- Spatial variability of ionic distribution in tropical estuaries were studied.
- Ca–Cl and Na–Cl water types suggest seawater mixing, evaporation and ion exchange.
- Isotopes reveal progressive evaporation in estuaries due to continental effect.
- PCA determines seawater influx, weathering and anthropogenic impacts in estuaries.
- High to medium level of metal pollution in MRE and KBE needs continuous monitoring.
- Open Access
- Article
Geochemical Characteristics of Tropical Estuarine Waters in Northern Borneo: Implication for Elemental Distribution, Mechanisms, Sources and Pollution Risk
- Prasanna Mohan Viswanathan 1,*,
- Benedict Won Yong Tian 1,
- Chidambaram Sabarathinam 2,
- Munirah Abdul Zali 3
Author Information
Received: 15 Jan 2026 | Revised: 03 Mar 2026 | Accepted: 11 Mar 2026 | Published: 18 Mar 2026
Highlights
Abstract
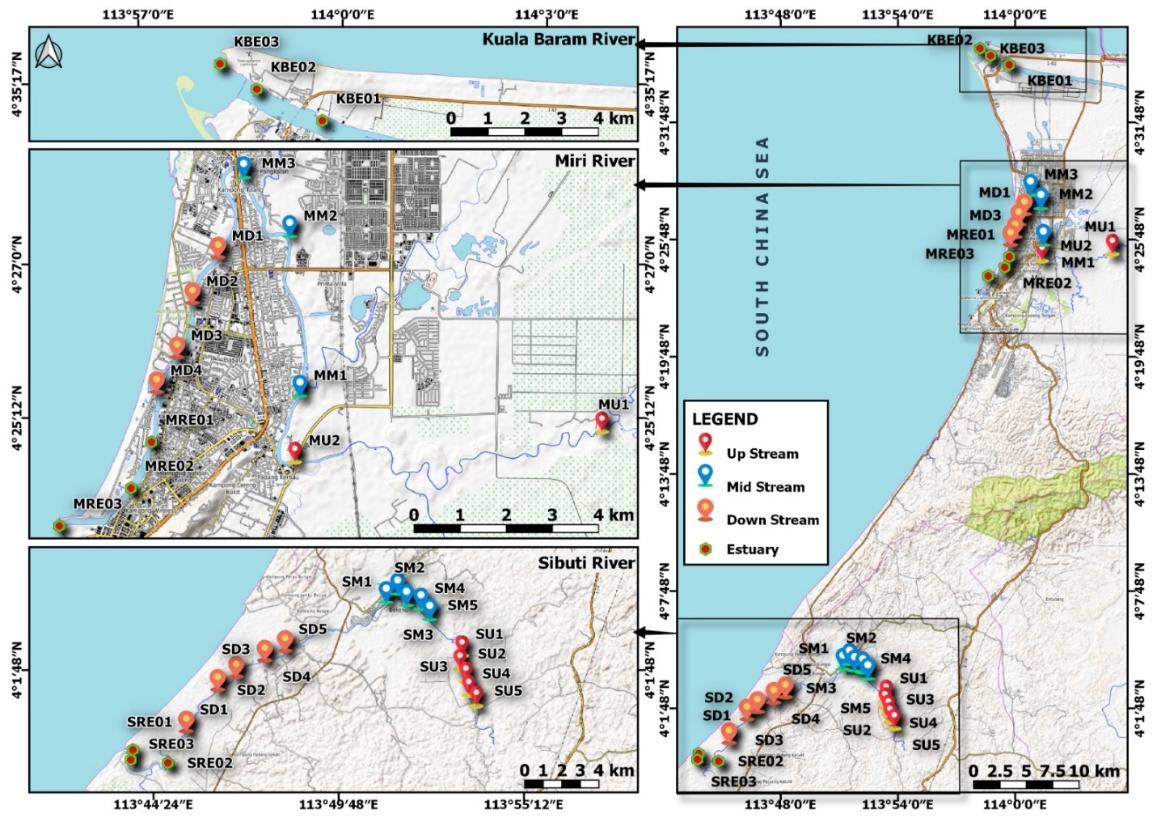
Estuaries are exceptional and vital ecosystems which create a dynamic region with biological, ecological, and environmental advantages. In this study, surface water samples were collected at three estuaries (Miri River, Sibuti River and Kuala Baram River) adjoining the South China Sea, and compared with the upper section of the rivers. The collected samples were analysed for in situ parameters, major ions, trace metals and stable isotopes using standard procedures. Results showing spatial variations in ionic concentrations across three estuaries, controlled by multi-geochemical processes such as weathering, ion exchange and seawater influx. Dominant water types varied: Ca–Cl in Miri and Sibuti rivers, and Na–Cl in Kuala Baram River, indicating distinct influences of seawater mixing and ion exchange reactions. Evaporation process increased the salinity in estuaries, coupled with the anthropogenic impacts in the downstream. Ionic ratios also prove seawater and freshwater mixing, followed by mineral precipitation and ion exchange. The undersaturation state of carbonate and sulphate minerals indicates the dissolution in the lower and upper reaches of the rivers, and approaching towards saturation state in estuaries might lead to precipitation of minerals in favour of pH and seawater influx. The higher LogpCO2 values in the upper sections of the rivers indicate the weathering of source rocks. The isotopic ratio of δ18O and δ2H and d-excess values confirm the progressive dominance of evaporation in estuaries due to the continental effect. Statistical analysis reveals key geochemical processes such as seawater mixing, weathering, ion exchange and anthropogenic activities, that impact the hydrochemistry of estuaries. Miri and Kuala Baram estuaries are at high and medium levels of pollution based on heavy metal evaluation index (HEI), which require continuous monitoring, particularly for the higher concentrations of Zn, Cu, Pb and Mn.
