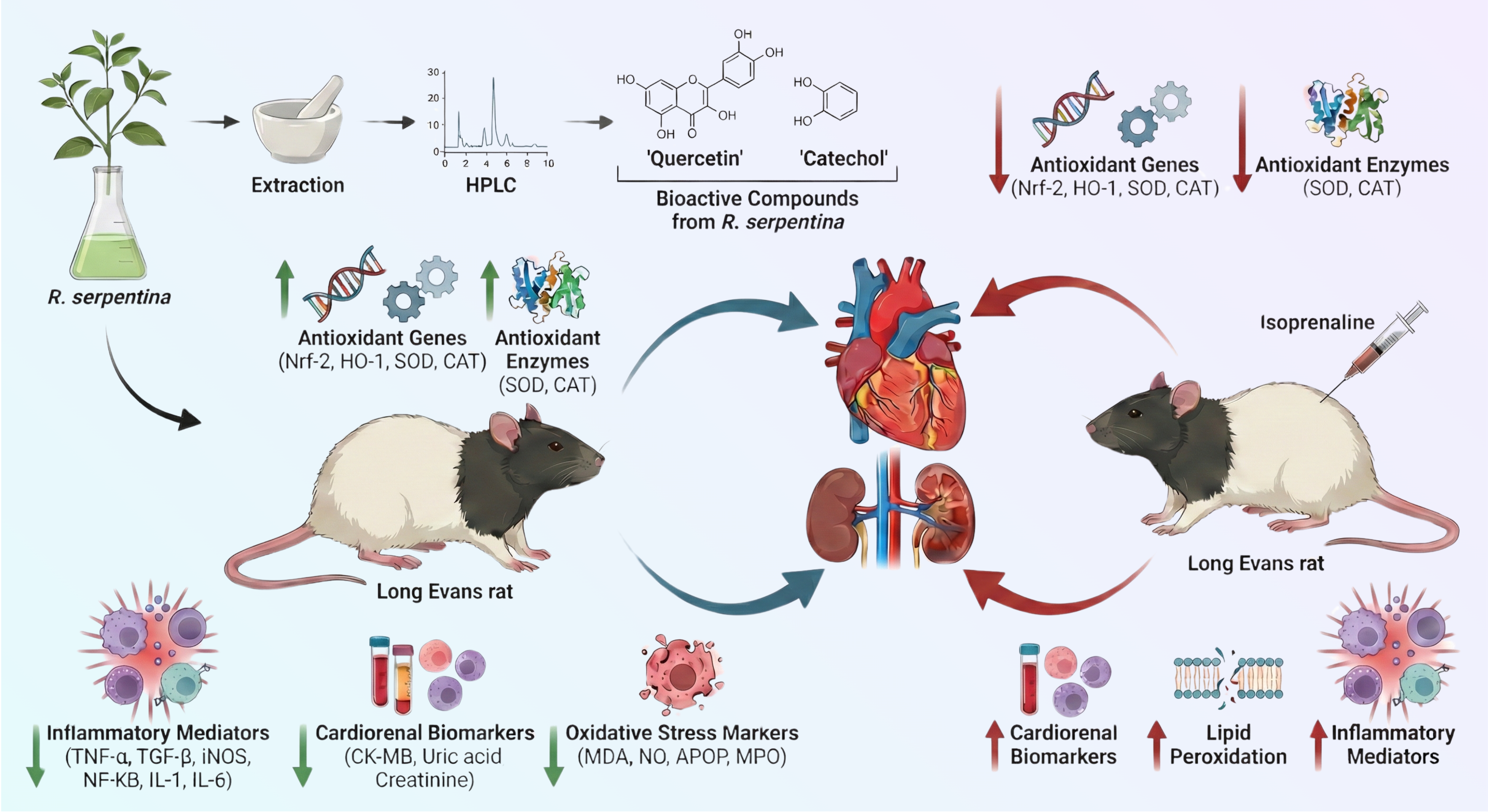
This study examined the protective effects of Rauwolfia serpentina root extract against isoprenaline (ISO)-induced oxidative stress, fibrosis, and cardiac injury in rats. ISO (50 mg/kg) was administered subcutaneously twice weekly. Biochemical markers, including uric acid, creatinine, CK-MB, MDA, NO, AOPP, and antioxidant enzymes, were evaluated, alongside histopathological analysis. ISO increased oxidative stress, inflammation, and cardiac damage, while R. serpentina treatment significantly reduced uric acid, creatinine, CK-MB, MDA, NO, AOPP, and myeloperoxidase levels, and restored antioxidant activity. Histology showed reduced fibrosis and inflammatory infiltration in treated rats. Gene expression analysis revealed downregulation of NF-κB, TNF-α, and IL-6, along with activation of the Nrf2-HO-1 pathway. Network pharmacology identified six key polyphenols in R. serpentina interacting with 141 cardiac hypertrophy-related proteins. Overall, R. serpentina mitigates ISO-induced cardiac and renal damage by reducing oxidative stress, inflammation, and fibrosis.
- Open Access
- Article
Rauwolfia serpentina Treatment Prevents Oxidative Stress, Inflammation, and Fibrosis in the Heart and Kidneys of Isoprenaline-Administered Rats
- Kamrun Nahar Ela 1,
- Mirza Alimullah 1,
- Asif Ul Haque Shuvo 1,
- Puspa Sornakar 1,
- Shamima Sultana 1,
- Md. Junaeid Rahman 1,
- Md. Sakil Amin 2,
- Khondoker Shahin Ahmed 3,
- Hemayet Hossain 3,
- Mohammad Borhan Uddin 1,
- Ferdous Khan 1,
- Md. Ashraful Alam 1,*,
- Nusrat Subhan 1,*
Author Information
Received: 30 Dec 2025 | Revised: 11 Feb 2026 | Accepted: 25 Feb 2026 | Published: 24 Mar 2026
Abstract
Graphical Abstract
Keywords
Rauwolfia serpentina | myocardial damage | isoprenaline | fibrosis | inflammation
References
- 1.
Szlagor, M.; Dybiec, J.; Młynarska, E.; et al. Chronic kidney disease as a comorbidity in heart failure. Int. J. Mol. Sci. 2023, 24, 2988.
- 2.
Liu, M.; Li, X.-C.; Lu, L.; et al. Cardiovascular disease and its relationship with chronic kidney disease. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 2918–2926.
- 3.
Schefold, J.C.; Filippatos, G.; Hasenfuss, G.; et al. Heart failure and kidney dysfunction: Epidemiology, mechanisms and management. Nat. Rev. Nephrol. 2016, 12, 610–623.
