Chuanxiong, one of the traditional Chinese medicines (TCM), was first documented in the Tang dynasty to promote blood circulation and remove blood stasis. Ligusticum chuanxiong Hort was shown as the most effective portion of chuanxiong. Later chemical analysis revealed that the main chemical component of ligusticum chuanxiong Hort is tetramethylpyrazine. Since then, numerous explorations have been made to examine the efficiency of tetramethylpyrazine in treating different diseases and understand the underlying mechanisms of its action. Like Chuanxiong, ligustrazine (Chuan Xiong Qin) improved the functions of the circulatory and nervous systems. Ligustrazine (Chuan Xiong Qin) was also used in combination with other medicines to achieve better effects on improving cardiovascular health or alleviating the adverse effects of chemotherapies in both basic and clinical studies. The present review briefly summarizes the existing studies of the combination of ligustrazine (Chuan Xiong Qin) with other medicines in the treatment of cardiovascular diseases (CVDs) and provides valuable insights into the future research direction and better utilization of this drug.
- Open Access
- Review
The Therapeutic Effects of Ligustrazine in Combination with Other Drugs in Cardiovascular Diseases
- Peihua Dong,
- Yu Huang,
- Yujie Pu *
Author Information
Received: 29 Dec 2022 | Accepted: 28 Jan 2023 | Published: 10 Feb 2023
Abstract
Graphical Abstract
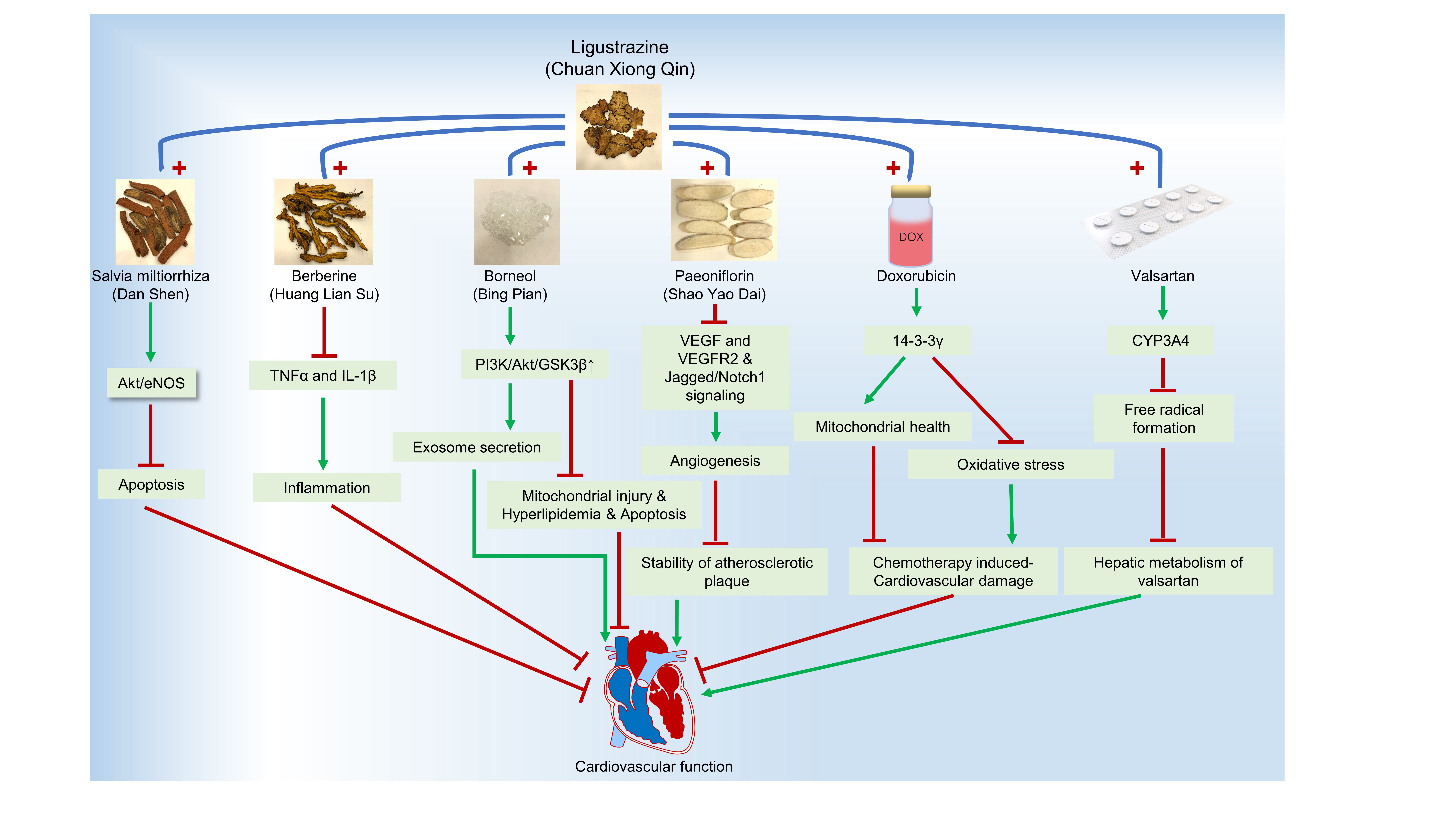
Keywords
Ligustrazine (Chuan Xiong Qin) | Salvia miltiorrhiza (Dan Shen) | Berberine (Huang Lian Su) | Paeoniflorin (Shao Yao Dai) | Suxiao jiuxin pill | Valsartan | Doxorubicin | Cardiovascular diseases
References
