Per- and polyfluoroalkyl substances (PFAS) are globally distributed anthropogenic contaminants of concern due to their environmental persistence, bioaccumulation potential, and adverse health effects. Despite increasing recognition of PFAS exposure in marine wildlife, data for penguins remain limited, in part due to challenges associated with traditional sampling methods. We present a proof-of-concept application of silicone passive samplers (SPS) as a minimally invasive approach to characterize contact-based environmental PFAS exposure in penguins. SPS bands (n = 55) were deployed (2–9 days) on Magellanic penguins (Spheniscus magellanicus) at two colonies along the Patagonian coast of Argentina across three consecutive breeding seasons. Forty PFAS were targeted and quantified using liquid chromatography-tandem mass spectrometry (LC-MS/MS). PFAS were detected on the vast majority (90.7%) of SPS bands, with a mean ΣPFAS concentration of 2.26 ± 2.46 ng/g (maximum = 16.23 ng/g). Five legacy (PFPeA, PFHxA, PFHxS, PFHpA, and PFOS) and four emerging compounds (HFPO-DA, 6:2 FTS, NMeFOSAA, and NEtFOSAA) were identified. The frequency and/or concentration of PFPeA, PFHxA, PFHpA, HFPO-DA, and 6:2 FTS varied significantly among colony-season cohorts, while PFHxA, 6:2 FTS, and NMeFOSAA varied significantly with the duration of the deployment period. SPS bands provide a minimally invasive, repeatable, and practical approach for comparing relative external exposure profiles of penguins, and they could be adapted for other sensitive species to improve our understanding of PFAS and contaminant exposure in wildlife. Future studies should pair SPS deployments with biological matrices (e.g., blood) to determine how SPS-derived profiles relate to internal biological burdens and to validate the interpretation of SPS measurements.
- Open Access
- Article
Penguins as Sentinel Species for Monitoring Per- and Polyfluoroalkyl Substances (PFAS): Evaluation of Silicone Passive Samplers as a Non-Invasive Tool
- Paige C. Montgomery 1,
- Ralph Eric Thijl Vanstreels 2,*,
- Luciana Gallo 3,4,