- 4.
Lim, S.H.; Lee, J. Xyloglucan intake attenuates myocardial injury by inhibiting apoptosis and improving energy metabolism in a rat model of myocardial infarction. Nutr. Res. 2017, 45, 19–29.
- 5.
Hong, K.N.; Fuster, V.; Rosenson, R.S.; et al. How low to go with glucose, cholesterol, and blood pressure in primary prevention of CVD. J. Am. Coll. Cardiol. 2017, 70, 2171–2185.
- 6.
Hurtubise, J.; McLellan, K.; Durr, K.; et al. The different facets of dyslipidemia and hypertension in atherosclerosis. Curr. Atheroscler. Rep. 2016, 18, 82.
- 7.
Di Domenico, F.; Tramutola, A.; Butterfield, D.A. Role of 4-hydroxy-2-nonenal (HNE) in the pathogenesis of alzheimer disease and other selected age-related neurodegenerative disorders. Free Radic. Biol. Med. 2017, 111, 253–261.
- 8.
Zhang, Y.; Martin, S. Redox proteins and radiotherapy. Clin. Oncol. 2014, 26, 289–300.
- 9.
Zhou, S.; Sun, W.; Zhang, Z.; et al. The role of Nrf2-mediated pathway in cardiac remodeling and heart failure. Oxidative Med. Cell. Longev. 2014, 2014, 260429.
- 10.
Wang, C.; Pei, Y.-Y.; Ma, Y.-H.; et al. Risk factors for acute kidney injury in patients with acute myocardial infarction. Chin. Med. J. 2019, 132, 1660–1665.
- 11.
Chiuariu, T.; Șalaru, D.; Ureche, C.; et al. Cardiac and Renal Fibrosis, the Silent Killer in the Cardiovascular Continuum: An Up-to-Date. J. Cardiovasc. Dev. Dis. 2024, 11, 62.
- 12.
Araujo, M.; Wilcox, C.S. Oxidative stress in hypertension: Role of the kidney. Antioxid. Redox Signal. 2014, 20, 74–101.
- 13.
AlQudah, M.; Hale, T.M.; Czubryt, M.P. Targeting the renin-angiotensin-aldosterone system in fibrosis. Matrix Biol. 2020, 91, 92–108.
- 14.
Singh, P.K.; Gari, M.; Choudhury, S.; et al. Oleic acid prevents isoprenaline-induced cardiac injury: Effects on cellular oxidative stress, inflammation and histopathological alterations. Cardiovasc. Toxicol. 2020, 20, 28–48.
- 15.
Huang, H.; Geng, Q.; Yao, H.; et al. Protective effect of scutellarin on myocardial infarction induced by isoprenaline in rats. Iran. J. Basic. Med. Sci. 2018, 21, 267.
- 16.
Manolis, A.S.; Manolis, A.A.; Manolis, T.A.; et al. Mitochondrial dysfunction in cardiovascular disease: Current status of translational research/clinical and therapeutic implications. Med. Res. Rev. 2021, 41, 275–313.
- 17.
Mukherjee, E.; Gantait, S.; Kundu, S.; et al. Biotechnological interventions on the genus Rauvolfia: Recent trends and imminent prospects. Appl. Microbiol. Biotechnol. 2019, 103, 7325–7354.
- 18.
Anavkar, A.; Patel, N.; Ali, A.; et al. Plantago ovata (Isabgol) and Rauvolfia serpentina (Indian Snakeroot). In Herbs, Shrubs, and Trees of Potential Medicinal Benefits; CRC Press: Boca Raton, FL, USA, 2022; pp. 235–260.
- 19.
Sen, S.; Chakraborty, R. Toward the integration and advancement of herbal medicine: A focus on traditional Indian medicine. Bot. Targets Ther. 2015, 5, 33–44.
- 20.
Wu, F.; Kerčmar, P.; Zhang, C.; et al. Sarpagan-Ajmalan-Type Indoles: Biosynthesis, structural biology, and chemo-enzymatic significance. Alkaloids Chem. Biol. 2016, 76, 1–61.
- 21.
Perk, A.A.; Shatynska-Mytsyk, I.; Gerçek, Y.C.; et al. Rutin mediated targeting of signaling machinery in cancer cells. Cancer Cell Int. 2014, 14, 124.
- 22.
Ciumărnean, L.; Milaciu, M.V.; Runcan, O.; et al. The effects of flavonoids in cardiovascular diseases. Molecules 2020, 25, 4320.
- 23.
Sánchez, M.; Romero, M.; Gómez-Guzmán, M.; et al. Cardiovascular effects of flavonoids. Curr. Med. Chem. 2019, 26, 6991–7034.
- 24.
Chaudhary, R.; Singh, B.; Chhillar, A. Ethnomedicinal Importance of Rauvolfia serpentina L. Benth. Ex Kurz in the Prevention and Treatment of Diseases. Nat. Prod. Res. Rev. 2016, 3, 305–326.
- 25.
Alshahrani, M.Y.; Rafi, Z.; Alabdallah, N.M.; et al. A comparative antibacterial, antioxidant, and antineoplastic potential of Rauwolfia serpentina (L.) leaf extract with its biologically synthesized gold nanoparticles (R-AuNPs). Plants 2021, 10, 2278.
- 26.
Gupta, A.K.; Irchhaiya, R.; Misra, C. Free radical scavenging activity of Rauwolfia serpentina rhizome against CCl4 induced liver injury. Int. J. Pharm. 2015, 2, 123–126.
- 27.
Shah, S.M.A.; Naqvi, S.A.R.; Munir, N.; et al. Antihypertensive and antihyperlipidemic activity of aqueous methanolic extract of Rauwolfia serpentina in albino rats. Dose-Response 2020, 18, 1559325820942077.
- 28.
Azmi, M.B.; Qureshi, S.A. Rauwolfia serpentina improves altered glucose and lipid homeostasis in fructose-induced type 2 diabetic mice. Pak. J. Pharm. Sci. 2016, 29, 1619–1624.
- 29.
Rohela, G.K.; Bylla, P.; Korra, R.; et al. Phytochemical screening and antimicrobial activity of leaf, stem, root and their callus extracts in Rauwolfia tetraphylla. Int. J. Agric. Biol. 2016, 18, 521–528.
- 30.
Hossain, H.; Rahman, S.E.; Akbar, P.N.; et al. HPLC profiling, antioxidant and in vivo anti-inflammatory activity of the ethanol extract of Syzygium jambos available in Bangladesh. BMC Res. Notes 2016, 9, 191.
