The vibrational circular dichroism (VCD) spectra of (R)-2-chloromethyl-oxirane and (R)-2-chloromethyl-thiirane have been recorded in a vast spectral region including mid-IR, CH-stretching fundamentals and bending-CC/CO(CS) overtone/combination regions in the mid-IR, and CH-stretching overtone/combination regions in the near infrared (NIR). The presence of sulfur is associated with intensification of the NIR-VCD spectra, similarly to what is monitored with electronic circular dichroism (ECD) and optical rotatory dispersion (ORD). DFT calculations, dealing with anharmonicity at the GVPT2 level or based on the local mode approximation, permit to correctly predict the large majority of observed VCD and IR/NIR bands and to explain the role of various molecular moieties. Also, ORD data are better interpreted by including anharmonic vibrational contributions.
- Open Access
- Article
The Influence of Sulphur on Anharmonic Features of Vibrational Circular Dichroism, on Electronic Circular Dichroism and on Optical Rotatory Dispersion: A Study of (R)-2-Chloromethyl-Oxirane, (R)-2-Methyl-Oxirane, (R)-2-Chloromethyl-Thiirane, and (R)-2-Methyl-Thiirane †
- Marco Fusè 1,
- Sergio Abbate 1,2,
- Giuseppe Mazzeo 1,
- Lorenzo Celio 3,
- Francesca Leonelli 3,
- Bruno Brunetti 4,
- Cecilia Cagliero 5,
- Carlo Bicchi 5,
- Stefano Stranges 6,7,
- Giovanna Longhi 1,2,*
Author Information
Received: 21 Nov 2025 | Revised: 08 Jan 2026 | Accepted: 09 Jan 2026 | Published: 04 Mar 2026
Abstract
Graphical Abstract
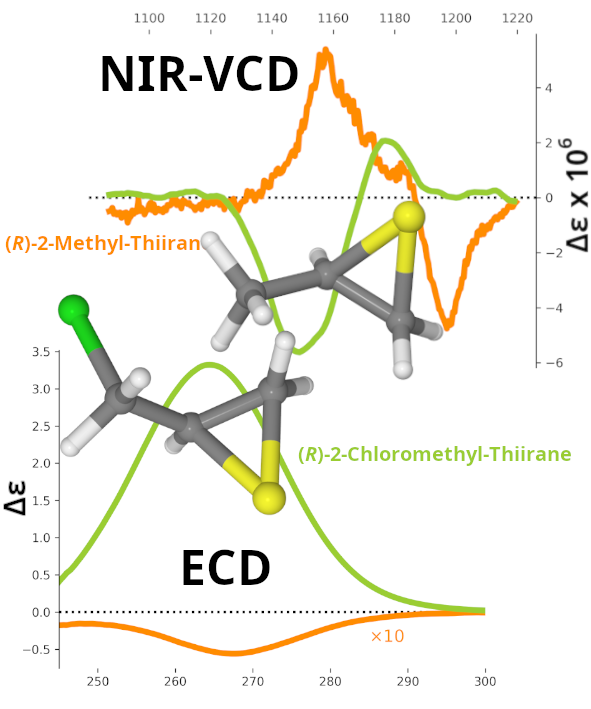
Keywords
methyloxirane | methylthiirane | chloromethyl derivatives | vibrational circular dichroism (VCD) | anharmonicity | GVPT2 DFT calculations | local mode calculations | optical rotatory dispersion (ORD) | electronic circular dichroism (ECD)
References
- 1.
McGuire, B.A.; Carroll, P.B.; Loomis, R.A.; et al. Discovery of the Interstellar Chiral Molecule Propylene Oxide (CH3CHCH2O). Science 2016, 352, 1449–1452. https://doi.org/10.1126/science.aae0328.
- 2.
Garcia, G.A.; Nahon, L.; Daly, S.; et al. Vibrationally Induced Inversion of Photoelectron Forward-Backward Asymmetry in Chiral Molecule Photoionization by Circularly Polarized Light. Nat. Commun. 2013, 4, 2132. https://doi.org/10.1038/ncomms3132.
- 3.
Stranges, S.; Turchini, S.; Alagia, M.; et al. Valence Photoionization Dynamics in Circular Dichroism of Chiral Free Molecules: The Methyl-Oxirane. J. Chem. Phys. 2005, 122, 244307. https://doi.org/10.1063/1.1940632.
- 4.
Turchini, S.; Zema, N.; Contini, G.; et al. Circular Dichroism in Photoelectron Spectroscopy of Free Chiral Molecules: Experiment and Theory on Methyl-Oxirane. Phys. Rev. A 2004, 70, 014502. https://doi.org/10.1103/physreva.70.014502.