- Gabriela S. Blanco 5,
- Flavio R. Quintana 3,
- Marcela M. Uhart 2,
- Katarzyna Kordas 6,
- Diana S. Aga 1,7,*
Author Information
Received: 11 Nov 2025 | Revised: 04 Mar 2026 | Accepted: 11 Mar 2026 | Published: 23 Mar 2026
Abstract
Graphical Abstract
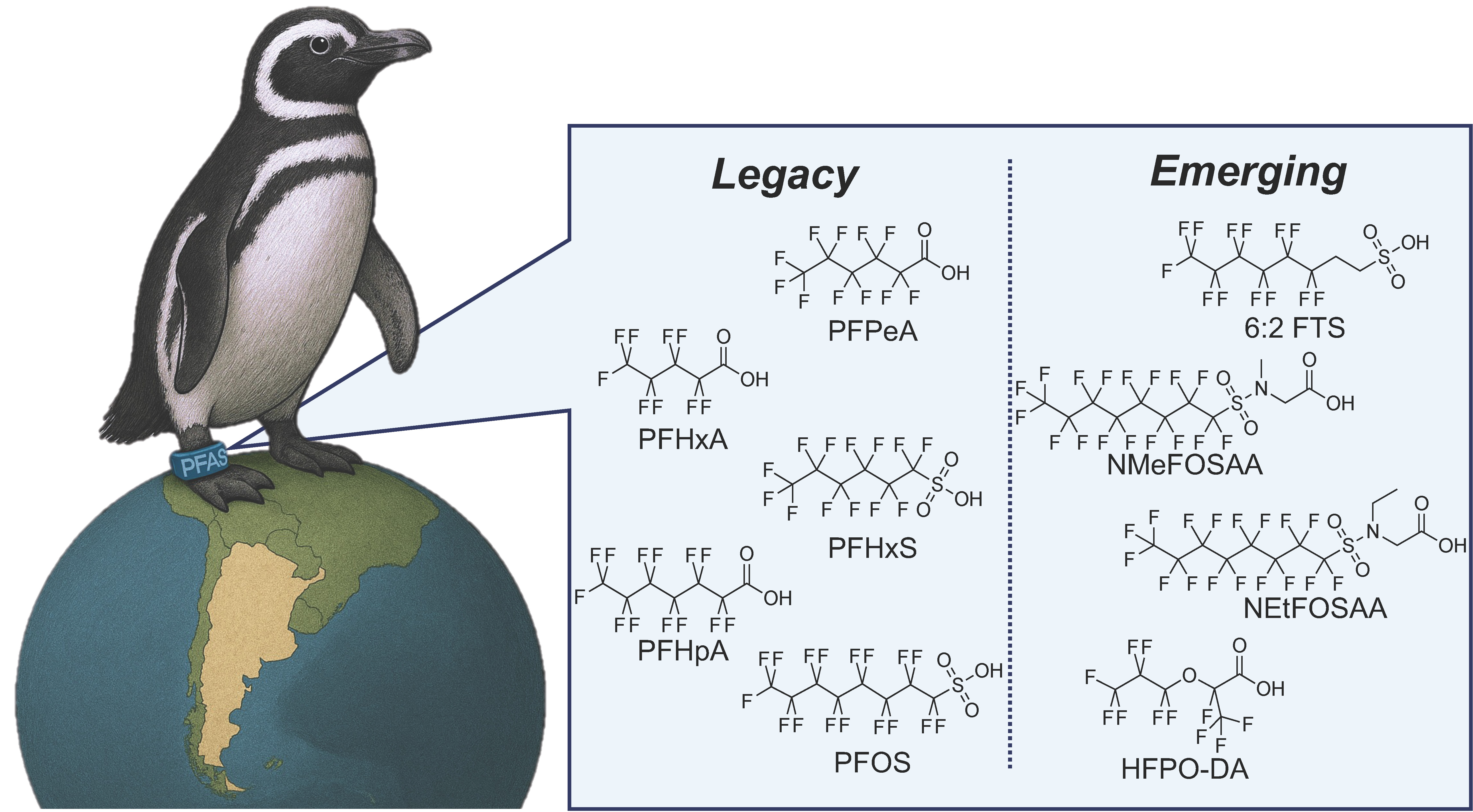
Keywords
forever chemicals | marine pollution | seabird | silicone bands | threatened species | wildlife health | biomonitoring
References
- 1.
Rhind, S.M. Anthropogenic Pollutants: A Threat to Ecosystem Sustainability? Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 3391–3401. https://doi.org/10.1098/rstb.2009.0122.
- 2.
Maneesh, T.P. Contaminant Types and Nature of Sources in General Aspects. In Contaminated Land and Water; Alshemmari, H., Hashmi, M.Z., Kavil, Y.N., Eds.; Springer: Cham, Switzerland, 2024; pp. 35–43. https://doi.org/10.1007/978-3-031-65129-8_3.
- 3.
Evich, M.G.; Davis, M.J.B.; McCord, J.P.; et al. Per- and Polyfluoroalkyl Substances in the Environment. Science 2022, 375, eabg9065. https://doi.org/10.1126/science.abg9065.
- 4.
Brase, R.A.; Mullin, E.J.; Spink, D.C. Legacy and Emerging Per- and Polyfluoroalkyl Substances: Analytical Techniques, Environmental Fate, and Health Effects. Int. J. Mol. Sci. 2021, 22, 995. https://doi.org/10.3390/ijms22030995.
- 5.
Zhang, Y.; Zhou, Y.; Dong, R.; et al. Emerging and Legacy Per- and Polyfluoroalkyl Substances (PFAS) in Fluorochemical Wastewater along Full-Scale Treatment Processes: Source, Fate, and Ecological Risk. J. Hazard. Mater. 2024, 465, 133270. https://doi.org/10.1016/j.jhazmat.2023.133270.
- 6.
Kurwadkar, S.; Dane, J.; Kanel, S.R.; et al. Per- and Polyfluoroalkyl Substances in Water and Wastewater: A Critical Review of Their Global Occurrence and Distribution. Sci. Total Environ. 2022, 809, 151003. https://doi.org/10.1016/j.scitotenv.2021.151003.
- 7.
Greaves, A.K.; Letcher, R.J.; Sonne, C.; et al. Brain Region Distribution and Patterns of Bioaccumulative Perfluoroalkyl Carboxylates and Sulfonates in East Greenland Polar Bears (Ursus maritimus). Environ. Toxicol. Chem. 2013, 32, 713–722. https://doi.org/10.1002/etc.2107.