- 31.
Mamun, F.; Rahman, M.M.; Zamila, M.; et al. Polyphenolic compounds of litchi leaf augment kidney and heart functions in 2K1C rats. J. Funct. Foods 2020, 64, 103662.
- 32.
Clark, J.D.; Gebhart, G.F.; Gonder, J.C.; et al. The 1996 guide for the care and use of laboratory animals. ILAR J. 1997, 38, 41–48.
- 33.
Lasker, S.; Rahman, M.M.; Parvez, F.; et al. High-fat diet-induced metabolic syndrome and oxidative stress in obese rats are ameliorated by yogurt supplementation. Sci. Rep. 2019, 9, 20026.
- 34.
Rahman, M.M.; Alimullah, M.; Yasmin, T.; et al. Cardioprotective action of apocynin in isoproterenol-induced cardiac damage is mediated through Nrf-2/HO-1 signaling pathway. Food Sci. Nutr. 2024, 12, 9108–9122. https://doi.org/10.1002/fsn3.4465.
- 35.
Jahan, I.; Hassan, S.M.H.; Alimullah, M.; et al. Evaluation of fenugreek (Trigonella foenum-graecum L.) powder supplementation on metabolic syndrome, oxidative stress and inflammation in high fat diet fed rats. Pharmacol. Res. Nat. Prod. 2024, 5, 100116. https://doi.org/10.1016/j.prenap.2024.100116.
- 36.
Tracey, W.R.; Tse, J.; Carter, G. Lipopolysaccharide-induced changes in plasma nitrite and nitrate concentrations in rats and mice: Pharmacological evaluation of nitric oxide synthase inhibitors. J. Pharmacol. Exp. Ther. 1995, 272, 1011–1015.
- 37.
Zahan, T.; Alimullah, M.; Jahan, I.; et al. Baccaurea ramiflora fruit peel powder supplementation prevented inflammatory cell infiltration, oxidative stress, and fibrosis in carbon tetrachloride (CCl4) administered ovariectomized rats. Phytomed Plus 2025, 5, 100719. https://doi.org/10.1016/j.phyplu.2024.100719.
- 38.
Witko-Sarsat, V.; Friedlander, M.; Capeillère-Blandin, C.; et al. Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 1996, 49, 1304–1313.
- 39.
Jahan, I.; Shuvo, A.U.H.; Alimullah, M.; et al. Purple potato extract modulates fat metabolizing genes expression, prevents oxidative stress, hepatic steatosis, and attenuates high-fat diet-induced obesity in male rats. PLoS ONE 2025, 20, e0318162. https://doi.org/10.1371/journal.pone.0318162.
- 40.
Khan, S.; Rahman, M.M.; Kabir, F.; et al. Trichosanthes dioica Roxb. prevents hepatic inflammation and fibrosis in CCl4-induced ovariectomized rats. Clin. Nutr. Exp. 2020, 33, 1–17.
- 41.
Alimullah, M.; Rahman, N.; Sornaker, P.; et al. Evaluation of Terminalia arjuna Bark Powder Supplementation on Isoprenaline-Induced Oxidative Stress and Inflammation in the Heart of Long Evans Rats, Understanding the Molecular Mechanism of This Old Medicinal Plant. J. Med. Nat. Prod. 2024, 1, 100004.
- 42.
Alimullah, M.; Shuvo, A.U.H.; Jahan, I.; et al. Evaluation of the modulating effect of epidermal growth factor receptor inhibitor cetuximab in carbon-tetrachloride induce hepatic fibrosis in rats. Biochem. Biophys. Rep. 2024, 38, 101689. https://doi.org/10.1016/j.bbrep.2024.101689.
- 43.
Bradley, P.P.; Christensen, R.D.; Rothstein, G. Cellular and extracellular myeloperoxidase in pyogenic inflammation. Blood 1982, 60, 618–622.
- 44.
Shuvo, A.U.H.; Alimullah, M.; Jahan, I.; et al. Evaluation of Xanthine Oxidase Inhibitors Febuxostat and Allopurinol on Kidney Dysfunction and Histological Damage in Two-Kidney, One-Clip (2K1C) Rats. Scientifica 2025, 2025, 7932075. https://doi.org/10.1155/sci5/7932075.
- 45.
Jollow, D.; Mitchell, J.; Zampaglione, N.a.; et al. Bromobenzene-induced liver necrosis. Protective role of glutathione and evidence for 3, 4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacology 1974, 11, 151–169.
- 46.
Kim, S.; Chen, J.; Cheng, T.; et al. PubChem 2023 update. Nucleic Acids Res. 2023, 51, D1373–D1380.
- 47.
Kim, S.; Thiessen, P.A.; Bolton, E.E.; et al. PubChem substance and compound databases. Nucleic Acids Res. 2016, 44, D1202–D1213.
- 48.
Kong, X.; Liu, C.; Zhang, Z.; et al. BATMAN-TCM 2.0: An enhanced integrative database for known and predicted interactions between traditional Chinese medicine ingredients and target proteins. Nucleic Acids Res. 2024, 52, D1110–D1120.
- 49.
Liu, Z.; Guo, F.; Wang, Y.; et al. BATMAN-TCM: A bioinformatics analysis tool for molecular mechANism of traditional Chinese medicine. Sci. Rep. 2016, 6, 21146.
- 50.
Gallo, K.; Goede, A.; Preissner, R.; et al. SuperPred 3.0: Drug classification and target prediction—A machine learning approach. Nucleic Acids Res. 2022, 50, W726–W731.
- 51.
Nickel, J.; Gohlke, B.-O.; Erehman, J.; et al. SuperPred: Update on drug classification and target prediction. Nucleic Acids Res. 2014, 42, W26–W31.
- 52.
Min, S.; Lee, B.; Yoon, S. TargetNet: Functional microRNA target prediction with deep neural networks. Bioinformatics 2022, 38, 671–677.
- 53.
Yao, Z.-J.; Dong, J.; Che, Y.-J.; et al. TargetNet: A web service for predicting potential drug–target interaction profiling via multi-target SAR models. J. Comput.-Aided Mol. Des. 2016, 30, 413–424.
- 54.
Druzhilovskiy, D.; Filimonov, D.; Rudik, A.; et al. Way2Drug Platform: From Biological Activity Prediction to Systems Pharmacology. In Proceedings of the 5th Belgrade Bioinformatics Conference, Serbia, Belgrade, 17–20 June 2024; p. 8.
- 55.