- 5.
Lipparini, F.; Egidi, F.; Cappelli, C.; et al. The Optical Rotation of Methyloxirane in Aqueous Solution: A Never Ending Story? J. Chem. Theory Comput. 2013, 9, 1880–1884. https://doi.org/10.1021/ct400061z.
- 6.
Nafie, L.A. Vibrational Optical Activity: Principles and Applications, 1st ed.; Wiley: Hoboken, NJ, USA, 2011. https://doi.org/10.1002/9781119976516.
- 7.
Šebestík, J.; Bouř, P. Raman Optical Activity of Methyloxirane Gas and Liquid. J. Phys. Chem. Lett. 2011, 2, 498–502. https://doi.org/10.1021/jz200108v.
- 8.
Polavarapu, P.L. Rotational–Vibrational Circular Dichroism. Chem. Phys. Lett. 1989, 161, 485–490. https://doi.org/10.1016/0009-2614(89)87025-3.
- 9.
Carnell, M.; Peyerimhoff, S.D.; Breest, A.; et al. Experimental and Quantum-Theoretical Investigation of the Circular Dichroism Spectrum of R-Methyloxirane. Chem. Phys. Lett. 1991, 180, 477–481. https://doi.org/10.1016/0009-2614(91)85153-N.
- 10.
Breest, A.; Ochmann, P.; Pulm, F.; et al. Experimental Circular Dichroism and VUV Spectra of Substituted Oxiranes and Thiiranes. Mol. Phys. 1994, 82, 539–551. https://doi.org/10.1080/00268979400100404.
- 11.
Basil, A.; Ben-Tzur, S.; Gedanken, A.; et al. An Extension of the Quadrant Rule in Oxiranes to Non-Alkyl Substituents: The CD of R (−)- and S (+)-Epichlorohydrin. Chem. Phys. Lett. 1991, 180, 482–484. https://doi.org/10.1016/0009-2614(91)85154-O.
- 12.
Fusè, M.; Longhi, G.; Mazzeo, G.; et al. Anharmonic Aspects in Vibrational Circular Dichroism Spectra from 900 to 9000 Cm−1 for Methyloxirane and Methylthiirane. J. Phys. Chem. A 2022, 126, 6719–6733. https://doi.org/10.1021/acs.jpca.2c05332.
- 13.
Wang, F.; Polavarapu, P.L. Conformational Stability of (+)-Epichlorohydrin. J. Phys. Chem. A 2000, 104, 6189–6196. https://doi.org/10.1021/jp000757c.
- 14.
Abbate, S.; Longhi, G.; Castiglioni, E. Near-Infrared Vibrational Circular Dichroism: NIR-VCD. In Comprehensive Chiroptical Spectroscopy; Berova, N., Polavarapu, P.L., Nakanishi, K., Eds.; Wiley: Hoboken, NJ, USA, 2011; pp 247–273. https://doi.org/10.1002/9781118120187.ch10.
- 15.
Wilson, S.M.; Wiberg, K.B.; Cheeseman, J.R.; et al. Nonresonant Optical Activity of Isolated Organic Molecules. J. Phys. Chem. A 2005, 109, 11752–11764. https://doi.org/10.1021/jp054283z.
- 16.
Crawford, T.D.; Tam, M.C.; Abrams, M.L. The Problematic Case of (S)-Methylthiirane: Electronic Circular Dichroism Spectra and Optical Rotatory Dispersion. Mol. Phys. 2007, 105, 2607–2617. https://doi.org/10.1080/00268970701598097.
- 17.
Fusè, M.; Longhi, G.; Mazzeo, G.; et al. The Role of Anharmonicity in the HCD Chromophore in Vibrational Circular Dichroism Spectra and Optical Rotation Data. J. Phys. Chem. A 2025, 129, 6615–6622. https://doi.org/10.1021/acs.jpca.5c03064.
- 18.
Kleiner, C.M.; Horst, L.; Würtele, C.; et al. Isolation of the Key Intermediates in the Catalyst-Free Conversion of Oxiranes to Thiiranes in Water at Ambient Temperature. Org. Biomol. Chem. 2009, 7, 1397–1402. https://doi.org/10.1039/b820232j.
- 19.
Polavarapu, P.L.; Hess, B.A.; Schaad, L.J.; et al. Vibrational Spectra of Methylthiirane. J. Chem. Phys. 1987, 86, 1140–1146. https://doi.org/10.1063/1.452257.