Graphical Abstract
Keywords
estuary | seawater mixing | ion exchange | evaporation | Miri coast
References
- 1.
McLusky, DS.; Elliott, M. The Estuarine Ecosystem: Ecology, Threats and Management, 3rd ed.; Oxford University Press: Oxford, UK, 2004. https://doi.org/10.1093/acprof:oso/9780198525080.001.0001
- 2.
Billah, M.; Kamal, A.; Hoque, M.; et al. Temporal distribution of water characteristics in the Miri estuary, east Malaysia. Zool. Ecol. 2016, 26, 134–140. https://doi.org/10.1080/21658005.2016.1148960
- 3.
Nagarajan, R.; Eswaramoorthi, S.G.; Anandkumar, A.; et al. Geochemical fractionation, mobility of elements and environmental significance of surface sediments in a tropical river, Borneo. Mar. Pollut. Bull. 2023, 192, 115090. https://doi.org/10.1016/j.marpolbul.2023.115090
- 4.
Saifullah, A.S.M.; Abu Hena, M.K.; Idris, M.H.; et al. Seasonal variation of water characteristics in Kuala Sibuti river estuary in Miri, Sarawak, Malaysia. Malays. J. Sci. 2014, 33, 9–22. https://doi.org/10.22452/mjs.vol33no1.3
- 5.
Prabakaran, K.; Eswaramoorthi, S.; Nagarajan, R.; et al. Geochemical behaviour and risk assessment of trace elements in a tropical river, Northwest Borneo. Chemosphere 2020, 252, 126430. https://doi.org/10.1016/j.chemosphere.2020.126430
- 6.
Rakesh Roshan, G.; Prasanna. MV.; Nagarajan. R.; et al. Spatial and temporal variations of geochemical processes and toxicity of water, sediments, and suspended solids in Sibuti River Estuary, NW Borneo. Environ. Sci. Pollut. Res. 2023, 30, 92692–92719. https://doi.org/10.1007/s11356-023-28596-5
- 7.
Claridge, P.N.; Potter, I.C.; Hardisty, M.W. Seasonal changes in movements, abundance, size composition and diversity of the fish fauna of the severn estuary. J. Mar. Biol. Assoc. U.K. 1986, 66, 229–258. https://doi.org/10.1017/S002531540003976X
- 8.
Fiona Bassy, W.; Prasanna, M.V.; Nagarajan, R.; et al. A comprehensive analysis of tracing the sources and dynamics of pollutants in a tropical Miri River, NW Borneo. Sustain. Water Resour. Manag. 2025, 11, 20. https://doi.org/10.1007/s40899-025-01203-w
- 9.
Maharjan, A.K.; Wong, D.R.E.; Rubiyatno, R.; Level and distribution of heavy metals in Miri River, Malaysia. Trop. Aquat. Soil. Pollut. 2021, 1, 74–86. https://doi.org/10.53623/tasp.v1i2.20
- 10.