Druzhilovskiy, D.; Rudik, A.; Filimonov, D.; et al. Computational platform Way2Drug: From the prediction of biological activity to drug repurposing. Russ. Chem. Bull. 2017, 66, 1832–1841.
- 56.
Wang, Z.; Liang, L.; Yin, Z.; et al. Improving chemical similarity ensemble approach in target prediction. J. Cheminform. 2016, 8, 20.
- 57.
Gu, S.; Lai, L. Associating 197 Chinese herbal medicine with drug targets and diseases using the similarity ensemble approach. Acta Pharmacol. Sin. 2020, 41, 432–438.
- 58.
Amberger, J.S.; Bocchini, C.A.; Schiettecatte, F.; et al. OMIM. org: Online Mendelian Inheritance in Man (OMIM®), an online catalog of human genes and genetic disorders. Nucleic Acids Res. 2015, 43, D789–D798.
- 59.
Hamosh, A.; Scott, A.F.; Amberger, J.S.; et al. Online Mendelian Inheritance in Man (OMIM), a knowledgebase of human genes and genetic disorders. Nucleic Acids Res. 2005, 33, D514–D517.
- 60.
Amberger, J.S.; Bocchini, C.A.; Scott, A.F.; et al. OMIM. org: Leveraging knowledge across phenotype–gene relationships. Nucleic Acids Res. 2019, 47, D1038–D1043.
- 61.
Safran, M.; Rosen, N.; Twik, M.; et al. The genecards suite. In Practical Guide to Life Science Databases; Springer Nature: Singapore, 2021; pp. 27–56.
- 62.
Stelzer, G.; Rosen, N.; Plaschkes, I.; et al. The GeneCards suite: From gene data mining to disease genome sequence analyses. Curr. Protoc. Bioinform. 2016, 54, 1–30.
- 63.
Consortium, U. UniProt: A worldwide hub of protein knowledge. Nucleic Acids Res. 2019, 47, D506–D515.
- 64.
The UniProt Consortium. UniProt: The universal protein knowledgebase in 2021. Nucleic Acids Res. 2021, 49, D480–D489.
- 65.
Wang, Z.; Li, X.; Chen, H.; et al. Decreased HLF expression predicts poor survival in lung adenocarcinoma. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2021, 27, e929333-1.
- 66.
Zúñiga-Hernández, S.R.; García-Iglesias, T.; Macías-Carballo, M.; et al. A Bioinformatic Assay of Quercetin in Gastric Cancer. Int. J. Mol. Sci. 2024, 25, 7934.
- 67.
Shannon, P.; Markiel, A.; Ozier, O.; et al. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504.
- 68.
Liu, H.; Yang, S.; Chen, B.; et al. Integrating network pharmacology and molecular docking to explore the pharmacological mechanism of tanshinone IIA in improving chronic obstructive pulmonary disease. Medicine 2025, 104, e41638.
- 69.
Szklarczyk, D.; Nastou, K.; Koutrouli, M.; et al. The STRING database in 2025: Protein networks with directionality of regulation. Nucleic Acids Res. 2025, 53, D730–D737.
- 70.
Wahyuni, S.; Jamil, A.S.; Muchlisin, M.A. A Network Pharmacology of Camellia sinensis (Green Tea). Proc. Int. Pharm. Ulul Albab Conf. Semin. 2023, 216–224. https://doi.org/10.18860/planar.v3i0.2487.
- 71.
Ge, S.X.; Jung, D.; Yao, R. ShinyGO: A graphical gene-set enrichment tool for animals and plants. Bioinformatics 2020, 36, 2628–2629.
- 72.
Bisht, A.; Tewari, D.; Kumar, S.; et al. Network pharmacology-based approach to investigate the molecular targets and molecular mechanisms of Rosmarinus officinalis L. for treating aging-related disorders. Biogerontology 2024, 25, 793–808.
- 73.
Hedayati, N.; Yaghoobi, A.; Salami, M.; et al. Impact of polyphenols on heart failure and cardiac hypertrophy: Clinical effects and molecular mechanisms. Front. Cardiovasc. Med. 2023, 10, 1174816.
- 74.
Sun, G.; Qiu, Z.; Wang, W.; et al. Flavonoids extraction from propolis attenuates pathological cardiac hypertrophy through PI3K/AKT signaling pathway. Evid. Based Complement. Altern. Med. 2016, 2016, 6281376.
- 75.
Guan, P.; Sun, Z.-M.; Wang, N.; et al. Resveratrol prevents chronic intermittent hypoxia-induced cardiac hypertrophy by targeting the PI3K/AKT/mTOR pathway. Life Sci. 2019, 233, 116748.
- 76.
Zhou, Y.; Zhou, B.; Pache, L.; et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 2019, 10, 1523.
- 77.
Ren, S.; Shen, L.; Lin, S.; et al. Mechanistic analysis of resveratrol in cardiac hypertrophy by network pharmacology and animal experiments. Mol. Med. Rep. 2022, 26, 324.
- 78.
Wang, X.; Wang, Y.; Yuan, T.; et al. Network pharmacology provides new insights into the mechanism of traditional Chinese medicine and natural products used to treat pulmonary hypertension. Phytomedicine 2024, 135, 156062.
- 79.
Yu, W.; Weng, Y.; Wang, J.; et al. Network Pharmacology Approach and Partial Experimental Validation of Aidi Injection Solution for the Treatment of Colorectal Cancer. Nat. Prod. Commun. 2024, 19, 1934578X241239169.
- 80.
Peng, Y.; Zhu, G.; Ma, Y.; et al. Network Pharmacology–Based Prediction and Pharmacological Validation of Effects of Astragali Radix on Acetaminophen-Induced Liver Injury. Front. Med. 2022, 9, 697644.
- 81.
Yang, X.; Qin, Y.; Zhou, H. Deciphering the Pharmacological Mechanisms of Wen-Jing-Zhi-Tong Decoction in Treating Primary Dysmenorrhea by UPLC-Q-Exactive-Orbitrap-MS/MS with GC-MS and Network Pharmacology. Comb. Chem. High. Throughput Screen. 2025, 28, 1011–1025.
- 82.
Liu, M.; Zheng, L.; Zhang, Y.; et al. Mechanistic insights into pachymic acid’s action on triple-negative breast Cancer through TOP2A targeting. Sci. Rep. 2025, 15, 2856.
- 83.
Jiang, S.; Huang, C.; Wang, S.; et al. Network pharmacology-based strategy for predicting therapy targets of Citri Reticulatae Pericarpium on myocardial hypertrophy. BioMed Res. Int. 2022, 2022, 4293265.
