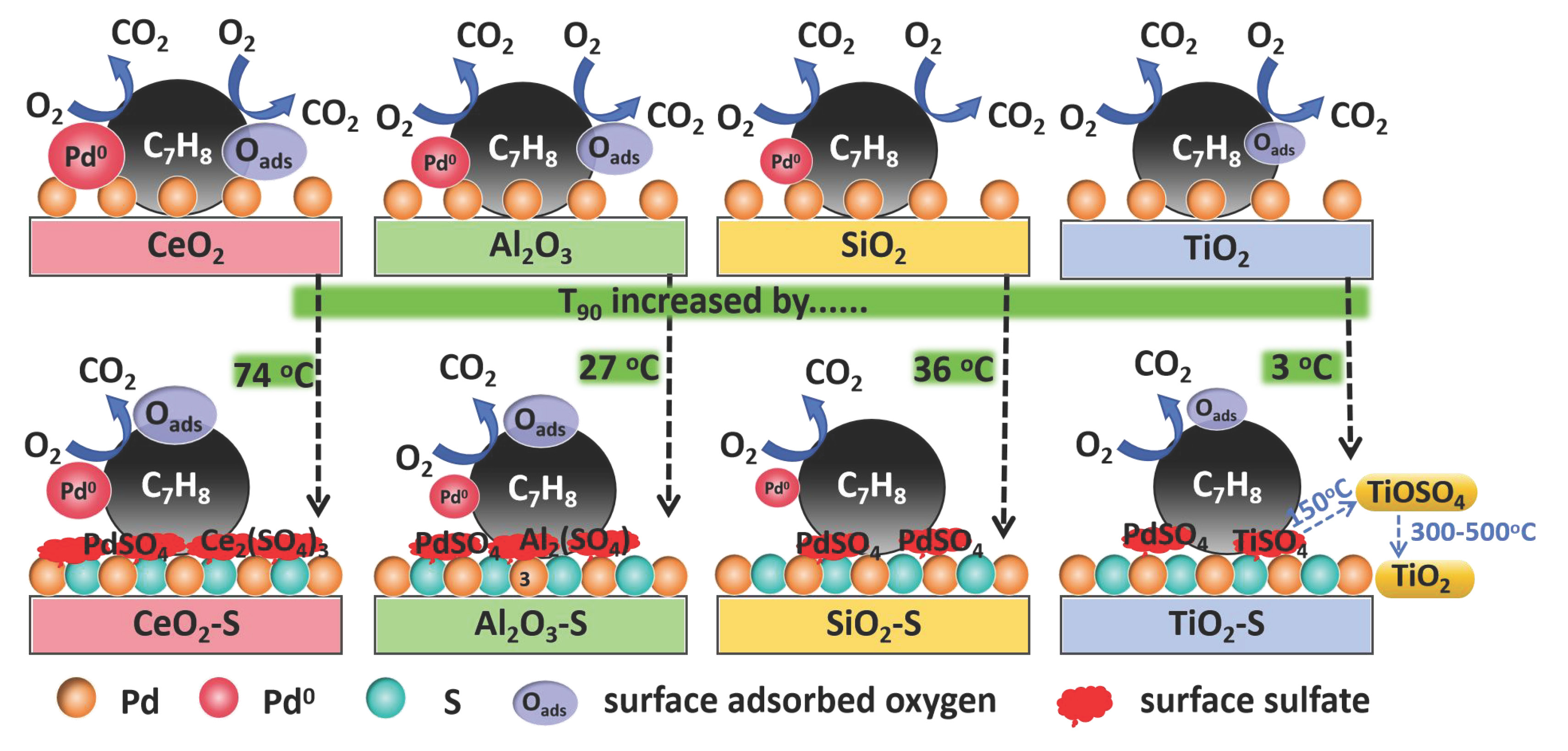
Developing sulfur tolerance catalyst for combustion of volatile organic compounds (VOCs) is of great significance in the removal of industrial waste gas. In this work, the roles of support over Pd-based catalyst for the sulfur tolerance during toluene oxidation were investigated by loading 0.5 wt% Pd onto CeO2, Al2O3, TiO2, and SiO2 with or without SO2 pretreatment. Results showed that the Pd/CeO2 displays an attractive toluene conversion of 90% at 220 °C, outperforming other supported Pd catalysts, especially the Pd/TiO2 catalyst (with a T90 of 253 °C). However, after SO2 pretreatment, the toluene oxidation performance of Pd/TiO2 ranked first, and its T90 was 38 °C lower than that of Pd/CeO2. Characterization results indicated that the Pd0 active species on the CeO2 surface is highly prone to reacts with SO2 to form stable Ce2(SO4)3 deposition. Additionally, the decrease in activated oxygen capacity inhibits its redox performance. These factors collectively result in poor sulfur resistance of Pd/CeO2. While the outstanding sulfur resistance of Pd/TiO2 was closely linked to the ready decomposition of sulfates on its surface. This work elucidates the critical role of the support in dictating catalytic performance and anti-poisoning capability, thereby offering new perspectives for the design of high-performance Pd-based catalysts for VOC remediation.