Parvin Raj, S.R.; Prasanna, M.V. Occurrence and distribution of geochemical elements in Miri estuary, NW Borneo: Evaluating for processes, sources and pollution status. Chemosphere 2023, 316, 137838. https://doi.org/10.1016/j.chemosphere.2023.137838
- 11.
Mondrag´on-D´ıaz, LF.; Molina, A.; Duque, G. Influence of environmental variables on the spatiotemporal dynamics of water quality in Buenaventura Bay, Colombian Pacific. Environ. Monit. Assess. 2022, 194, 720. https://doi.org/10.1007/s10661-022-10388-y
- 12.
Dessai, D.V.G.; Nayak, G.N.; Basavaiah, N. Grain size, geochemistry, magnetic susceptibility: Proxies in identifying sources and factors controlling distribution of metals in a tropical estuary, India. Estuar. Coast. Shelf Sci. 2009, 85, 307–318. https://doi.org/10.1016/j.ecss.2009.08.020
- 13.
Thanh-Nho, N.; Strady, E.; Nhu-Trang, T.T., et al. Trace metals partitioning between particulate and dissolved phases along a tropical mangrove estuary (Can Gio, Vietnam). Chemosphere 2018, 196, 311–322. https://doi.org/10.1016/j.chemosphere.2017.12.189
- 14.
Akhtar, S.; Equeenuddin, S.M.; Roy, P.D. Spatial and seasonal variability of dissolved metals in a monsoonal estuarine environment. Reg. Stud. Mar. Sci. 2024, 73, 103463. https://doi.org/10.1016/j.rsma.2024.103463
- 15.
Balachandran, K.K.; Laluraj, C.M.; Martin, G.D.; et al. Environmental analysis of heavy metal deposition in a flow-restricted tropical estuary and its adjacent shelf. Environ. Forensics 2006, 7, 345–351. https://doi.org/10.1080/15275920600996339
- 16.
Amorim, R.M.; Delgado, J.de F.; Baptista Neto, J.A.; et al. The benthic macrofauna along the estuarine gradient of the Paranagua estuary. Reg. Stud. Mar. Sci. 2020, 39, 101459. https://doi.org/10.1016/j.rsma.2020.101459
- 17.
Liechti, P.; Roe, F.W.; Haile, N.S.; et al. The Geology of Sarawak, Brunei and theWestern Part of North Borneo; Geological Survey Department, British Territories in Borneo, Bulletin 3; Geological Survey Department, British Territories in Borneo: Kuching, Sarawak, 1960; p. 360.
- 18.
Nagarajan, R.; Armstrong-Altrin, J.S.; Kessler, F.L.; et al. Provenance and tectonic setting of Miocene siliciclastic sediments, Sibuti formation, northwestern Borneo. Arab. J. Geosci. 2015, 8, 8549–8565. https://doi.org/10.1007/s12517-015-1833-4
- 19.
Vijith, H.; Dodge-Wan, D. Spatial and temporal characteristics of rainfall over a forested river basin in NW Borneo. Meteorol. Atmos. Phys. 2019, 132, 683–702. https://doi.org/10.1007/s00703-019-00714-4
- 20.
Hoque, M.M.; Kamal, AH.; Idris, MH.; et al. Status of some fishery resources in a tropical mangrove estuary of Sarawak, Malaysia. Mar. Biol. Res. 2015, 11, 834–846. https://doi.org/10.1080/17451000.2015.101.6970
- 21.
Fiona Bassy, W.; Prasanna, MV.; Nagarajan, R. Spatial and temporal distribution of geochemical elements and their processes in different size fractions–Miri River (NW Borneo). Kuwait J. Sci. 2024. 51, 100136. https://doi.org/10.1016/j.kjs.2023.10.004
- 22.
APHA. Standard Methods for the Examination ofWater andWastewater, 19th Edition; American Public Health Association (APHA); American Water Works Association (AWWA); Water Environment Federation (WEF): Washington, DC, USA, 1998.
- 23.
Freeze, A.R.; Cherry, J.A. Groundwater; Prentice-Hall, Inc: Englewood Cliffs, NJ, USA, 1979; p. 604.
- 24.
Craig, H. Isotopic variation in meteoric water. Science 1961, 133, 1702–1703.