- 8.
Trilla-Prieto, N.; Dachs, J.; Iriarte, J.; et al. Accumulation of Perfluoroalkyl Acids as Forever Chemicals in Antarctic Waters. Commun. Earth Environ. 2025, 6, 545. https://doi.org/10.1038/s43247-025-02535-3.
- 9.
Fenton, S.E.; Ducatman, A.; Boobis, A.; et al. Per- and Polyfluoroalkyl Substance Toxicity and Human Health Review: Current State of Knowledge and Strategies for Informing Future Research. Environ. Toxicol. Chem. 2020, 40, 606–630. https://doi.org/10.1002/etc.4890.
- 10.
Panieri, E.; Baralic, K.; Djukic-Cosic, D.; et al. PFAS Molecules: A Major Concern for the Human Health and the Environment. Toxics 2022, 10, 44. https://doi.org/10.3390/toxics10020044.
- 11.
Sebastiano, M.; Angelier, F.; Blevin, P.; et al. Exposure to PFAS Is Associated with Telomere Length Dynamics and Demographic Responses of an Arctic Top Predator. Environ. Sci. Technol. 2020, 54, 10217–10226. https://doi.org/10.1021/acs.est.0c03099.
- 12.
Androulakakis, A.; Alygizakis, N.; Gkotsis, G.; et al. Determination of 56 Per- and Polyfluoroalkyl Substances in Top Predators and Their Prey from Northern Europe by LC-MS/MS. Chemosphere 2022, 287, 131775. https://doi.org/10.1016/j.chemosphere.2021.131775.
- 13.
Casas, G.; Iriarte, J.; D’Agostino, L.A.; et al. Inputs, Amplification and Sinks of Perfluoroalkyl Substances at Coastal Antarctica. Environ. Pollut. 2023, 338, 122608. https://doi.org/10.1016/j.envpol.2023.122608.
- 14.
Bailes, I.R.; Phillips, R.A.; Barber, J.L.; et al. Per- and Polyfluoroalkyl Substances (PFAS) in Sub-Antarctic Seabirds: Insights into Long-Range Transport and Bioaccumulation of Legacy and Replacement Chemicals. ACS Environ. Au 2025, 5, 603–615. https://doi.org/10.1021/acsenvironau.5c00102.
- 15.
Gimeno, M.; Rossell, L.; Julia, L.; et al. Assessing Mercury Contamination in Southern Hemisphere Marine Ecosystems: The Role of Penguins as Effective Bioindicators. Environ. Pollut. 2024, 343, 123159. https://doi.org/10.1016/j.envpol.2023.123159.
- 16.
Velarde, E.; Anderson, D.W.; Ezcurra, E. Seabird Clues to Ecosystem Health. Science 2019, 365, 116–117. https://doi.org/10.1126/science.aaw9999.
- 17.
International Union for Conservation of Nature and Natural Resources. The IUCN Red List of Threatened Species. Available online: https://www.iucnredlist.org/ (accessed on 7 October 2025).
- 18.
Ropert-Coudert, Y.; Chiaradia, A.; Ainley, D.; et al. Happy Feet in a Hostile World? The Future of Penguins Depends on Proactive Management of Current and Expected Threats. Front. Mar. Sci. 2019, 6, 248.
- 19.
Dreyer, S.; Marcu, D.; Keyser, S.; et al. Factors in the Decline of the African Penguin: Are Contaminants of Emerging Concern (CECs) a Potential New Age Stressor? Mar. Pollut. Bull. 2024, 206, 116688. https://doi.org/10.1016/j.marpolbul.2024.116688.
- 20.
Wells, M.R.; Coggan, T.L.; Stevenson, G.; et al. Per- and Polyfluoroalkyl Substances (PFAS) in Little Penguins and Associations with Urbanization and Health Parameters. Sci. Total Environ. 2024, 912, 169084. https://doi.org/10.1016/j.scitotenv.2023.169084.
- 21.
Mollier, M.; Bustamante, P.; Martinez-Alvarez, I.; et al. Blood Kinetics of Lipophilic and Proteinophilic Pollutants during Two Types of Long-Term Fast in King Penguins. Environ. Sci. Technol. 2024, 58, 6138–6148. https://doi.org/10.1021/acs.est.3c10822.
