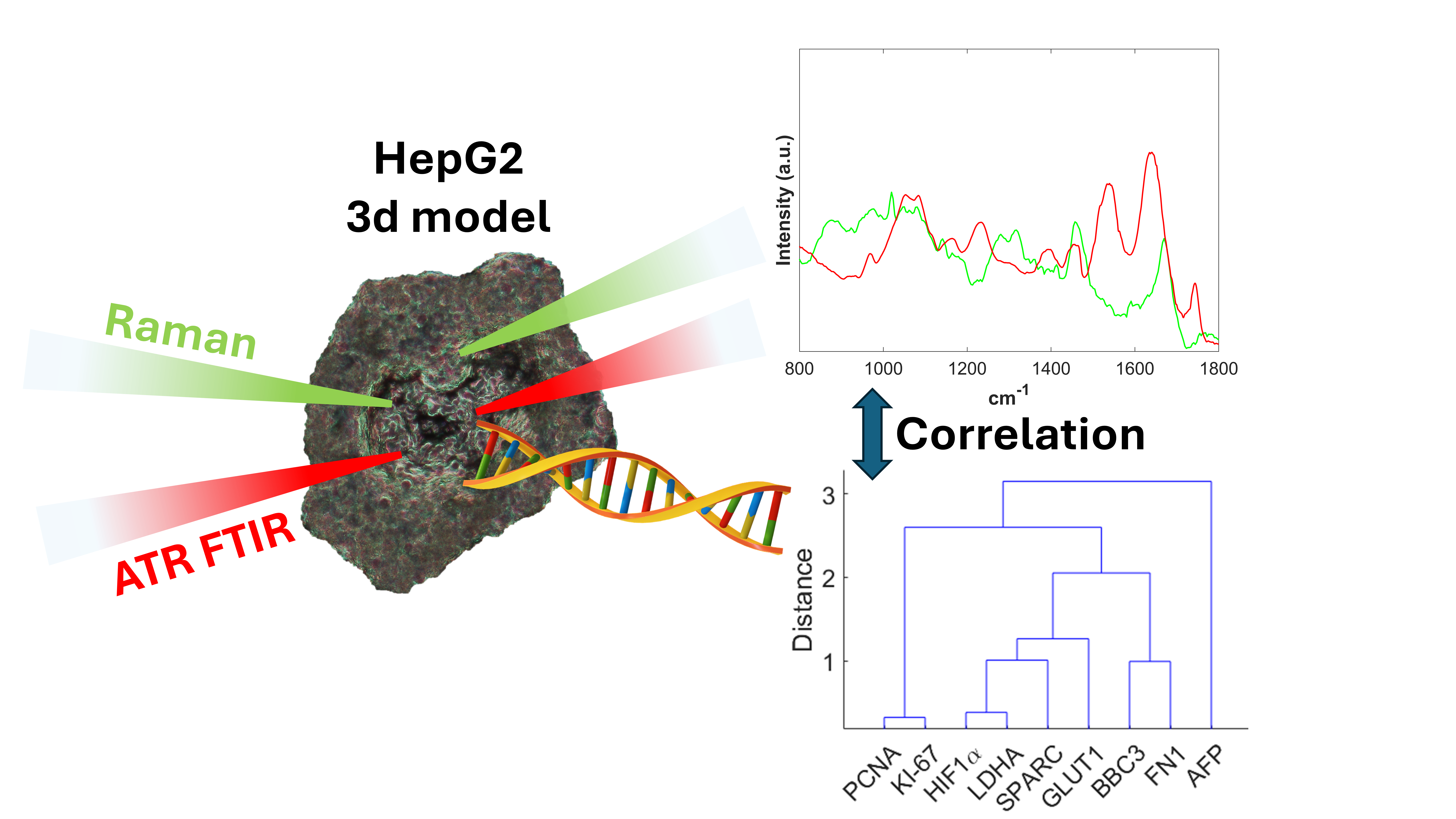
The integration of micro-Raman and ATR-FTIR spectroscopies with qRT-PCR gene expression analysis provides a multimodal strategy for profiling three-dimensional hepatocellular carcinoma (HCC) spheroids. This approach enables temporal monitoring of tumor growth, metabolic adaptation, and microenvironmental remodeling. Gene expression revealed an early proliferative phase (PCNA, KI-67, AFP), followed by activation of hypoxia- and stress-associated pathways (HIF1α, SLC2A1/GLUT1, LDHA) and modulation of extracellular matrix (ECM) components (SPARC, FN1) alongside the pro-apoptotic regulator BBC3. In parallel, Raman and ATR-FTIR spectroscopies non-invasively detected biochemical variations related to oxidative stress, lipid accumulation, apoptosis, and ECM remodeling. The observed correlations between spectral signatures and molecular markers demonstrate that these label-free spectroscopic fingerprints are reliable indicators of physiopathological processes. Our research integrates molecular spectroscopy with transcriptomics to provide an innovative and comprehensive approach for a quick and first evidence of tumor progression.