- 84.
Wang, J.; Yang, Z.; Jiang, J.; et al. Systematically Investigating the Pharmacological Mechanism of Momordica grosvenori in the Treatment of Spinal Cord Injury by Network Pharmacology and Experimental Verification. Evid. Based Complement. Altern. Med. 2023, 2023, 1638966.
- 85.
Zhang, Z.; Wang, C. Exploring key genes and pathways of cardiac hypertrophy based on bioinformatics. Dis. Markers 2022, 2022, 2081590.
- 86.
Safran, M.; Dalah, I.; Alexander, J.; et al. GeneCards Version 3: The human gene integrator. Database 2010, 2010, baq020.
- 87.
Huang, X.; Huang, F.; Shu, H.; et al. Garlic’s anti-mastitis mechanism studied via network pharmacology & molecular docking. In Proceedings of the 2025 5th International Conference on Bioinformatics and Intelligent Computing, Shenyang, China, 10–12 January 2025; pp. 385–390.
- 88.
Kong, J.; Huang, X.; Yang, X.; et al. Network pharmacology-based prediction of potential targets of ethnic medicine Blumea balsamifera (L.) DC acting on anti-infammatory effect. J. Phys. Conf. Ser. 2020, 1486, 022032.
- 89.
Shi, X.; Luo, Y.; Yang, L.; et al. Protective effect of Gastrodia elata Blume in a Caenorhabditis elegans model of Alzheimer's disease based on network pharmacology. Biomed. Rep. 2023, 18, 37.
- 90.
Gong, K.; Yang, K.; Xie, T.; et al. Identification of circRNA-miRNA-mRNA regulatory network and its role in cardiac hypertrophy. PLoS ONE 2023, 18, e0279638.
- 91.
Chen, L.; Li, M.; Shen, M.; et al. Bioinformatics exploration of potential common therapeutic targets for systemic and pulmonary arterial hypertension-induced myocardial hypertrophy: Common therapeutic target for left and right ventricular hypertrophy. Acta Biochim. Biophys. Sin. 2023, 55, 831.
- 92.
Singh, M. Evaluating the therapeutic efficiency and drug targeting ability of alkaloids present in Rauwolfia serpentina. Int. J. Green Pharm. 2017, 11, 132–142.
- 93.
Ma, Y.-Q.; Zhang, M.; Sun, Z.-H.; et al. Identification of anti-gastric cancer effects and molecular mechanisms of resveratrol: From network pharmacology and bioinformatics to experimental validation. World J. Gastrointest. Oncol. 2024, 16, 493.
- 94.
Wijaya, P.; Tallei, T.E.; Tendean, L.E.N.; et al. Network Pharmacology Insights into Broccoli Microgreens for Prostate Cancer. Heca J. Appl. Sci. 2025, 3, 1–16.
- 95.
Li, S.; Fan, T.-P.; Jia, W.; et al. Network pharmacology in traditional Chinese medicine. Evid. -Based Complement. Altern. Med. Ecam 2014, 2014, 138460.
- 96.
Chang, C.; Jia, R.; Fang, B.; et al. Network pharmacological analysis and in vitro testing of the rutin effects on triple-negative breast cancer. Open Med. 2025, 20, 20241079.
- 97.
Liu, Y.; Li, Q.; Shao, C.; et al. Exploring the potential mechanisms of guanxinshutong capsules in treating pathological cardiac hypertrophy based on network pharmacology, computer-aided drug design, and animal experiments. ACS Omega 2024, 9, 18083–18098.
- 98.
Wu, Q.; Zhou, Q.; Wan, C.; et al. Mechanism Actions of Coniferyl Alcohol in Improving Cardiac Dysfunction in Renovascular Hypertension Studied by Experimental Verification and Network Pharmacology. Int. J. Mol. Sci. 2024, 25, 10063.
- 99.
Zhou, L.; Ding, H.-m.; Du, Y.; et al. Liquiritin ameliorates acute myocardial infarction via the COX-2/NLRP3 signaling pathway: Network pharmacology and experimental validation. Ital. J. Food Sci. 2024, 36, 1.
- 100.
Pathania, S.; Randhawa, V.; Bagler, G. Prospecting for novel plant-derived molecules of Rauvolfia serpentina as inhibitors of Aldose Reductase, a potent drug target for diabetes and its complications. PLoS ONE 2013, 8, e61327.
- 101.
Azmi, M.B.; Sultana, S.; Naeem, S.; et al. In silico investigation on alkaloids of Rauwolfia serpentina as potential inhibitors of 3-hydroxy-3-methyl-glutaryl-CoA reductase. Saudi J. Biol. Sci. 2021, 28, 731–737.
- 102.
Yang, K.; Shan, X.; Yang, S.; et al. Network pharmacology integrated with experimental validation to elucidate the mechanisms of action of the Guizhi-Gancao Decoction in the treatment of phenylephrine-induced cardiac hypertrophy. Pharm. Biol. 2024, 62, 456–471.
- 103.
Ramly, B.; Afiqah-Aleng, N.; Mohamed-Hussein, Z.-A. Protein–protein interaction network analysis reveals several diseases highly associated with polycystic ovarian syndrome. Int. J. Mol. Sci. 2019, 20, 2959.
- 104.
Xia, J.; Benner, M.J.; Hancock, R.E. NetworkAnalyst-integrative approaches for protein–protein interaction network analysis and visual exploration. Nucleic Acids Res. 2014, 42, W167–W174.
- 105.
Szklarczyk, D.; Gable, A.L.; Lyon, D.; et al. STRING v11: Protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, D607–D613.
- 106.
Rezaei-Tavirani, M.; Rezaei-Tavirani, S.; Mansouri, V.; et al. Protein-protein interaction network analysis for a biomarker panel related to human esophageal adenocarcinoma. Asian Pac. J. Cancer Prev. 2017, 18, 3357.
- 107.
Majeed, A.; Mukhtar, S. Protein–protein interaction network exploration using cytoscape. In Protein-Protein Interactions: Methods and Protocols; Springer: New York, NY, USA, 2023; pp. 419–427.
- 108.
Kardos, O.; London, A.; Vinkó, T. Stability of network centrality measures: A numerical study. Soc. Netw. Anal. Min. 2020, 10, 80.
- 109.
Sabah, L.; Şimşek, M. A new fast entropy-based method to generate composite centrality measures in complex networks. Concurr. Comput. Pract. Exp. 2023, 35, e7657.
- 110.
