A.; Campanale, A.; Cheishvili, D.; et al. Modulation of DNA methylation and protein expression in the prefrontal cortex by repeated administration of D-lysergic acid diethylamide (LSD): Impact on neurotropic, neurotrophic, and neuroplasticity signaling. Prog. Neuropsychopharmacol. Biol. Psychiatry 2022, 119, 110594. https://doi.org/10.1016/j.pnpbp.2022.110594.
- 25.
de la Fuente Revenga, M.; Zhu, B.; Guevara, C.A.; et al. Prolonged epigenomic and synaptic plasticity alterations following single exposure to a psychedelic in mice. Cell Rep. 2021, 37, 109836. https://doi.org/10.1016/j.celrep.2021.109836.
- 26.
Ly, C.; Greb, A.C.; Cameron, L.P.; et al. Psychedelics promote structural and functional neural plasticity. Cell Rep. 2018, 23, 3170–3182.
- 27.
De Gregorio, D.; Inserra, A.; Enns, J.P.; et al. Repeated lysergic acid diethylamide (LSD) reverses stress-induced anxiety-like behavior, cortical synaptogenesis deficits and serotonergic neurotransmission decline. Neuropsychopharmacology 2022, 47, 1188–1198. https://doi.org/10.1038/s41386-022-01301-9.
- 28.
De Gregorio, D.; Popic, J.; Enns Justine, P.; et al. Lysergic acid diethylamide (LSD) promotes social behavior through mTORC1 in the excitatory neurotransmission. Proc. Natl. Acad. Sci. USA 2021, 118, e2020705118. https://doi.org/10.1073/pnas.2020705118.
- 29.
Richardson, B.; Inserra, A.; Pileggi, M.; et al. Differential effects of psilocybin and lisuride on serotonin and dopamine neuronal activity and behavior. Prog. Neuropsychopharmacol. Biol. Psychiatry 2025, 142, 111522. https://doi.org/10.1016/j.pnpbp.2025.111522.
- 30.
Jaster, A.M.; Hadlock, T.M.; Buzzi, B.; et al. Sex-specific role of the 5-HT2A receptor in psilocybin-induced extinction of opioid reward. Nat. Commun. 2025, 16, 10206. https://doi.org/10.1038/s41467-025-64887-w.
- 31.
Siegel, J.S.; Liston, C.; Nicol, G.E.; et al. The science of psychedelic medicine. Nat. Med. 2026, 32, 449–462. https://doi.org/10.1038/s41591-025-04194-5.
- 32.
Inserra, A.; Campanale, A.; Rezai, T.; et al. Epigenetic mechanisms of rapid-acting antidepressants. Transl. Psychiatry 2024, 14, 359. https://doi.org/10.1038/s41398-024-03055-y.
- 33.
de Camargo, R.W.; Joaquim, L.; Machado, R.S.; et al. Ayahuasca Pretreatment Prevents Sepsis-Induced Anxiety-Like Behavior, Neuroinflammation, and Oxidative Stress, and Increases Brain-Derived Neurotrophic Factor. Mol. Neurobiol. 2025, 62, 5695–5719. https://doi.org/10.1007/s12035-024-04597-4.
- 34.
Inserra, A.; De Gregorio, D.; Gobbi, G. Psychedelics in Psychiatry: Neuroplastic, Immunomodulatory, and Neurotransmitter Mechanisms. Pharmacol. Rev. 2021, 73, 202–277. https://doi.org/10.1124/pharmrev.120.000056.
- 35.
Floris, G.; Dabrowski, K.R.; Zanda, M.T.; et al. Psilocybin reduces heroin seeking behavior and modulates inflammatory gene expression in the nucleus accumbens and prefrontal cortex of male rats. Mol. Psychiatry 2025, 30, 1801–1816. https://doi.org/10.1038/s41380-024-02788-y.
- 36.
Leccisotti, I.; Moretti, M.C.; Altamura, M.; et al. The epigenetic mechanisms of ketamine in the treatment of depression: A systematic review. Epigenomics 2025, 17, 1641–1658. https://doi.org/10.1080/17501911.2025.2583892.
- 37.
Carnicella, S.; Kharazia, V.; Jeanblanc, J.; et al. GDNF is a fast-acting potent inhibitor of alcohol consumption and relapse. Proc. Natl. Acad. Sci. USA 2008, 105, 8114–8119. https://doi.org/10.1073/pnas.0711755105.
- 38.
Marton, S.; González, B.; Rodríguez, S.; et al. Ibogaine Modifies GDNF, BDNF and NGF Expression in Brain Regions Involved in Mesocorticolimbic and Nigral Dopaminergic Circuits. ChemRxiv 2018. https://doi.org/10.26434/chemrxiv.7261559.v1.
- 39.
Nardou, R.; Sawyer, E.; Song, Y.J.; et al. Psychedelics reopen the social reward learning critical period. Nature 2023, 618, 790–798. https://doi.org/10.1038/s41586-023-06204-3.
- 40.
Meshkat, S.; Malik, G.; Zeifman, R.J.; et al. Efficacy and safety of psilocybin for the treatment of substance use disorders: A systematic review. Neurosci. Biobehav. Rev. 2025, 173, 106163. https://doi.org/10.1016/j.neubiorev.2025.106163.
- 41.
Rathore, B.S.; Singh, S.; Gupta, M.; et al. Safety and efficacy of ketamine for the treatment of patients with alcohol use disorder: A systematic review. Am. J. Drug Alcohol Abus. 2025, 51, 563–576.

This work is licensed under a Creative Commons Attribution 4.0 International License.