- 25.
Piper, A.M. A graphic procedure in the geochemical interpretation of water analyses. Eos Trans. Am. Geophys. Union 1944, 25, 914–928. https://doi.org/10.1029/ TR025i006p00914
- 26.
Gibbs, R. Mechanisms controlling world water chemistry. Science 1970, 170, 1088–1090. https://doi.org/10.1126/science.170.3962.1088
- 27.
Ball, J.W.; Nordstrom, D.K. User’s Manual for WATEQ4F, with Revised Thermodynamic Data Base and Test Cases for Calculating Speciation of Major, Trace, and Redox Elements in Natural Waters; U.S. Geological Survey Water-Resources Investigations Report 91-4183; U.S. Geological Survey (USGS): Denver, CO, USA, 1991; pp. 1–188.
- 28.
Ninu Krishnan, M.V.; Prasanna, M.V.; Vijith, H. Isoscapes to address the regional precipitation trends in the equatorial region of southeast Asia. Phys. Chem. Earth A/B/C 2022, 127, 103159. https://doi.org/10.1016/j.pce.2022.103159
- 29.
Edet, A.E.; Offiong, O.E. Evaluation of water quality pollution indices for heavy metal contamination monitoring. A study case from Akpabuyo-Odukpani area, Lower Cross River Basin (southeastern Nigeria). GeoJournal 2002, 57, 295–304. https://doi.org/10.1023/B:GEJO.0000007250.92458.de
- 30.
Shin, K.; Koh, D.-C.; Jung, H. The hydrogeochemical characteristics of groundwater subjected to seawater intrusion in the archipelago, Korea. Water 2020, 12, 1542. https://doi.org/10.3390/w12061542
- 31.
Collins, D.S.; Johnson, H.D.; Allison, P.A. Coupled ‘storm-flood’ depositional model: Application to the Miocene–Modern Baram Delta Province, north-west Borneo. Sedimentology 2017, 64, 1203–1235. https://doi.org/ 10.1111/sed.12316
- 32.
Matson, E.A.; Brinson, M.M. Sulfate enrichments in estuarine waters of North Carolina. Estuaries 1985, 8, 279–289. https://doi.org/10.2307/1351488
- 33.
Yin, H.; Hu X.; Dias, L.M. Sulfate enrichment in estuaries of the northwestern Gulf of Mexico: The potential effect of sulfide oxidation on carbonate chemistry under a changing climate. Limnol. Oceanogr. 2023, 8, 685–798. https://doi.org/10.1002%2Flol2.10335
- 34.
Benedict Wong, Y.T. Geochemical Characteristics and Metals Dynamics in Estuaries of Miri Coastal Environment, Unpublished Bachelor’s Thesis, Curtin University, Malaysia, 2024.
- 35.
Zhao, G.; Ye, S.; Yuan, H. Surface sediment properties and heavy metal pollution assessment in the Pearl River Estuary, China. Environ. Sci. Pollut. Res. 2017, 24, 2966–2979. https://doi.org/10.1007/s11356-016-8003-4
- 36.
Essien, J.P.; Antai, S.P.; Olajire, A.A. Distribution, seasonal variations and ecotoxicological significance of heavy metals in sediments of cross river estuary mangrove swamp. Water Air Soil Pollut. 2009, 197, 91–105. https://doi.org/10.1007/s11270-008-9793-x
- 37.
Taghavi, S.M.; Nasrabadi, T.; Kachoueiyan, F. Effect of oxidationreduction potential on flocculation and mobility of metals in an estuarine environment. Reg. Stud. Mar. Sci. 2025, 89, 104278. https://doi.org/10.1016/j.rsma.2025.104278
- 38.
Oyuntsetseg, D.; Ganchimeg, D.; Minjigmaa, A. Isotopic and chemiocal studies of hot and cold springs in western part of Khangai Mounbtain region, Mongolia for geothermal exploration. Geothermics 2015, 53, 488–497. https://doi.org/10.1016/j.geothermics.2014.08.010
- 39.
Omar, T.F.T.; Aris, A.Z.; Yusoff, F.M. Occurrence, distribution, and sources of emerging organic contaminants in tropical coastal sediments of anthropogenically impacted Klang River estuary, Malaysia. Mar. Pollut. Bull. 2018, 131, 284–293. https://doi.org/10.1016/j.marpolbul.2018.04.019
- 40.