Mukhtar, M.F.; Anuar, S.H.H.; Abas, Z.A.; et al. Bibliometric Analysis of the Global Trend in Centrality Measures. Int. J. Acad. Res. Bus. Soc. Sci. 2024, 14, 1078–1098.
- 111.
Xiong, W.; Xie, L.; Zhou, S.; et al. The centrality of cancer proteins in human protein-protein interaction network: A revisit. Int. J. Comput. Biol. Drug Des. 2014, 7, 146–156.
- 112.
Giles, A.P.; Georgiou, O.; Dettmann, C.P. Betweenness centrality in dense random geometric networks. In Proceedings of the 2015 IEEE International Conference on Communications (ICC), London, UK, 8–12 June 2015; pp. 6450–6455.
- 113.
Ochieng, P.J.; Hussain, A.; Dombi, J.; et al. An efficient weighted network centrality approach for exploring mechanisms of action of the Ruellia herbal formula for treating rheumatoid arthritis. Appl. Netw. Sci. 2023, 8, 7.
- 114.
Chiranjeevi, M.; Dhuli, V.S.; Enduri, M.K.; et al. Icdc: Ranking influential nodes in complex networks based on isolating and clustering coefficient centrality measures. IEEE Access 2023, 11, 126195–126208.
- 115.
Nandini, Y.; Lakshmi, T.J.; Enduri, M.K.; et al. Link prediction in complex networks using average centrality-based similarity score. Entropy 2024, 26, 433.
- 116.
Parisutham, N. The Impact of Centrality Measures in Protein–Protein Interaction Networks: Tools, Databases, Challenges and Future Directions. J. Comput. Biophys. Chem. 2024, 23, 815–836.
- 117.
Madonna, R.; Geng, Y.-J.; Bolli, R.; et al. Co-activation of nuclear factor-κB and myocardin/serum response factor conveys the hypertrophy signal of high insulin levels in cardiac myoblasts. J. Biol. Chem. 2014, 289, 19585–19598.
- 118.
Jin, L.; Hou, P. Yixin-Fumai granules modulate autophagy through the PI3K/AKT/FOXO pathway and lead to amelioration of aging mice with sick sinus syndrome. Immun. Ageing 2024, 21, 46.
- 119.
Zhao, M.; Feng, L.; Li, W. Network Pharmacology and Experimental Verification: SanQi-DanShen Treats Coronary Heart Disease by Inhibiting the PI3K/AKT Signaling Pathway. Drug Des. Dev. Ther. 2024, 18, 4529–4550.
- 120.
Zhang, Y.; Zhang, X.; Zhang, X.; et al. Molecular targets and pathways contributing to the effects of wenxin keli on atrial fibrillation based on a network pharmacology approach. Evid. Based Complement. Altern. Med. 2020, 2020, 8396484.
- 121.
Chen, C.; Zou, L.-X.; Lin, Q.-Y.; et al. Resveratrol as a new inhibitor of immunoproteasome prevents PTEN degradation and attenuates cardiac hypertrophy after pressure overload. Redox Biol. 2019, 20, 390–401.
- 122.
Aoyagi, T.; Matsui, T. Phosphoinositide-3 kinase signaling in cardiac hypertrophy and heart failure. Curr. Pharm. Des. 2011, 17, 1818–1824.
- 123.
Zhao, A.; Zhang, X.; Hu, G.; et al. Novel Gene Signatures for Prostate Cancer Detection: Network Centralitybased Screening with Experimental Validation. Curr. Bioinform. 2023, 18, 842–852.
- 124.
Ni, X.; Bao, H.; Guo, J.; et al. Discussion on the mechanism of Danggui Sini decoction in treating diabetic foot based on network pharmacology and molecular docking and verification of the curative effect by meta-analysis. Front. Endocrinol. 2024, 15, 1347021.
- 125.
Azmi, M.B.; Qureshi, S.A. Methanolic root extract of Rauwolfia serpentina benth improves the glycemic, antiatherogenic, and cardioprotective indices in alloxan-induced diabetic mice. Adv. Pharmacol. Pharm. Sci. 2012, 2012, 376429.
- 126.
Pathania, S.; Ramakrishnan, S.M.; Randhawa, V.; et al. SerpentinaDB: A database of plant-derived molecules of Rauvolfia serpentina. BMC Complement. Altern. Med. 2015, 15, 262.
- 127.
Afsheen, N.; Jahan, N.; Ijaz, M.; et al. Cardioprotective and metabolomic profiling of selected medicinal plants against oxidative stress. Oxidative Med. Cell. Longev. 2018, 2018, 9819360.
- 128.
Netala, V.R.; Teertam, S.K.; Li, H.; et al. A comprehensive review of cardiovascular disease management: Cardiac biomarkers, imaging modalities, pharmacotherapy, surgical interventions, and herbal remedies. Cells 2024, 13, 1471.
- 129.
Ananta, M.F.; Oyshi, S.A.; Mim, M.M.A.; et al. Multipurpose Drug from Rouwolfia serpentina and Nigella sativa: A Herbal Approach to Treat Hypertension and Hyperlipidemia in Experimental Rodent Model. J. Complement. Altern. Med. Res. 2023, 24, 9–15.
- 130.
Piñero, J.; Bravo, À.; Queralt-Rosinach, N.; et al. DisGeNET: A comprehensive platform integrating information on human disease-associated genes and variants. Nucleic Acids Res. 2017, 45, D833–D839. https://doi.org/10.1093/nar/gkw943.
- 131.
Qin, W.; Cao, L.; Massey, I.Y. Role of PI3K/Akt signaling pathway in cardiac fibrosis. Mol. Cell. Biochem. 2021, 476, 4045–4059.
- 132.
Ghafouri-Fard, S.; Khanbabapour Sasi, A.; Hussen, B.M.; et al. Interplay between PI3K/AKT pathway and heart disorders. Mol. Biol. Rep. 2022, 49, 9767–9781.
- 133.
McLean, B.A.; Zhabyeyev, P.; Pituskin, E.; et al. PI3K inhibitors as novel cancer therapies: Implications for cardiovascular medicine. J. Card. Fail. 2013, 19, 268–282.
- 134.
Zhang, P.; Li, H.; Zhang, A.; et al. Mechanism of myocardial fibrosis regulation by IGF-1R in atrial fibrillation through the PI3K/Akt/FoxO3a pathway. Biochem. Cell Biol. 2023, 101, 432–442.
- 135.