- 22.
Carravieri, A.; Bustamante, P.; Labadie, P.; et al. Trace Elements and Persistent Organic Pollutants in Chicks of 13 Seabird Species from Antarctica to the Subtropics. Environ. Int. 2020, 134, 105225. https://doi.org/10.1016/j.envint.2019.105225.
- 23.
Wild, S.; Eulaers, I.; Covaci, A.; et al. South Polar Skua (Catharacta maccormicki) as Biovectors for Long-Range Transport of Persistent Organic Pollutants to Antarctica. Environ. Pollut. 2022, 292, 118358. https://doi.org/10.1016/j.envpol.2021.118358.
- 24.
Padilha, J.; De Carvalho, G.O.; Willems, T.; et al. Perfluoroalkylated Compounds in the Eggs and Feathers of Resident and Migratory Seabirds from the Antarctic Peninsula. Environ. Res. 2022, 214, 114157. https://doi.org/10.1016/j.envres.2022.114157.
- 25.
Pala, N.; Vorkamp, K.; Bossi, R.; et al. Chemical Threats for the Sentinel Pygoscelis adeliae from the Ross Sea (Antarctica): Occurrence and Levels of POPs, PFAS and Mercury within the Largest Marine Protected Area Worldwide. Sci. Total Environ. 2024, 947, 174562. https://doi.org/10.1016/j.scitotenv.2024.174562.
- 26.
Bouwman, H.; Govender, D.; Underhill, L.; et al. Chlorinated, Brominated and Fluorinated Organic Pollutants in African Penguin Eggs: 30 Years since the Previous Assessment. Chemosphere 2015, 126, 1–10.
- 27.
Mwangi, J.K.; Lee, W.-J.; Wang, L.-C.; et al. Persistent Organic Pollutants in the Antarctic Coastal Environment and Their Bioaccumulation in Penguins. Environ. Pollut. 2016, 216, 924–934. https://doi.org/10.1016/j.envpol.2016.07.001.
- 28.
Celis, J.E.; Espejo, W.; Groffen, T.; et al. Per- and Polyfluoroalkylated Substances (PFAS) in the Feathers and Excreta of Gentoo Penguins (Pygoscelis papua) from the Antarctic Peninsula. Sci. Total Environ. 2025, 959, 178333. https://doi.org/10.1016/j.scitotenv.2024.178333.
- 29.
Andrews, D.Q.; Stoiber, T.; Temkin, A.M.; et al. Has the Human Population Become a Sentinel for the Adverse Effects of PFAS Contamination on Wildlife Health and Endangered Species? Sci. Total Environ. 2023, 901, 165939. https://doi.org/10.1016/j.scitotenv.2023.165939.
- 30.
O’Connell, S.G.; Kincl, L.D.; Anderson, K.A. Silicone Wristbands as Personal Passive Samplers. Environ. Sci. Technol. 2014, 48, 3327–3335. https://doi.org/10.1021/es405022f.
- 31.
Samon, S.M.; Hammel, S.C.; Stapleton, H.M.; et al. Silicone Wristbands as Personal Passive Sampling Devices: Current Knowledge, Recommendations for Use, and Future Directions. Environ. Int. 2022, 169, 107339. https://doi.org/10.1016/j.envint.2022.107339.
- 32.
Travis, S.C.; Aga, D.S.; Queirolo, E.I.; et al. Catching Flame Retardants and Pesticides in Silicone Wristbands: Evidence of Exposure to Current and Legacy Pollutants in Uruguayan Children. Sci. Total Environ. 2020, 740, 140136. https://doi.org/10.1016/j.scitotenv.2020.140136.
- 33.
Levasseur, J.L.; Hoffman, K.; Herkert, N.J.; et al. Characterizing Firefighters’ Exposure to over 130 SVOCs Using Silicone Wristbands: A Pilot Study Comparing On-Duty and Off-Duty Exposures. Sci. Total Environ. 2022, 834, 155237. https://doi.org/10.1016/j.scitotenv.2022.155237.
- 34.
Niu, S.; Zhu, X.; Chen, R.; et al. Personal Wearable Sampler for Per- and Polyfluoroalkyl Substances Exposure Assessment. Environ. Sci. Technol. Lett. 2024, 11, 301–307. https://doi.org/10.1021/acs.estlett.4c00026.