- Open Access
- Article
Raman and ATR-FTIR Spectroscopic Profiling Coupled with Gene Expression to Monitor Physiopathological Dynamics in 3D HepG2 Liver Cancer Spheroids †
- Enza Fazio 1,*,
- Carmelo Corsaro 1,
- Vincenza Crupi 1,
- Sabrina Conoci 2,3,4,
- Fortunato Neri 1,
- Maria Giovanna Rizzo 2
Author Information
Received: 30 Oct 2025 | Revised: 29 Dec 2025 | Accepted: 21 Jan 2026 | Published: 13 Apr 2026
Abstract
Graphical Abstract
Keywords
micro-Raman | infrared spectroscopy | qRT-PCR | hepatocellular carcinoma | hypoxia | apoptosis | oxidative stress | extracellular matrix remodeling | tumor microenvironment | physiopathological processes
References
- 1.
Zhu, L.; Cheng, C.; Liu, S.; et al. Advancements and Application Prospects of Three-Dimensional Models for Primary Liver Cancer: A Comprehensive Review. Front. Bioeng. Biotechnol. 2023, 11, 1343177. https://doi.org/10.3389/fbioe.2023.1343177.
- 2.
Pastore, M.; Giachi, A.; Spínola-Lasso, E.; et al. Organoids and Spheroids: Advanced in Vitro Models for Liver Cancer Research. Front. Cell Dev. Biol. 2025, 12, 1536854. https://doi.org/10.3389/fcell.2024.1536854.
- 3.
Štampar, M.; Breznik, B.; Filipič, M.; et al. Characterization of In Vitro 3D Cell Model Developed from Human Hepatocellular Carcinoma (HepG2) Cell Line. Cells 2020, 9, 2557. https://doi.org/10.3390/cells9122557.
- 4.
Monaghan, J.F.; Byrne, H.J.; Lyng, F.M.; et al. Radiobiological Applications of Vibrational Spectroscopy: A Review of Analyses of Ionising Radiation Effects in Biology and Medicine. Radiation 2024, 4, 276–308. https://doi.org/10.3390/radiation4030022.
- 5.
Allakhverdiev, E.S.; Kossalbayev, B.D.; Sadvakasova, A.K.; et al. Spectral Insights: Navigating the Frontiers of Biomedical and Microbiological Exploration with Raman Spectroscopy. J. Photochem. Photobiol. B 2024, 252, 112870. https://doi.org/10.1016/j.jphotobiol.2024.112870.
- 6.
Vu, K.H.P.; Blankenburg, G.H.; Lesser-Rojas, L.; et al. Applications of Single-Molecule Vibrational Spectroscopic Techniques for the Structural Investigation of Amyloid Oligomers. Molecules 2022, 27, 6448. https://doi.org/10.3390/molecules27196448.
- 7.