- 20.
Castiglioni, E.; Lebon, F.; Longhi, G.; et al. Vibrational Circular Dichroism in the Near Infrared: Instrumental Developments and Applications. Enantiomer 2002, 7, 161–173. https://doi.org/10.1080/10242430212877.
- 21.
Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; et al. Gaussian 16, Revision C.01; Gaussian, Inc.: Wallingford, CT, USA, 2019.
- 22.
Bloino, J. A VPT2 Route to Near-Infrared Spectroscopy: The Role of Mechanical and Electrical Anharmonicity. J. Phys. Chem. A 2015, 119, 5269–5287. https://doi.org/10.1021/jp509985u.
- 23.
Lee, M.J.; Hur, S.W.; Durig, J.R. Conformational Stability, Vibrational Assignments, and Normal Coordinate Analysis from FT-IR Spectra of Xenon Solutions and Ab Initio Calculations of Epichlorohydrin. J. Mol. Struct. 1998, 444, 99–113. https://doi.org/10.1016/S0022-2860(97)00344-X.
- 24.
Durig, J.R.; Drew, B.R.; Shoop, J.A.; et al. Conformational Stability Determination of Chloromethyl Thiirane from Variable Temperature FT-IR Studies of Rare Gas Solutions, Structural Parameters, and Ab Initio Calculations. J. Mol. Struct. 2001, 569, 195–212. https://doi.org/10.1016/S0022-2860(01)00430-6.
- 25.
Palumbo, O.; Paolone, A.; Campetella, M.; et al. New Insights into Chloromethyl-Oxirane and Chloromethyl-Thiirane in Liquid and Solid Phase from Low-Temperature Infrared Spectroscopy and Ab Initio Modeling. Spectrochim. Acta A 2021, 247, 119061. https://doi.org/10.1016/j.saa.2020.119061.
- 26.
Fusè, M.; Mazzeo, G.; Longhi, G.; et al. Ledragna/Lcmodslib: v0.1.2. Available online: https://zenodo.org/records/18155720 (accessed on 20 November 2025)
- 27.
Cahn, R.S.; Ingold, C.; Prelog, V. Specification of Molecular Chirality. Angew. Chem. Int. Ed. 1966, 5, 385–415. https://doi.org/10.1002/anie.196603851.
- 28.
Fusè, M.; Mazzeo, G.; Bloino, J.; et al. Pushing Measurements and Interpretation of VCD Spectra in the IR, NIR and Visible Ranges to the Detectability and Computational Complexity Limits. Spectrochim. Acta A 2024, 305, 123496. https://doi.org/10.1016/j.saa.2023.123496.
- 29.
Mendolicchio, M. Harnessing the Power of Curvilinear Internal Coordinates: From Molecular Structure Prediction to Vibrational Spectroscopy. Theor. Chem. Acc. 2023, 142, 133. https://doi.org/10.1007/s00214-023-03069-7.
- 30.
Mendolicchio, M.; Bloino, J.; Barone, V. Perturb-Then-Diagonalize Vibrational Engine Exploiting Curvilinear Internal Coordinates. J. Chem. Theory Comput. 2022, 18, 7603–7619. https://doi.org/10.1021/acs.jctc.2c00773.
- 31.
Longhi, G.; Abbate, S.; Zagano, C.; et al. Analysis of the Transition from Normal Modes to Local Modes in a System of Two Harmonically Coupled Morse Oscillators. Theor. Chim. Acta 1992, 82, 321–337. https://doi.org/10.1007/BF01113262.
- 32.
Abbate, S.; Havel, H.A.; Laux, L.; et al. Vibrational Optical Activity in Deuteriated Phenylethanes. J. Phys. Chem. 1988, 92, 3302–3311. https://doi.org/10.1021/j100322a045.
- 33.
Bendazzoli, G.L.; Gottarelli, G.; Palmieri, P.; et al. The Optical Activity of R-(+)-Propylene Sulphide. Mol. Phys. 1973, 25, 473–477. https://doi.org/10.1080/00268977300100421.
- 34.
Gottarelli, G.; Samori, B.; Torre, G. Dynamic-Coupling Approach to the Optical Activity of the 260 Nm Transition of Chiral Thiirans. J. Chem. Soc. Chem. Commun. 1974, 398–399. https://doi.org/10.1039/C39740000398.
- 35.
Moscowitz, A. Some Remarks on the Interpretation of Natural and Magnetically Induced Optical Activity Data. Proc. R. Soc. Lond. A 1967, 297, 16–26.
- 36.