- 1.Zhang Z.G.; Zhang X.L.; Wang X.Y.; et al. Inhibition of acid sensing ion channel by ligustrazine on angina model in rat. Am. J. Transl. Res., 2015, 7(10): 1798-1811.
- 2.Lin J.G.; Wang Q.Q.; Zhou S.M.; et al. Tetramethylpyrazine: a review on its mechanisms and functions. Biomed. Pharmacother., 2022, 150: 113005.
- 3.Jiang R.D.; Xu J.Q.; Zhang Y.Z.; et al. Ligustrazine alleviates psoriasis-like inflammation through inhibiting TRAF6/c-JUN/NFκB signaling pathway in keratinocyte. Biomed. Pharmacother., 2022, 150: 113010.
- 4.Zhao T.F.; Fu Y.X.; Sun H.; et al. Ligustrazine suppresses neuron apoptosis via the Bax/Bcl-2 and caspase-3 pathway in PC12 cells and in rats with vascular dementia. IUBMB life, 2018, 70(1): 60-70.
- 5.Guo M.; Liu Y.; Shi D.Z. Cardiovascular actions and therapeutic potential of tetramethylpyrazine (active component isolated from Rhizoma Chuanxiong): roles and mechanisms. BioMed Res. Int., 2016, 2016: 2430329.
- 6.Meng D.M.; Lu H.Y.; Huang S. S.; et al. Comparative pharmacokinetics of tetramethylpyrazine phosphate in rat plasma and extracellular fluid of brain after intranasal, intragastric and intravenous administration. Acta Pharm. Sin. B, 2014, 4(1): 74-78.
- 7.Kücükköylü S.; Rump L.C. [Cardiovascular morbidity and mortality in renal diseases]. Dtsch. Med. Wochenschr., 2013, 138(14): 721-724.
- 8.Cai T.; Abel L.; Langford O.; et al. Associations between statins and adverse events in primary prevention of cardiovascular disease: systematic review with pairwise, network, and dose-response meta-analyses. BMJ, 2021, 374: n1537.
- 9.Sridharan K.; Sequeira R.P. Drugs for treating severe hypertension in pregnancy: a network meta-analysis and trial sequential analysis of randomized clinical trials. Br. J. Clin. Pharmacol., 2018, 84(9): 1906-1916.