Rizzo, M.G.; Yadav, N.; Morganti, D.; et al. 3D MicroOrganoSpheres Formation of Hepatocellular Carcinoma on Chip as a Novel in Vitro Model for Physiological and Therapeutic Studies. In Sensors and Microsystems; Sabrina, C., Corrado, D.N., Luca, P., et al, Eds.; Springer Nature: Cham, Switzerland, 2025; pp. 9–18.
- 8.
Rizzo, M.G.; Morganti, D.; Smeriglio, A.; et al. Formation of 3D Human Osteoblast Spheroids Incorporating Extracellular Matrix-Mimetic Phage Peptides as a Surrogate Bone Tissue Model. Int. J. Mol. Sci. 2025, 26, 8482. https://doi.org/10.3390/ijms26178482.
- 9.
Rizzo, M.G.; Cordaro, M.; Morganti, D.; et al. Osteogenic Activity and Bone Matrix Mineralization Induced by Vitis Vinifera Leaves Extract in Human Osteoblastic Cells. Food Sci. Nutr. 2025, 13, e70785. https://doi.org/10.1002/fsn3.70785.
- 10.
Chaudhary, I.; Jackson, N.; Denning, D.; et al. Contributions of Vibrational Spectroscopy to Virology: A Review. Clin. Spectrosc. 2022, 4, 100022. https://doi.org/10.1016/j.clispe.2022.100022.
- 11.
Zhang, S.; Qi, Y.; Tan, S.P.H.; et al. Molecular Fingerprint Detection Using Raman and Infrared Spectroscopy Technologies for Cancer Detection: A Progress Review. Biosensors 2023, 13, 557. https://doi.org/10.3390/bios13050557.
- 12.
Siddhanta, S.; Kuzmin, A.N.; Pliss, A.; et al. Advances in Raman Spectroscopy and Imaging for Biomedical Research. Adv. Opt. Photonics 2023, 15, 318. https://doi.org/10.1364/AOP.479884.
- 13.
Rizzo, M.G.; Fazio, E.; De Pasquale, C.; et al. Physiopathological Features in a Three-Dimensional In Vitro Model of Hepatocellular Carcinoma: Hypoxia-Driven Oxidative Stress and ECM Remodeling. Cancers 2025, 17, 3082. https://doi.org/10.3390/cancers17183082.
- 14.
Rizzo, M.G.; Corsaro, C.; Marrara, S.; et al. Raman Spectral Analyses to Investigate the Physiological and Metabolic Development of a 3D Hepatocellular Carcinoma Model. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2025, 343, 126564. https://doi.org/10.1016/j.saa.2025.126564.
- 15.
Takahashi, Y.; Hori, Y.; Yamamoto, T.; et al. 3D Spheroid Cultures Improve the Metabolic Gene Expression Profiles of HepaRG Cells. Biosci. Rep. 2015, 35, e00208. https://doi.org/10.1042/BSR20150034.
- 16.
Sae-be, A.; Wiwatpanit, T.; Varatthan, T.; et al. Comparative Study between the 3D-Liver Spheroid Models Developed from HepG2 and Immortalized Hepatocyte-Like Cells with Primary Hepatic Stellate Coculture for Drug Metabolism Analysis and Anticancer Drug Screening. Adv. Ther. 2023, 6, 2200169. https://doi.org/10.1002/adtp.202200169.
- 17.
Gao, J.; Senthil, M.; Ren, B.; et al. IRF-1 Transcriptionally Upregulates PUMA, Which Mediates the Mitochondrial Apoptotic Pathway in IRF-1-Induced Apoptosis in Cancer Cells. Cell Death Differ. 2010, 17, 699–709. https://doi.org/10.1038/cdd.2009.156.
- 18.
Apicella, C.; Ruano, C.S.M.; Jacques, S.; et al. Urothelial Cancer Associated 1 (UCA1) and MiR-193 Are Two Non-Coding RNAs Involved in Trophoblast Fusion and Placental Diseases. Front. Cell Dev. Biol. 2021, 9, 633937. https://doi.org/10.3389/fcell.2021.633937.