- 35.
Vaezafshar, S.; Wolk, S.; Arrandale, V.H.; et al. Young Children’s Exposure to Chemicals of Concern in Their Sleeping Environment: An In-Home Study. Environ. Sci. Technol. Lett. 2025, 12, 468–475. https://doi.org/10.1021/acs.estlett.5c00051.
- 36.
Craig, J.A.; Ceballos, D.M.; Fruh, V.; et al. Exposure of Nail Salon Workers to Phthalates, Di(2-ethylhexyl) Terephthalate, and Organophosphate Esters: A Pilot Study. Environ. Sci. Technol. 2019, 53, 14630–14637. https://doi.org/10.1021/acs.est.9b02474.
- 37.
Hoxie, T.; Zhang, S.; Herkert, N.J.; et al. Silicone Wristbands as a Personal Passive Sampler to Evaluate Indoor Exposure to Volatile and Non-volatile PFASs. Environ. Sci. Technol. 2024, 58, 16316–16326. https://doi.org/10.1021/acs.est.4c05707.
- 38.
Wise, C.F.; Hammel, S.C.; Herkert, N.J.; et al. Comparative Assessment of Pesticide Exposures in Domestic Dogs and Their Owners Using Silicone Passive Samplers and Biomonitoring. Environ. Sci. Technol. 2022, 56, 1149–1161. https://doi.org/10.1021/acs.est.1c06819.
- 39.
Wise, C.F.; Herkert, N.J.; Hoffman, K.; et al. Environmental Exposures and Canine Bladder Cancer: A Case Control Study Using Silicone Passive Samplers. Environ. Sci. Technol. 2025, 59, 1121–1132. https://doi.org/10.1021/acs.est.4c09271.
- 40.
Matheson, R.; Sexton, C.L.; Wise, C.F.; et al. Silicone Tags as an Effective Method of Monitoring Environmental Contaminant Exposures in a Geographically Diverse Sample of Dogs from the Dog Aging Project. Front. Vet. Sci. 2024, 11, 1394061. https://doi.org/10.3389/fvets.2024.1394061.
- 41.
Poutasse, C.M.; Herbstman, J.B.; Peterson, M.E.; et al. Silicone Pet Tags Associate Tris(1,3-dichloro-2-isopropyl) Phosphate Exposures with Feline Hyperthyroidism. Environ. Sci. Technol. 2019, 53, 9203–9213. https://doi.org/10.1021/acs.est.9b02226.
- 42.
Swanson, J.E.; Muths, E.; Pierce, C.L.; et al. Exploring the Amphibian Exposome in an Agricultural Landscape Using Telemetry and Passive Sampling. Sci. Rep. 2018, 8, 10045. https://doi.org/10.1038/s41598-018-28132-3.
- 43.
Yaw, T.J.; Swanson, J.E.; Pierce, C.L.; et al. Placement of Intracoelomic Radiotransmitters and Silicone Passive Sampling Devices in Northern Leopard Frogs (Lithobates pipiens). J. Herpetol. Med. Surg. 2017, 27, 111–115. https://doi.org/10.5818/16-10-090.1.
- 44.
Kaidarova, A.; Geraldi, N.R.; Wilson, R.P.; et al. Wearable Sensors for Monitoring Marine Environments and Their Inhabitants. Nat. Biotechnol. 2023, 41, 1208–1220. https://doi.org/10.1038/s41587-023-01827-3.
- 45.
Quintana, F.; Wilson, R.; Gomez-Laich, A. Sintesis y Revision Critica del Uso de Bio-registradores para Aves Marinas en Sudamerica. El Hornero 2024, 39, 65–94. https://doi.org/10.56178/eh.v39i2.1488.
- 46.
Boersma, P.D.; Frere, E.; Kane, O.J.; et al. Magellanic Penguin (Spheniscus magellanicus). In Penguins: Natural History and Conservation; Garcia-Borboroglu, P., Boersma, P.D., Eds.; University of Washington Press: Seattle, WA, USA, 2013; pp. 285–302.
- 47.
Wilson, R.P. A Method for Restraining Penguins. Mar. Ornithol. 1997, 25, 72–73.
- 48.