- 10.Murphy E.; Curneen J.M.G.; McEvoy J.W. Aspirin in the modern era of cardiovascular disease prevention. Methodist DeBakey Cardiovascular Journal, 2021, 17(4): 36-47.
- 11.Huang W. D.; Yang Y. F.; Zeng Z.; et al. Effect of Salvia miltiorrhiza and ligustrazine injection on myocardial ischemia/reperfusion and hypoxia/reoxygenation injury. Mol. Med. Rep., 2016, 14(5): 4537-4544.
- 12.Zhang Y.; Ma X.J.; Guo C.Y.; et al. Pretreatment with a combination of ligustrazine and berberine improves cardiac function in rats with coronary microembolization. Acta Pharmacol. Sin., 2016, 37(4): 463-472.
- 13.Yuan R.; Shi W.L.; Xin Q.Q.; et al. Tetramethylpyrazine and paeoniflorin inhibit oxidized LDL-induced angiogenesis 6 of 7 in human umbilical vein endothelial cells via VEGF and notch pathways. Evidence-Based Complementary Altern. Med., 2018, 2018: 3082507.
- 14.Guo Q.X.; Zhang J.; Li Y.Q.; et al. Study on anti-atherosclerotic effect of Suxiao Jiuxin Pill and its mechanism. Afr. J. Tradit., Complementary Altern. Med., 2013, 11(1): 97-102.
- 15.Li Y. P.; Ruan X. F.; Chen T. J.; et al. Anti-apoptotic effect of Suxiao Jiuxin Pills against hypoxia-induced injury through PI3K/Akt/GSK3β pathway in HL-1 cardiomyocytes. J. Chin. Med. Assoc., 2018, 81(9): 816-824.
- 16.Liu Y.; Zhang J.Q.; Wu D.; et al. Pharmacokinetic interaction study between ligustrazine and valsartan in rats and its potential mechanism. Pharm. Biol., 2020, 58(1): 1290-1293.
- 17.Yang B.; Li H.W.; Qiao Y.; et al. Tetramethylpyrazine attenuates the endotheliotoxicity and the mitochondrial dysfunction by doxorubicin via 14-3-3γ/Bcl-2. Oxid. Med. Cell. Longevity, 2019, 2019: 5820415.
- 18.Ren J.; Fu L.; Nile S.H.; et al. Salvia miltiorrhiza in treating cardiovascular diseases: a review on its pharmacological and clinical applications. Front. Pharmacol., 2019, 10: 753.
- 19.Wang L.L.; Ma R.F.; Liu C.Y.; et al. Salvia miltiorrhiza: a potential red light to the development of cardiovascular diseases. Curr. Pharm. Des., 2017, 23(7): 1077-1097.
- 20.Hung Y. C.; Tseng Y. J.; Hu W. L.; et al. Demographic and prescribing patterns of Chinese herbal products for individualized therapy for ischemic heart disease in Taiwan: population-based study. PLoS One, 2015, 10(8): e0137058.
- 21.Hung I.L.; Hung Y.C.; Wang L.Y.; et al. Chinese herbal products for ischemic stroke. Am. J. Chin. Med., 2015, 43(7): 1365-1379.
- 22.Zhu T.; Wang L.; Feng Y. C.; et al. Classical active ingredients and extracts of Chinese herbal medicines: pharmacokinetics, pharmacodynamics, and molecular mechanisms for ischemic stroke. Oxid. Med. Cell. Longevity, 2021, 2021: 8868941.
- 23.Kim J. S.; Lee J. H.; Hong S.M.; et al. Salvia miltiorrhiza prevents methylglyoxal-induced glucotoxicity via the regulation of apoptosis-related pathways and the glyoxalase system in human umbilical vein endothelial cells. Biol. Pharm. Bull., 2022, 45(1): 51-62.
- 24.Rascio F.; Spadaccino F.; Rocchetti M.T.; et al. The pathogenic role of PI3K/AKT pathway in cancer onset and drug resistance: an updated review. Cancers, 2021, 13(16): 3949.