- 19.
Zhu, C.; Fang, X.; Liu, X.; et al. Squalene Monooxygenase Facilitates Bladder Cancer Development in Part by Regulating PCNA. Biochim. Biophys. Acta BBA Mol. Cell Res. 2024, 1871, 119681. https://doi.org/10.1016/j.bbamcr.2024.119681.
- 20.
Huang, S.-W.; Kao, J.-K.; Wu, C.-Y.; et al. Targeting Aerobic Glycolysis and HIF-1α Expression Enhance Imiquimod-Induced Apoptosis in Cancer Cells. Oncotarget 2014, 5, 1363–1381. https://doi.org/10.18632/oncotarget.1734.
- 21.
Franco, D.; Leonardi, A.A.; Rizzo, M.G.; et al. Biological Response Evaluation of Human Fetal Osteoblast Cells and Bacterial Cells on Fractal Silver Dendrites for Bone Tissue Engineering. Nanomaterials 2023, 13, 1107. https://doi.org/10.3390/nano13061107.
- 22.
Ghimire, H.; Garlapati, C.; Janssen, E.A.M.; et al. Protein Conformational Changes in Breast Cancer Sera Using Infrared Spectroscopic Analysis. Cancers 2020, 12, 1708. https://doi.org/10.3390/cancers12071708.
- 23.
Butler, M.; Salem, N.; Hoss, W.; et al. Raman Spectral Analysis of the 1300 Cm−1 Region for Lipid and Membrane Studies. Chem. Phys. Lipids 1979, 24, 99–102. https://doi.org/10.1016/0009-3084(79)90099-9.
- 24.
Ami, D.; Bovio, F.; Forcella, M.; et al. Identifying Label-Free and Untargeted Biomarkers of SH-SY5Y Cell Fate in ATRA-Induced Differentiation through FTIR Spectroscopy and Integrated Omics. Talanta 2026, 296, 128464. https://doi.org/10.1016/j.talanta.2025.128464.
- 25.
Al-Kelani, M.; Buthelezi, N. Advancements in Medical Research: Exploring Fourier Transform Infrared (FTIR) Spectroscopy for Tissue, Cell, and Hair Sample Analysis. Skin. Res. Technol. 2024, 30, e13733. https://doi.org/10.1111/srt.13733.
- 26.
Sandt, C.; Nadaradjane, C.; Richards, R.; et al. Use of Infrared Microspectroscopy to Elucidate a Specific Chemical Signature Associated with Hypoxia Levels Found in Glioblastoma. Analyst 2016, 141, 870–883. https://doi.org/10.1039/C5AN02112J.
- 27.
Cao, R.; Zhao, X.; Li, S.; et al. Hypoxia Induces Dysregulation of Lipid Metabolism in HepG2 Cells via Activation of HIF-2a. Cell. Physiol. Biochem. 2014, 34, 1427–1441. https://doi.org/10.1159/000366348.
- 28.
Chrabaszcz, K.; Kaminska, K.; Augustyniak, K.; et al. Tracking Extracellular Matrix Remodeling in Lungs Induced by Breast Cancer Metastasis. Fourier Transform Infrared Spectroscopic Studies. Molecules 2020, 25, 236. https://doi.org/10.3390/molecules25010236.
- 29.
Ma, C.; Zhang, L.; He, T.; et al. Single Cell Raman Spectroscopy to Identify Different Stages of Proliferating Human Hepatocytes for Cell Therapy. Stem Cell Res. Ther. 2021, 12, 555. https://doi.org/10.1186/s13287-021-02619-9.
- 30.
Paramitha, P.N.; Zakaria, R.; Maryani, A.; et al. Raman Study on Lipid Droplets in Hepatic Cells Co-Cultured with Fatty Acids. Int. J. Mol. Sci. 2021, 22, 7378. https://doi.org/10.3390/ijms22147378.