- Open Access
- Article
The Crucial Role of Support in the Pd/MO Catalyst for SO2 Resistance during Toluene Combustion
- Gehui Wang 1,
- Junrong Lai 1,
- Zhengxuan Zhu 1,
- Haiqin Wan 1,2,*,
- Lin Dong 1,2
Author Information
Received: 01 Feb 2026 | Revised: 08 Apr 2026 | Accepted: 10 Apr 2026 | Published: 22 Apr 2026
Abstract
Graphical Abstract
Keywords
toluene oxidation | Pd-based catalysts | SO2 poisoning | support effects | sulfur tolerance
References
- 1.
Zheng, Y.F.; Su, Y. Interface-Enhanced Oxygen Vacancies of CoCuOx Catalysts in Situ Grown on Monolithic Cu Foam for VOC Catalytic Oxidation. Environ. Sci. Technol. 2022, 56, 1905–1916.
- 2.
Yang, Y.; Si, W.Z. Oxygen Vacancy Engineering on Copper-Manganese Spinel Surface for Enhancing Toluene Catalytic Combustion: A Comparative Study of Acid Treatment and Alkali Treatment. Appl. Catal. B Environ. 2024, 340, 123142.
- 3.
Zhang, K.; Ding, H.L. Research Progress of a Composite Metal Oxide Catalyst for VOC Degradation. Environ. Sci. Technol. 2022, 56, 9220–9236.
- 4.
Feng, Y.; Wang, C.C. Catalytic Stability Enhancement for Pollutant Removal via Balancing Lattice Oxygen Mobility and VOCs Adsorption. J. Hazard. Mater. 2022, 424, 127337.
- 5.
Zhang, B.; Shen, Y. Boosting Ozone Catalytic Oxidation of Toluene at Room Temperature by Using Hydroxyl-Mediated MnOx/Al2O3 Catalysts. Environ. Sci. Technol. 2023, 57, 7041–7050.
- 6.
Li, L.M.; Wahab, M.A. Pt-Modulated CuMnOx Nanosheets as Catalysts for Toluene Oxidation. ACS Appl. Nano Mater. 2021, 4, 6637–6647.
- 7.
Ning, H.Q.; Xie, K.Y. Boosting Sulfur Tolerance in Pd/Beta Zeolite Catalyst for Toluene Oxidation: The Role of CeO2. J. Rare Earths 2025, 43, 1635–1642.
- 8.
Nunotani, N.; Saeki, S. Novel Catalysts Based on Lanthanum Oxyfluoride for Toluene Combustion. Mater. Lett. 2020, 258, 126802.
- 9.
Qi, Y.; Yang, Z.Y. Platinum Copper Bimetallic Nanoparticles Supported on TiO2 as Catalysts for Photo-Thermal Catalytic Toluene Combustion. ACS Appl. Nano Mater. 2022, 5, 1845–1854.
- 10.
Wu, C.; Liu, Q.L. Insights into the Surface Structure-Sensitive Photocatalytic Oxidation of Gaseous Toluene on Pd/TiO2 Catalysts. Chem. Eng. J. 2023, 475, 146294.
- 11.
Shen, Y.; Liu, S.S. Photocatalytic Degradation of Toluene by a TiO2 p-n Homojunction Nanostructure. ACS Appl. Nano Mater. 2022, 5, 18612–18621.
- 12.
Shan, C.P.; Hou, X.Y. Recent Advances of Gaseous Pollutant Catalytic Oxidation over Precious Metal Catalysts with SO2 Exposure. Environ. Sci. Technol. 2025, 59, 2348–2367.
- 13.
Xie, K.Y.; Wang, Z. Synthesis of Quantitative Sulfur-Poisoned Pd/γ-Al2O3 and Its Deactivation Mechanism for Catalytic Combustion of Toluene. Appl. Catal. A Gen. 2022, 639, 118641.
- 14.