Pandey, P.; Khan, F.; Upadhyay, T.K.; et al. New insights about the PDGF/PDGFR signaling pathway as a promising target to develop cancer therapeutic strategies. Biomed. Pharmacother. 2023, 161, 114491.
- 136.
Ivey, M.J.; Kuwabara, J.T.; Riggsbee, K.L.; et al. Platelet-derived growth factor receptor-α is essential for cardiac fibroblast survival. Am. J. Physiol.-Heart Circ. Physiol. 2019, 317, H330–H344.
- 137.
Mouton, A.J. Platelet-Derived Growth Factors: A New Therapeutic Opportunity for Treating Cardiac Fibrosis? Basic Transl. Sci. 2023, 8, 675–676.
- 138.
Asli, N.S.; Xaymardan, M.; Forte, E.; et al. PDGFRα signaling in cardiac fibroblasts modulates quiescence, metabolism and self-renewal, and promotes anatomical and functional repair. BioRxiv 2017, 225979. https://doi.org/10.1101/225979.
- 139.
McMullen, J.R.; Amirahmadi, F.; Woodcock, E.A.; et al. Protective effects of exercise and phosphoinositide 3-kinase (p110α) signaling in dilated and hypertrophic cardiomyopathy. Proc. Natl. Acad. Sci. 2007, 104, 612–617.
- 140.
Proud, C.G. Ras, PI3-kinase and mTOR signaling in cardiac hypertrophy. Cardiovasc. Res. 2004, 63, 403–413.
- 141.
Gao, W.; Guo, N.; Zhao, S.; et al. HTR2A promotes the development of cardiac hypertrophy by activating PI3K-PDK1-AKT-mTOR signaling. Cell Stress. Chaperones 2020, 25, 899–908.
- 142.
Di, R.; Wu, X.; Chang, Z.; et al. S6K inhibition renders cardiac protection against myocardial infarction through PDK1 phosphorylation of Akt. Biochem. J. 2012, 441, 199–207.
- 143.
Völkers, M.; Toko, H.; Doroudgar, S.; et al. Pathological hypertrophy amelioration by PRAS40-mediated inhibition of mTORC1. Proc. Natl. Acad. Sci. 2013, 110, 12661–12666.
- 144.
Sharma, A.K.; Bhatia, S.; Al-Harrasi, A.; et al. Crosstalk between GSK-3β-actuated molecular cascades and myocardial physiology. Heart Fail. Rev. 2021, 26, 1495–1504.
- 145.
Pillai, V.B.; Sundaresan, N.R.; Gupta, M.P. Regulation of Akt signaling by sirtuins: Its implication in cardiac hypertrophy and aging. Circ. Res. 2014, 114, 368–378.
- 146.
Selvetella, G.; Hirsch, E.; Notte, A.; et al. Adaptive and maladaptive hypertrophic pathways: Points of convergence and divergence. Cardiovasc. Res. 2004, 63, 373–380.
- 147.
Bernardo, V.S.; Torres, F.F.; Da Silva, D.G.H. FoxO3 and oxidative stress: A multifaceted role in cellular adaptation. J. Mol. Med. 2023, 101, 83–99.
- 148.
Xin, Z.; Ma, Z.; Jiang, S.; et al. FOXOs in the impaired heart: New therapeutic targets for cardiac diseases. Biochim. Et. Biophys. Acta (BBA)-Mol. Basis Dis. 2017, 1863, 486–498.
- 149.
Shafaati, T.; Gopal, K. Forkhead box O1 transcription factor; a therapeutic target for diabetic cardiomyopathy. J. Pharm. Pharm. Sci. 2024, 27, 13193.
- 150.
Meng, X.; Cui, J.; He, G. Bcl-2 is involved in cardiac hypertrophy through PI3K-Akt pathway. BioMed Res. Int. 2021, 2021, 6615502.
- 151.
Agrawal, Y.O.; Sharma, P.K.; Shrivastava, B.; et al. Hesperidin produces cardioprotective activity via PPAR-γ pathway in ischemic heart disease model in diabetic rats. PLoS ONE 2014, 9, e111212.
- 152.
Mohany, M.; Ahmed, M.M.; Al-Rejaie, S.S. Molecular mechanistic pathways targeted by natural antioxidants in the prevention and treatment of chronic kidney disease. Antioxidants 2021, 11, 15.
- 153.
Vasques-Nóvoa, F.; Angélico-Gonçalves, A.; Alvarenga, J.M.; et al. Myocardial oedema: Pathophysiological basis and implications for the failing heart. ESC Heart Fail. 2022, 9, 958–976.
- 154.
Losada-Barreiro, S.; Bravo-Diaz, C. Free radicals and polyphenols: The redox chemistry of neurodegenerative diseases. Eur. J. Med. Chem. 2017, 133, 379–402.
- 155.
Jiang, M.; Xie, X.; Cao, F.; et al. Mitochondrial metabolism in myocardial remodeling and mechanical unloading: Implications for ischemic heart disease. Front. Cardiovasc. Med. 2021, 8, 789267.
- 156.
Aydin, S.; Ugur, K.; Aydin, S.; et al. Biomarkers in acute myocardial infarction: Current perspectives. Vasc. Health Risk Manag. 2019, 15, 1–10.
- 157.
Battelli, M.G.; Polito, L.; Bortolotti, M.; et al. Xanthine oxidoreductase-derived reactive species: Physiological and pathological effects. Oxidative Med. Cell. Longev. 2016, 2016, 3527579.
- 158.
Zhang, S.; Wang, Y.; Cheng, J.; et al. Hyperuricemia and cardiovascular disease. Curr. Pharm. Des. 2019, 25, 700–709.
- 159.
Sánchez-Lozada, L.G. The pathophysiology of uric acid on renal diseases. In Uric Acid in Chronic Kidney Disease; Karger Publishers: Basel, Switzerland, 2018; Volume 192, pp. 17–24.
- 160.
Zhang, W. Uric acid en route to gout. Adv. Clin. Chem. 2023, 116, 209–275.
- 161.
Damman, K.; Valente, M.A.; Voors, A.A.; et al. Renal impairment, worsening renal function, and outcome in patients with heart failure: An updated meta-analysis. Eur. Heart J. 2014, 35, 455–469.
- 162.
Chen, X.; Li, X.; Xu, X.; et al. Ferroptosis and cardiovascular disease: Role of free radical-induced lipid peroxidation. Free Radic. Res. 2021, 55, 405–415.
- 163.
Vida, C.; Oliva, C.; Yuste, C.; et al. Oxidative stress in patients with advanced CKD and renal replacement therapy: The key role of peripheral blood leukocytes. Antioxidants 2021, 10, 1155.