Mazzeo, G.; Ghidinelli, S.; Ruzziconi, R.; et al. Circularly Polarized Luminescence of Some [2]Paracyclo[2](5,8)quinoliphane Derivatives with Planar and Central Chirality. ChemPhotoChem 2022, 6, e202100222. https://doi.org/10.1002/cptc.202100222.
- 37.
Moscowitz, A. Theoretical Aspects of Optical Activity Part One: Small Molecules. In Advances in Chemical Physics; Prigogine, I., Ed.; Wiley: New York, NY, USA, 1962; Volume 4, pp. 67–112. https://doi.org/10.1002/9780470143506.ch2.
- 38.
Moffitt, W.; Moscowitz, A. Optical Activity in Absorbing Media. J. Chem. Phys. 1959, 30, 648–660. https://doi.org/10.1063/1.1730025.
- 39.
Brockman, M.W.; Moscowitz, A. Macroscopic Sum Rules in Natural Optical Activity. Mol. Phys. 1981, 43, 1385–1393. https://doi.org/10.1080/00268978100102141.
- 40.
Polavarapu, P.L. Kramers–Kronig Transformation for Optical Rotatory Dispersion Studies. J. Phys. Chem. A 2005, 109, 7013–7023. https://doi.org/10.1021/jp0524328.
- 41.
Giorgio, E.; Viglione, R.G.; Zanasi, R.; et al. Ab Initio Calculation of Optical Rotatory Dispersion (ORD) Curves: A Simple and Reliable Approach to the Assignment of the Molecular Absolute Configuration. J. Am. Chem. Soc. 2004, 126, 12968–12976. https://doi.org/10.1021/ja046875l.
- 42.
Ruud, K.; Taylor, P.R.; Åstrand, P.-O. Zero-Point Vibrational Effects on Optical Rotation. Chem. Phys. Lett. 2001, 337, 217–223. https://doi.org/10.1016/S0009-2614(01)00187-7.
- 43.
Pedersen, T.B.; Kongsted, J.; Crawford, T.D.; et al. On the Importance of Vibrational Contributions to Small-Angle Optical Rotation: Fluoro-Oxirane in Gas Phase and Solution. J. Chem. Phys. 2009, 130, 034310. https://doi.org/10.1063/1.3054301.
- 44.
Mort, B.C.; Autschbach, J. Magnitude of Zero-Point Vibrational Corrections to Optical Rotation in Rigid Organic Molecules: A Time-Dependent Density Functional Study. J. Phys. Chem. A 2005, 109, 8617–8623. https://doi.org/10.1021/jp051685y.
- 45.
Faintich, B.; Parsons, T.; Balduf, T.; et al. Theoretical Study of the Isotope Effect in Optical Rotation. J. Phys. Chem. A 2024, 128, 8045–8059. https://doi.org/10.1021/acs.jpca.4c03728.
- 46.
Scholten, K.; Engelage, E.; Merten, C. Basis Set Dependence of S=O Stretching Frequencies and Its Consequences for IR and VCD Spectra Predictions. Phys. Chem. Chem. Phys. 2020, 22, 27979–27986. https://doi.org/10.1039/D0CP05420H.
- 47.
Puente, A.; Chhetri, B.; Kubanek, J.; et al. Revisiting the Absolute Configuration of Peyssonnoside A Using Vibrational Circular Dichroism Spectroscopy. Symmetry 2024, 16, 133. https://doi.org/10.3390/sym16020133.
- 48.
Mangiavacchi, F.; Mazzeo, G.; Graziani, M.C.; et al. Vibrational and Electronic Circular Dichroism Study of Chiral Seleno Compounds Prepared from a Naphthol Based Diselenide. Eur. J. Org. Chem. 2022, 2022, e202200282. https://doi.org/10.1002/ejoc.202200282.
- 49.
Ravutsov, M.; Dobrikov, G.M.; Dangalov, M.; et al. 1,2-Disubstituted Planar Chiral Ferrocene Derivatives from Sulfonamide-Directed Ortho-Lithiation: Synthesis, Absolute Configuration, and Chiroptical Properties. Organometallics 2021, 40, 578–590. https://doi.org/10.1021/acs.organomet.0c00712.
- 50.
Moretti, I.; Torre, G.; Gottarelli, G. The Circular Dichroism and Absolute Configuration of Aryl-Thiiranes. Tetrahedron Lett. 1971, 12, 4301–4304. https://doi.org/10.1016/S0040-4039(01)97425-8.

This work is licensed under a Creative Commons Attribution 4.0 International License.