Wang, Y.F.; Liu, X.Y. Promotion of Catalytic Performance of Pd/Al2O3 for o-Xylene Oxidation by Morphological Control. Chem. Eng. J. 2023, 472, 145013.
- 15.
Xu, F.P.; Wang, J. Catalytic Performance and Mechanism of Toluene Oxidation in High Humidity over the Mesoporous Titania-Ceria-Supported Pt or Pd Catalysts. Appl. Catal. A Gen. 2025, 699, 120277.
- 16.
Wang, S.; Wang, S. CO Oxidation with Pt Catalysts Supported on Different Supports: A Comparison of Their Sulfur Tolerance Properties. Appl. Catal. A Gen. 2023, 654, 119083.
- 17.
Hao, X.H.; Wu, S.N. In Situ Reaction Induced Strong Metal-Support Interaction to Enhance Catalytic Performance and Stability of Toluene Oxidation. Sep. Purif. Technol. 2025, 358, 130266.
- 18.
Gil, S.; Garcia-Vargas, J.M. Catalytic Oxidation of Propene over Pd Catalysts Supported on CeO2, TiO2, Al2O3 and M/Al2O3 Oxides (M = Ce, Ti, Fe, Mn). Catalysts 2015, 5, 671–689.
- 19.
Li, G.; Shen, K. SO2 Poisoning Mechanism of the Multi-Active Center Catalyst for Chlorobenzene and NOx Synergistic Degradation at Dry and Humid Environments. Environ. Sci. Technol. 2021, 55, 13186–13197.
- 20.
Luo, N.; Gao, F. Hierarchical Structured Ti-Doped CeO2 Stabilized CoMn2O4 for Enhancing the Low-Temperature NH3-SCR Performance within Highly H2O and SO2 Resistance. Appl. Catal. B Environ. 2024, 343, 123442.
- 21.
Jiang, B.S.; Xie, K.Y. Study on the Mechanism of Sulfur Poisoning in Toluene Catalyzed by Co3O4/γ-Al2O3 Sulfur Tolerant Catalyst Containing Spinel Structure. J. Environ. Chem. Eng. 2023, 11, 10518.
- 22.
Payan, A.; Soltan, J. Intensified Catalytic Decomposition of Acetone at Room Temperature Using a Ag-Modified CeO2–Al2O3 Binary Metal Oxide Support: Enhancing Synergies, Role of Relative Humidity, and in Situ Mechanistic Interpretation. Ind. Eng. Chem. Res. 2025, 64, 8047–8063.
- 23.
Payan, A.; Soltan, J. Impact of Support on Acetone Oxidation by Ag@S (S = CeO2, Al2O3, ZSM-5) Catalysts under VUV Illumination at Room Temperature: Improving Synergy, Influence of Relative Humidity, and In-Situ Mechanistic Studies. J. Environ. Chem. Eng. 2025, 13, 117334.
- 24.
Escandón, L.S.; Ordóñez, S. Sulphur Poisoning of Palladium Catalysts Used for Methane Combustion: Effect of the Support. J. Hazard. Mater. 2008, 153, 742–750.
- 25.
Ding, Y.; Wang, S. Investigation of Supported Palladium Catalysts for Combustion of Methane: The Activation Effect Caused by SO2. Chem. Eng. J. 2020, 382, 122969.
- 26.
Murindababisha, D.; Yusuf, A. Low-Temperature Catalytic Oxidation of Toluene Using CeO2@MIL-101(Fe): Stability against Water and SO2. Process Saf. Environ. Prot. 2025, 197, 107005.
- 27.
Wang, Z.; Xie, K.Y.; Zheng, J.; et al. Studies of Sulfur Poisoning Process via Ammonium Sulfate on MnO2/γ-Al2O3 Catalyst for Catalytic Combustion of Toluene. Appl. Catal. B Environ. 2021, 298, 120595.
- 28.
Feng, Z.T.; Ren, Q.M. Effect of CeO2 Morphologies on Toluene Catalytic Combustion. Catal. Today 2019, 332, 177–182.
- 29.
Wang, Z.W.; Li, S.X. Supported Ultralow Loading Pt Catalysts with High H2O-, CO2-, and SO2-Resistance for Acetone Removal. Appl. Catal. A Gen. 2019, 579, 106–115.
- 30.
Xiang, N.; Han, X.J. Size Effect of γ-Al2O3 Supports on the Catalytic Performance of Pd/γ-Al2O3 Catalysts for HCHO Oxidation. Mol. Catal. 2020, 494, 111112.
- 31.