Pozzi, L.M.; Borboroglu, P.G.; Boersma, P.D.; et al. Population Regulation in Magellanic Penguins: What Determines Changes in Colony Size? PLoS ONE 2015, 10, e0119002.
- 49.
Instituto Nacional de Estadistica y Censos. Censo Nacional de Poblacion, Hogares y Viviendas 2022, Resultados Definitivos: Indicadores Demograficos, por Sexo y Edad. Available online: https://www.indec.gob.ar/ftp/cuadros/poblacion/censo2022_indicadores_demograficos.pdf (accessed on 7 October 2025).
- 50.
García-Borboroglu, P.; Pozzi, L.M.; Parma, A.M.; et al. Population Distribution Shifts of Magellanic Penguins in Northern Patagonia, Argentina: Implications for Conservation and Management Strategies. Ocean Coast. Manag. 2022, 226, 106259. https://doi.org/10.1016/j.ocecoaman.2022.106259.
- 51.
Blanco, G.S.; Gallo, L.; Pisoni, J.P.; et al. At-Sea Distribution, Movements and Diving Behavior of Magellanic Penguins Reflect Small-Scale Changes in Oceanographic Conditions around the Colony. Mar. Biol. 2022, 169, 29. https://doi.org/10.1007/s00227-021-04016-5.
- 52.
Sala, J.; Wilson, R.; Frere, E.; et al. Foraging Effort in Magellanic Penguins in Coastal Patagonia, Argentina. Mar. Ecol. Prog. Ser. 2012, 464, 273–287.
- 53.
Gomez-Laich, A.; Wilson, R.P.; Sala, J.E.; et al. Moving Northward: Comparison of the Foraging Effort of Magellanic Penguins from Three Colonies of Northern Patagonia. Mar. Biol. 2015, 162, 1451–1461.
- 54.
Rebstock, G.A.; Garcia Borboroglu, P.; Boersma, P.D. Variability in Foraging Range and Direction among Colonies in a Widespread Seabird, the Magellanic Penguin. Front. Mar. Sci. 2022, 9, 815706. https://doi.org/10.3389/fmars.2022.815706.
- 55.
Yamamoto, T.; Yoda, K.; Blanco, G.S.; et al. Female-Biased Stranding in Magellanic Penguins. Curr. Biol. 2019, 29, R12–R13.
- 56.
Barrionuevo, M.; Frere, E.; Quintana, F.; et al. Within- and Among-Colony Variation in Non-breeding Dispersion of Magellanic Penguins Breeding along the Coast of Argentina. Mar. Ecol. Prog. Ser. 2023, 721, 151–160. https://doi.org/10.3354/meps14412.
- 57.
Bertellotti, M.; Tella, J.L.; Godoy, J.A.; et al. Determining Sex of Magellanic Penguins Using Molecular Procedures and Discriminant Functions. Waterbirds 2002, 25, 479–484.
- 58.
Camdzic, D.; Welgama, H.K.; Crawley, M.R.; et al. Rapid Capture of Per- and Polyfluoroalkyl Substances Using a Self-Assembling Zirconium-Based Metal-Organic Cage. ACS Appl. Eng. Mater. 2024, 2, 87–95. https://doi.org/10.1021/acsaenm.3c00592.
- 59.
R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2023.
- 60.
Ogle, D.H.; Doll, J.C.; Wheeler, A.P.; et al. FSA (Fisheries Stock Assessment), 2025. Available online: https://fishr-core-team.github.io/FSA/ (accessed on 7 October 2025).
- 61.
Wickham, H. ggplot2, 2016. Available online: https://ggplot2.tidyverse.org (accessed on 7 October 2025).
- 62.
Wickham, H. plyr: The split-apply-combine strategy for R, 2023. Available online: http://had.co.nz/plyr/ (accessed on 7 October 2025).
- 63.
Mangiafico, S.S. Summary and Analysis of Extension Program Evaluation in R, 2016. Available online: https://rcompanion.org/handbook/ (accessed on 7 October 2025).
- 64.
Gao, K.; Miao, X.; Fu, J.; et al. Occurrence and Trophic Transfer of Per- and Polyfluoroalkyl Substances in an Antarctic Ecosystem. Environ. Pollut. 2020, 257, 113383. https://doi.org/10.1016/j.envpol.2019.113383.