- 164.
Dal Monte, M.; Fornaciari, I.; Nicchia, G.P.; et al. β3-adrenergic receptor activity modulates melanoma cell proliferation and survival through nitric oxide signaling. Naunyn-Schmiedeberg's Arch. Pharmacol. 2014, 387, 533–543.
- 165.
Kaptoge, S.; Seshasai, S.R.K.; Gao, P.; et al. Inflammatory cytokines and risk of coronary heart disease: New prospective study and updated meta-analysis. Eur. Heart J. 2014, 35, 578–589.
- 166.
Seropian, I.M.; Toldo, S.; Van Tassell, B.W.; et al. Anti-inflammatory strategies for ventricular remodeling following ST-segment elevation acute myocardial infarction. J. Am. Coll. Cardiol. 2014, 63, 1593–1603.
- 167.
Satoh, N.; Nakamura, M.; Suzuki, A.; et al. Effects of nitric oxide on renal proximal tubular Na+ transport. BioMed Res. Int. 2017, 2017, 6871081.
- 168.
de Brum, G.F.; Bochi, G.V. Are Advanced Oxidation Protein Products (AOPPs) Levels Altered in Neuropsychiatric Disorders? An Integrative Review. Mol. Neurobiol. 2024, 61, 9043–9059.
- 169.
Ou, H.; Huang, Z.; Mo, Z.; et al. The characteristics and roles of advanced oxidation protein products in atherosclerosis. Cardiovasc. Toxicol. 2017, 17, 1–12.
- 170.
Wan, C.; Su, H.; Zhang, C. Role of NADPH oxidase in metabolic disease-related renal injury: An update. Oxidative Med. Cell. Longev. 2016, 2016, 7813072.
- 171.
Gryszczyńska, B.; Formanowicz, D.; Budzyń, M.; et al. Advanced oxidation protein products and carbonylated proteins as biomarkers of oxidative stress in selected atherosclerosis-mediated diseases. BioMed Res. Int. 2017, 2017, 4975264.
- 172.
Lin, W.; Chen, H.; Chen, X.; et al. The roles of neutrophil-derived myeloperoxidase (MPO) in diseases: The new progress. Antioxidants 2024, 13, 132.
- 173.
Khan, A.A.; Alsahli, M.A.; Rahmani, A.H. Myeloperoxidase as an active disease biomarker: Recent biochemical and pathological perspectives. Med. Sci. 2018, 6, 33.
- 174.
Ghali, J.R.; Wang, Y.M.; Holdsworth, S.R.; et al. Regulatory T cells in immune-mediated renal disease. Nephrology 2016, 21, 86–96.
- 175.
Costa, T.J.; Barros, P.R.; Arce, C.; et al. The homeostatic role of hydrogen peroxide, superoxide anion and nitric oxide in the vasculature. Free Radic. Biol. Med. 2021, 162, 615–635.
- 176.
Nandi, A.; Yan, L.-J.; Jana, C.K.; et al. Role of catalase in oxidative stress-and age-associated degenerative diseases. Oxidative Med. Cell. Longev. 2019, 2019, 9613090.
- 177.
Otterbein, L.E.; Foresti, R.; Motterlini, R. Heme oxygenase-1 and carbon monoxide in the heart: The balancing act between danger signaling and pro-survival. Circ. Res. 2016, 118, 1940–1959.
- 178.
Akter, N.; Chowdhury, F.I.; Selim, S.; et al. Polyphenolics in ramontchi protect cardiac tissues via suppressing isoprenaline-induced oxidative stress and inflammatory responses in Long-Evans rats. J. Funct. Foods 2020, 75, 104250.
- 179.
Saha, S.; Buttari, B.; Panieri, E.; et al. An overview of Nrf2 signaling pathway and its role in inflammation. Molecules 2020, 25, 5474.
- 180.
Prabhu, S.D.; Frangogiannis, N.G. The biological basis for cardiac repair after myocardial infarction: From inflammation to fibrosis. Circ. Res. 2016, 119, 91–112.
- 181.
Tian, M.; Yuan, Y.-C.; Li, J.-Y.; et al. Tumor necrosis factor-α and its role as a mediator in myocardial infarction: A brief review. Chronic Dis. Transl. Med. 2015, 1, 18–26.
- 182.
Shahrivari, M.; Wise, E.; Resende, M.; et al. Peripheral blood cytokine levels after acute myocardial infarction: IL-1β–and IL-6–related impairment of bone marrow function. Circ. Res. 2017, 120, 1947–1957.
- 183.
Chen, C.; Du, P.; Wang, J. Paeoniflorin ameliorates acute myocardial infarction of rats by inhibiting inflammation and inducible nitric oxide synthase signaling pathways. Mol. Med. Rep. 2015, 12, 3937–3943.
- 184.
Frangogiannis, N.G. The role of transforming growth factor (TGF)-β in the infarcted myocardium. J. Thorac. Dis. 2017, 9, S52.
- 185.
Soriano, F.G.; Guido, M.C.; Barbeiro, H.V.; et al. Endotoxemic myocardial dysfunction: Subendocardial collagen deposition related to coronary driving pressure. Shock 2014, 42, 472–479.
- 186.
Ferreira, V.M.; Marcelino, M.; Piechnik, S.K.; et al. Pheochromocytoma is characterized by catecholamine-mediated myocarditis, focal and diffuse myocardial fibrosis, and myocardial dysfunction. J. Am. Coll. Cardiol. 2016, 67, 2364–2374.
- 187.
Neri, M.; Fineschi, V.; Di Paolo, M.; et al. Cardiac oxidative stress and inflammatory cytokines response after myocardial infarction. Curr. Vasc. Pharmacol. 2015, 13, 26–36.
- 188.
Saparov, A.; Ogay, V.; Nurgozhin, T.; et al. Role of the immune system in cardiac tissue damage and repair following myocardial infarction. Inflamm. Res. 2017, 66, 739–751.
- 189.
Oliveira, J.B.; Soares, A.A.; Sposito, A.C. Inflammatory response during myocardial infarction. Adv. Clin. Chem. 2018, 84, 39–79.
- 190.
Abuzenadah, A.M.; Al-Sayes, F.; Mahafujul Alam, S.S.; et al. Elucidating antiangiogenic potential of Rauwolfia serpentina: VEGFR-2 targeting-based molecular docking study. Evid. Based Complement. Altern. Med. 2022, 2022, 6224666.

This work is licensed under a Creative Commons Attribution 4.0 International License.