Xu, J.Y.; Zhu, L.H. SiO2 Supported Pd Nanoparticles for Highly Efficient, Selective and Stable Phenol Hydrogenation to Cyclohexanone. Mol. Catal. 2023, 538, 112975.
- 32.
Bazin, P.; Saur, O. A Thermogravimetric and FT IR Study of the Reduction by H2 of Sulfated Pt/CexZr1−xO2 Solids. Appl. Catal. B Environ. 2009, 90, 368–379.
- 33.
Zhang, X.F.; Liu, Y. Catalytic Performance and SO2 Resistance of Zirconia Supported Platinum-Palladium Bimetallic Nanoparticles for Methane Combustion. Catal. Today 2022, 402, 138–148.
- 34.
Huang, Z.; Fan, Z. Enhanced NOx Catalytic Reduction by NH3 over Polymeric Sulfur Species in CeO2 via Tailoring Ce–O Bonds. Inorg. Chem. 2025, 64, 20517–20526.
- 35.
Srinithi, S.; Chen, S.M. Fabrication of N Rich Graphitic Carbon Nitride Supported CeO2 for Improved Photocatalytic Charge Separation and Electrochemical Electron Transfer Properties. Surf. Interfaces 2023, 39, 102877.
- 36.
Arooj, A.; Tahir, K. One-Step Fabrication of Surfactant Mediated Pd/SiO2, a Prospect toward Therapeutic and Photocatalytic Applications. Inorg. Chem. Commun. 2022, 142, 109692.
- 37.
Gao, Q.; Dong, C.Q. Effect of the Fe2O3@TiO2 Core-Shell Structure on CO Catalytic Oxidation and SO2 Poisoning Resistance. Mol. Catal. 2023, 547, 113308.
- 38.
Heshmatpour, F.; Aghakhanpour, R.B. Synthesis and Characterization of Superfine Pure Tetragonal Nanocrystalline Sulfated Zirconia Powder by a Non-Alkoxide Sol-Gel Route. Adv. Powder Technol. 2012, 23, 80–87.
- 39.
Dun, Y.H.; Liu, Y.F. Pt0-MnSO4 Active Centers on Modified SmMn2O5 Mullite Oxides for Efficient Propane Oxidation. Appl. Catal. B Environ. 2025, 371, 125223.
- 40.
Li, J.; Wu, Y. Carbothermal Reduction Synthesis of Superfined TiC Powders from TiOSO4. Adv. Mater. Res. 2011, 415, 510–513.
- 41.
Kinnunen, N.M.; Hirvi, J.T. Case Study of a Modern Lean-Burn Methane Combustion Catalyst for Automotive Applications: What Are the Deactivation and Regeneration Mechanisms? Appl. Catal. B Environ. 2017, 207, 114–119.
- 42.
Jiang, D.; Khivantsev, K. Low-Temperature Methane Oxidation for Efficient Emission Control in Natural Gas Vehicles: Pd and Beyond. ACS Catal. 2020, 10, 14304–14314.
- 43.
Wang, L.; Sun, Y.G. Electron Donation Promotes the Dual Activation of Lattice Oxygen and Molecular Oxygen: The Pt-Pd/CeO2 Catalyst Efficiently Catalyzes Toluene. J. Catal. 2023, 428, 115133.
- 44.
Liu, T.; Yan, H.M. Promoting Pd/Al2O3 Catalysts for Toluene Combustion by DBD Plasma Treating in Different Working Gas Atmospheres. Catal. Today 2023, 421, 114177.
- 45.
Zhang, W.J.; Liu, G.F. Temperature Sensitivity of the Selective Catalytic Reduction (SCR) Performance of Ce-TiO2 in the Presence of SO2. Chemosphere 2019, 243, 125419.
- 46.
Byun, M.Y.; Park, D.W. Effect of Oxide Supports on the Activity of Pd Based Catalysts for Furfural Hydrogenation. Catalysts 2020, 10, 837.
- 47.
Fan, L.P.; Zhang, J. Ceria Morphology-Dependent Pd-CeO2 Interaction and Catalysis in CO2 Hydrogenation into Formate. J. Catal. 2021, 397, 116–127.
- 48.
Liu, X.G.; Zhang, X. Pd Nanoparticles Supported on N-Doped TiO2 Nanosheets: Crystal Facets, Defective Sites, and Metal-Support Interactions Boost Reforming of Formaldehyde Solution for Hydrogen Production. Langmuir 2022, 38, 13532–13542.

This work is licensed under a Creative Commons Attribution 4.0 International License.