- 65.
Ahrens, L.; Rakovic, J.; Ekdahl, S.; et al. Environmental Distribution of Per- and Polyfluoroalkyl Substances (PFAS) on Svalbard: Local Sources and Long-Range Transport to the Arctic. Chemosphere 2023, 345, 140463. https://doi.org/10.1016/j.chemosphere.2023.140463.
- 66.
Van Der Schyff, V.; Kwet Yive, N.S.C.; Polder, A.; et al. Perfluoroalkyl Substances (PFAS) in Tern Eggs from St. Brandon’s Atoll, Indian Ocean. Mar. Pollut. Bull. 2020, 154, 111061. https://doi.org/10.1016/j.marpolbul.2020.111061.
- 67.
National Oceanic and Atmospheric Administration Physical Sciences Laboratory. Multivariate ENSO Index Version 2 (MEI.v2). Available online: https://psl.noaa.gov/enso/mei/ (accessed on 7 October 2025).
- 68.
Chen, C.; Wang, J.; Li, L.; et al. Comparison of Fluorotelomer Alcohol Emissions from Wastewater Treatment Plants into Atmospheric and Aquatic Environments. Environ. Int. 2020, 139, 105718. https://doi.org/10.1016/j.envint.2020.105718.
- 69.
Bohannon, M.E.; Narizzano, A.M.; Guigni, B.A.; et al. Next-Generation PFAS 6:2 Fluorotelomer Sulfonate Reduces Plaque Formation in Exposed White-Footed Mice. Toxicol. Sci. 2023, 192, 97–105. https://doi.org/10.1093/toxsci/kfad006.
- 70.
Secretaria de Gobierno de Ambiente y Desarrollo Sustentable. Resolucion 451/2019 (RESOL-2019-451-APN-SGAYDS#SGP). Available online: https://www.boletinoficial.gob.ar/detalleAviso/primera/222134/20191128 (accessed on 7 October 2025).
- 71.
Newsted, J.L.; Jones, P.D.; Coady, K.; et al. Avian Toxicity Reference Values for Perfluorooctane Sulfonate. Environ. Sci. Technol. 2005, 39, 9357–9362. https://doi.org/10.1021/es050989v.
- 72.
Haque, F.; Soerensen, A.L.; Sköld, M.; et al. Per- and Polyfluoroalkyl Substances (PFAS) in White-Tailed Sea Eagle Eggs from Sweden: Temporal Trends (1969–2021), Spatial Variations, Fluorine Mass Balance, and Suspect Screening. Environ. Sci. Processes Impacts 2023, 25, 1549–1563. https://doi.org/10.1039/D3EM00141E.
- 73.
Rayne, S.; Forest, K. Perfluoroalkyl Sulfonic and Carboxylic Acids: A Critical Review of Physicochemical Properties, Levels and Patterns in Waters and Wastewaters, and Treatment Methods. J. Environ. Sci. Health Part A 2009, 44, 1145–1199. https://doi.org/10.1080/10934520903139811.
- 74.
Higgins, C.P.; Luthy, R.G. Sorption of Perfluorinated Surfactants on Sediments. Environ. Sci. Technol. 2006, 40, 7251–7256. https://doi.org/10.1021/es061000n.
- 75.
Goss, K.-U. The pKa Values of PFOA and Other Highly Fluorinated Carboxylic Acids. Environ. Sci. Technol. 2008, 42, 456–458. https://doi.org/10.1021/es702192c.
- 76.
Wang, Q.; Shao, Y.; Leung, K.M.Y.; et al. Per- and Polyfluoroalkyl Substances (PFAS) in the Marine Environment: An Overview and Prospects. Mar. Pollut. Bull. 2025, 216, 117993. https://doi.org/10.1016/j.marpolbul.2025.117993.
- 77.
Gomis, M.I.; Wang, Z.; Scheringer, M.; et al. A Modeling Assessment of the Physicochemical Properties and Environmental Fate of Emerging and Novel Per- and Polyfluoroalkyl Substances. Sci. Total Environ. 2015, 505, 981–991. https://doi.org/10.1016/j.scitotenv.2014.10.062.
- 78.