Vipindas, P.V.; Anas, A.; Jayalakshmy, K.V. Impact of seasonal changes in nutrient loading on distribution and activity of nitrifiers in a tropical estuary. Cont. Shelf Res. 2018, 154, 37–45. https://doi.org/10.1016/j.csr.2018.01.003
- 41.
Scholeller, H. Qualitative evaluation of groundwater resources. In Methods and Techniques of Groundwater Investigations and Development; UNESCO: Paris, France, 1965; pp. 54–83.
- 42.
Al Farrah, N.; Walraevens, K. Hydrogeological and hydrogeochemical investigation of the coastal area of Jifarah Plain, NW Libya. Afr. Focus 2011, 24. 95–99. https://doi.org/10.21825/af.v24i1.18029
- 43.
Gopinath, S.; Srinivasamoorthy, K.; Saravanan, K. Tracing groundwater salinization using geochemical and isotopic signature in Southeastern coastal Tamilnadu, India. Chemosphere 2019, 236, 124305. https://doi.org/10.1016/j.chemosphere.2019.07.036
- 44.
Stephan, O.; Prasanna, M.V.; Anshuman, M. A multiproxy analysis of rainwater chemistry and moisture sources in Borneo. Earth Syst. Environ. 2025, 9, 3271–3288. https://doi.org/10.1007/s41748-025-00653-8
- 45.
Rozanski, K.; Araguas-Araguas, L.; Gonfiantini, R. Isotopic patterns in modern global precipitation. In Climate Change in Continental Isotopic Records; American Geophysical Union Monograph, Volume 78; American Geophysical Union (AGU): Washington, DC, USA, 1993; pp. 1–36.
- 46.
Winnick, M.J.; Chamberlain, C.P.; Caves, J.K. Quantifying the isotopic ‘continental effect’. Earth Planet. Sci. Lett. 2014, 406, 123–133.
- 47.
Aggarwal, P.; Gibson, J.J.; Froehlich, K.; et al. Isotopic evidence for climatic conditions on southeast Asia at the last glacial maximum. In Conference: Study of Environmental Change using Isotope Techniques, Vienna, Austria; IAEA C&S Papers Series 13/P; IAEA: Vienna, Austria, 1993; pp. 28–40.
- 48.
Dansgaard,W. Stable isotopes in precipitation. Tellus 1964, 16, 436–468.
- 49.
Zheng, L.; Jiang, C.; Chen, X. Combining hydrochemistry and hydrogen and oxygen stable isotopes to reveal the influence of human activities on surface water quality in Chaohu Lake Basin. J. Environ. Manag. 2022, 312, 114933. https://doi.org/10.1016/j.jenvman.2022.114933
- 50.
Shilla, D.J.; Tsuchiya, M.; Shilla, D.A. Terrigenous nutrient and organic matter in a subtropical river estuary, Okinawa, Japan: Origin, distribution and pattern across the estuarine salinity gradient. Chem. Ecol. 2011, 27, 523–542. https://doi.org/10.1080/02757540.2011.600831
- 51.
Akita, L.G.; Laudien, J.; Nyarko, E. Geochemical contamination in the Densu Estuary, Gulf of Guinea, Ghana. Environ. Sci. Pollut. Res. 2020, 27, 42530–42555. https://doi.org/10.1007/s11356-020-10035-4
- 52.
Tang, Z.; Liu, X.; Niu, X. Ecological risk assessment of aquatic organisms induced by heavy metals in the estuarine waters of the Pearl River. Sci. Rep. 2023, 13, 9145. https://doi.org/10.1038/s41598-023-35798-x
- 53.
Sultan, M.B.; Anik, A.H.; Rahman, Md.M. Emerging contaminants and their potential impacts on estuarine ecosystems: Are we are of it? Mar. Pollut. Bull. 2024, 199, 115982. https://doi.org/10.1016/j.marpolbul.2023.115982
- 54.
Basraoui, N-e.; Ben-tahar, R.; Deliege, J-F. Potentially toxic elements contamination and ecological risk assessment in surface sediments of Moulouya Estuary (Northeastern, Morocco). Sci. Afr. 2024, 24, e02295. https://doi.org/10.1016/j.sciaf.2024.e02295

This work is licensed under a Creative Commons Attribution 4.0 International License.