- 25.Feng X. J.; Sureda A.; Jafari S.; et al. Berberine in cardiovascular and metabolic diseases: from mechanisms to therapeutics. Theranostics, 2019, 9(7): 1923-1951.
- 26.Pang B.; Yu X.T.; Zhou Q.; et al. Effect of Rhizoma coptidis (Huang Lian) on treating diabetes mellitus. Evidence-Based Complementary Altern. Med., 2015, 2015: 921416.
- 27.Rui R.; Yang H.L.; Liu Y.K.; et al. Effects of berberine on atherosclerosis. Front. Pharmacol., 2021, 12: 764175.
- 28.Abdulredha A.; Abosaooda M.; Al-Amran F.; et al. Berberine protests the heart from ischemic reperfusion injury via interference with oxidative and inflammatory pathways. Med Arch., 2021, 75(3): 174-179.
- 29.Zhang J.; Huang L. L.; Shi X.; et al. Metformin protects against myocardial ischemia-reperfusion injury and cell pyroptosis via AMPK/NLRP3 inflammasome pathway. Aging, 2020, 12(23): 24270-24287.
- 30.Yang Q.; Huang D. D.; Li D. G.; et al. Tetramethylpyrazine exerts a protective effect against injury from acute myocardial ischemia by regulating the PI3K/Akt/GSK-3β signaling pathway. Cell. Mol. Biol. Lett., 2019, 24: 17.
- 31.Jiao F.; Varghese K.; Wang S. X.; et al. Recent insights into the protective mechanisms of paeoniflorin in neurological, cardiovascular, and renal diseases. J. Cardiovasc. Pharmacol., 2021, 77(6): 728-734.
- 32.Ngo T.; Kim K.; Bian Y.Y.; et al. Antithrombotic effects of paeoniflorin from Paeonia suffruticosa by selective inhibition on shear stress-induced platelet aggregation. Int. J. Mol. Sci., 2019, 20(20): 5040.
- 33.Chen H.W.; Dong Y.; He X.H.; et al. Paeoniflorin improves cardiac function and decreases adverse postinfarction left ventricular remodeling in a rat model of acute myocardial infarction. Drug Des., Dev. Ther., 2018, 12: 823-836.
- 34.Fan X.W.; Wu J.T.; Yang H.T.; et al. Paeoniflorin blocks the proliferation of vascular smooth muscle cells induced by platelet‑derived growth factor‑BB through ROS mediated ERK1/2 and p38 signaling pathways. Mol. Med. Rep., 2018, 17(1): 1676-1682.
- 35.Jiang J.T.; Dong C.J.; Zhai L.; et al. Paeoniflorin suppresses TBHP-induced oxidative stress and apoptosis in human umbilical vein endothelial cells via the Nrf2/HO-1 signaling pathway and improves skin flap survival. Front. Pharmacol., 2021, 12: 735530.
- 36.Chen Q.; Zhang D.N.; Bi Y.H.; et al. The protective effects of liguzinediol on congestive heart failure induced by myocardial infarction and its relative mechanism. Chin. Med., 2020, 15: 63.
- 37.Bai X.Y.; Zhang P.; Yang Q.; et al. Suxiao jiuxin pill induces potent relaxation and inhibition on contraction in human artery and the mechanism. Evidence-Based Complementary Altern. Med., 2014, 2014: 956924.
- 38.Ruan X.F.; Ju C.W.; Shen Y.; et al. Suxiao Jiuxin pill promotes exosome secretion from mouse cardiac mesenchymal stem cells in vitro. Acta Pharmacol. Sin., 2018, 39(4): 569-578.
- 39.Ruan X.F.; Chen T.J.; Wang X.L.; et al. Suxiao Jiuxin Pill protects cardiomyocytes against mitochondrial injury and alters gene expression during ischemic injury. Exp. Ther. Med., 2017, 14(4): 3523-3532.