Marciano, J.; Crawford, L.; Mukhopadhyay, L.; et al. Per/Polyfluoroalkyl Substances (PFASs) in a Marine Apex Predator (White Shark, Carcharodon carcharias) in the Northwest Atlantic Ocean. ACS Environ. Au 2024, 4, 152–161. https://doi.org/10.1021/acsenvironau.3c00055.
- 79.
Khan, B.; Burgess, R.M.; Cantwell, M.G. Occurrence and Bioaccumulation Patterns of Per- and Polyfluoroalkyl Substances (PFAS) in the Marine Environment. ACS EST Water 2023, 3, 1243–1259. https://doi.org/10.1021/acsestwater.2c00296.
- 80.
Khalid, N.K.; Le Calvez, M.; Lemire, M.; et al. Occurrence of 80 Per and Polyfluorinated Alkyl Substances (PFAS) in Muscle and Liver Tissues of Marine Mammals of the St. Lawrence Estuary and Gulf, Quebec, Canada. Front. Environ. Chem. 2024, 5, 1403728. https://doi.org/10.3389/fenvc.2024.1403728.
- 81.
Xie, Z.; Kallenborn, R. Legacy and Emerging Per- and Poly-fluoroalkyl Substances in Polar Regions. Curr. Opin. Green Sustain. Chem. 2023, 42, 100840. https://doi.org/10.1016/j.cogsc.2023.100840.
- 82.
Richdale, L.E. Banding and Marking Penguins. Bird-Banding 1951, 22, 47–54.
- 83.
Ratcliffe, N.; Takahashi, A.; Oulton, C.; et al. A Leg-Band for Mounting Geolocator Tags on Penguins. Mar. Ornithol. 2014, 42, 23–26.
- 84.
Wilson, R.P. The Price Tag. Nature 2011, 469, 164–165. https://doi.org/10.1038/469164a.
- 85.
Jackson, S.; Wilson, R.P. The Potential Costs of Flipper-Bands to Penguins. Funct. Ecol. 2002, 16, 141–148. https://doi.org/10.1046/j.0269-8463.2001.00606.x.
- 86.
Draghi, S.; Curone, G.; Risoluti, R.; et al. Comparative Analysis of PFASs Concentrations in Fur, Muscle, and Liver of Wild Roe Deer as Biomonitoring Matrices. Front. Vet. Sci. 2024, 11, 1500651. https://doi.org/10.3389/fvets.2024.1500651.
- 87.
Robuck, A.R.; McCord, J.P.; Strynar, M.J.; et al. Tissue-Specific Distribution of Legacy and Novel Per- and Polyfluoroalkyl Substances in Juvenile Seabirds. Environ. Sci. Technol. Lett. 2021, 8, 457–462. https://doi.org/10.1021/acs.estlett.1c00222.
- 88.
Anderson, K.A.; Points, G.L.; Donald, C.E.; et al. Preparation and Performance Features of Wristband Samplers and Considerations for Chemical Exposure Assessment. J. Expo. Sci. Environ. Epidemiol. 2017, 27, 551–559. https://doi.org/10.1038/jes.2017.9.
- 89.
Wang, L.; Liu, R.; Liu, X.; et al. Sampling Rate of Polar Organic Chemical Integrative Sampler (POCIS): Influence Factors and Calibration Methods. Appl. Sci. 2020, 10, 5548. https://doi.org/10.3390/app10165548.
- 90.
Kaserzon, S.L.; Vermeirssen, E.L.M.; Hawker, D.W.; et al. Passive Sampling of Perfluorinated Chemicals in Water: Flow Rate Effects on Chemical Uptake. Environ. Pollut. 2013, 177, 58–63. https://doi.org/10.1016/j.envpol.2013.02.002.
- 91.
Rey, A.R.; Putz, K.; Scioscia, G.; et al. Sexual Differences in the Foraging Behaviour of Magellanic Penguins Related to Stage of Breeding. Emu 2012, 112, 90–96.
- 92.
Wilson, R.P.; Scolaro, J.A.; Gremillet, D.; et al. How Do Magellanic Penguins Cope with Variability in Their Access to Prey? Ecol. Monogr. 2005, 75, 379–401.
- 93.
Garcia-Borboroglu, P.; Boersma, P.D. Penguins: Natural History and Conservation; University of Washington Press: Seattle, WA, USA, 2013.

This work is licensed under a Creative Commons Attribution 4.0 International License.