- 40.Ren L.; Wang J.; Feng L.; et al. Efficacy of suxiao jiuxin pill on coronary heart disease: a Meta-Analysis of randomized controlled trials. Evidence-Based Complementary Altern. Med., 2018, 2018: 9745804.
- 41.Shen Z.J.; Chen T.J.; Deng B.; et al. Effects on Suxiao Jiuxin Pills in the treatment of patients with acute coronary syndrome undergoing early percutaneous coronary intervention: a multicenter randomized double-blind placebocontrolled trial. J. Altern. Complementary Med., 2020, 26(11): 1055-1063.
- 42.Lee S.; Oh J.; Kim H.; et al. Sacubitril/valsartan in patients with heart failure with reduced ejection fraction with endstage of renal disease. ESC heart failure, 2020, 7(3): 1125-1129.
- 43.Hermida R.C.; Calvo C.; Ayala D.E.; et al. Administration time-dependent effects of valsartan on ambulatory blood pressure in hypertensive subjects. Hypertension, 2003, 42(3): 283-290.
- 44.Ho C.Y.; Day S.M.; Axelsson A.; et al. Valsartan in early-stage hypertrophic cardiomyopathy: a randomized phase 2 trial. Nat. Med., 2021, 27(10): 1818-1824.
- 45.Mann D.L.; Greene S. J.; Givertz M.M.; et al. Sacubitril/valsartan in advanced heart failure with reduced ejection fraction: rationale and design of the LIFE trial. JACC: Heart Failure, 2020, 8(10): 789-799.
- 46.Qin L.L.; Deng S.; Zhang Z.; et al. [Protective effect of valsartan or/and ligustrazine on hippocampal neuronal loss in rats with vascular dementia]. Sichuan Da Xue Xue Bao Yi Xue Ban, 2011, 42(1): 56-60, 100.
- 47.Hiensch A. E.; Bolam K. A.; Mijwel S.; et al. Doxorubicin-induced skeletal muscle atrophy: elucidating the underlying molecular pathways. Acta Physiol., 2020, 229(2): e13400.
- 48.Catanzaro M.P.; Weiner A.; Kaminaris A.; et al. Doxorubicin-induced cardiomyocyte death is mediated by unchecked mitochondrial fission and mitophagy. FASEB J., 2019, 33(10): 11096-11108.
- 49.Kalyanaraman B. Teaching the basics of the mechanism of doxorubicin-induced cardiotoxicity: have we been barking up the wrong tree? Redox Biol., 2020, 29: 101394.
- 50.De Souza C.A.; Simões R.; Borges K.B.G.; et al. Arterial stiffness use for early monitoring of cardiovascular adverse events due to anthracycline chemotherapy in breast cancer patients. A pilot study. Arq. Bras. Cardiol., 2018, 111(5): 721-728.
- 51.Christidi E.; Brunham L.R. Regulated cell death pathways in doxorubicin-induced cardiotoxicity. Cell Death Dis., 2021, 12(4):339.
- 52.Chen X.Y.; Peng X. P.; Luo Y.; et al. Quercetin protects cardiomyocytes against doxorubicin-induced toxicity by suppressing oxidative stress and improving mitochondrial function via 14-3-3γ. Toxicol. Mech. Methods, 2019, 29 (5): 344-354.
- 53.He H.; Luo Y.; Qiao Y.; et al. Curcumin attenuates doxorubicin-induced cardiotoxicity via suppressing oxidative stress and preventing mitochondrial dysfunction mediated by 14-3-3γ. Food Funct., 2018, 9(8): 4404-4418.
How to Cite
Dong, P.; Huang, Y.; Pu, Y. The Therapeutic Effects of Ligustrazine in Combination with Other Drugs in Cardiovascular Diseases. International Journal of Drug Discovery and Pharmacology 2023, 2 (1), 11–17. https://doi.org/10.53941/ijddp.0201005.
RIS
BibTex
Copyright & License

Peihua Dong, Yu Huang, Yujie Pu
This work is licensed under a Creative Commons Attribution 4.0 International License.
Contents
References