- 31.
Rak, S.; De Zan, T.; Stefulj, J.; et al. FTIR Spectroscopy Reveals Lipid Droplets in Drug Resistant Laryngeal Carcinoma Cells through Detection of Increased Ester Vibrational Bands Intensity. Analyst 2014, 139, 3407–3415. https://doi.org/10.1039/C4AN00412D.
- 32.
Wu, B.-B.; Gong, Y.-P.; Wu, X.-H.; et al. Fourier Transform Infrared Spectroscopy for the Distinction of MCF-7 Cells Treated with Different Concentrations of 5-Fluorouracil. J. Transl. Med. 2015, 13, 108. https://doi.org/10.1186/s12967-015-0468-2.
- 33.
Xu, J.; Huang, X. Lipid Metabolism at Membrane Contacts: Dynamics and Functions Beyond Lipid Homeostasis. Front. Cell Dev. Biol. 2020, 8, 615856. https://doi.org/10.3389/fcell.2020.615856.
- 34.
Zhang, C.; Liu, S.; Wan, J.; et al. In-Situ Non-Invasive Detection of Cellular Reactive Oxygen Species by Integrating Raman Spectrum and Bidirectional Gated Recurrent Unit Models. Biomed. Opt. Express 2025, 16, 4541. https://doi.org/10.1364/BOE.560107.
- 35.
Campos-Espinosa, A.; Guzmán, C.; Medina-Ávila, K.Z.; et al. In Vitro Lipid Overload Affects Cellular Proliferation, Apoptosis, and Senescence in a Time-Dependent Manner in HepG2 Hepatocytes and LX-2 Hepatic Stellate Cells. Cells 2024, 13, 282. https://doi.org/10.3390/cells13030282.
- 36.
Bonsignore, M.; Trusso, S.; De Pasquale, C.; et al. A Multivariate Analysis of Multiple Myeloma Subtype Plasma Cells. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2021, 258, 119813. https://doi.org/10.1016/j.saa.2021.119813.
- 37.
Lin, J.; Ruan, Q.; Liao, F.; et al. Characterization of Secreted Proteins in HepG2 and LO2 Cells by Raman Spectroscopy. In Optics in Health Care and Biomedical Optics VI; Luo, Q., Li, X., Gu, Y., et al., Eds.; SPIE: Bellingham, WA, USA, 2014; p. 92682M.
- 38.
Bradshaw, A.D. Diverse Biological Functions of the SPARC Family of Proteins. Int. J. Biochem. Cell Biol. 2012, 44, 480–488. https://doi.org/10.1016/j.biocel.2011.12.021.
- 39.
Said, N. Roles of SPARC in Urothelial Carcinogenesis, Progression and Metastasis. Oncotarget 2016, 7, 67574–67585. https://doi.org/10.18632/oncotarget.11590.
- 40.
Roos, W.P.; Kaina, B. DNA Damage-Induced Cell Death by Apoptosis. Trends Mol. Med. 2006, 12, 440–450. https://doi.org/10.1016/j.molmed.2006.07.007.
- 41.
Pridgeon, C.S.; Airavaara, K.; Monola, J.; et al. Chronic Hypoxia for the Adaptation of Extracellular Vesicle Phenotype. Sci. Rep. 2024, 14, 25189. https://doi.org/10.1038/s41598-024-73453-1.
- 42.
Shchulkin, A.V.; Abalenikhina, Y.V.; Kosmachevskaya, O.V.; et al. Regulation of P-Glycoprotein during Oxidative Stress. Antioxidants 2024, 13, 215. https://doi.org/10.3390/antiox13020215.
- 43.
Liang, X.; Weng, J.; You, Z.; et al. Oxidative Stress in Cancer: From Tumor and Microenvironment Remodeling to Therapeutic Frontiers. Mol. Cancer 2025, 24, 219. https://doi.org/10.1186/s12943-025-02375-x.
- 44.
Luo, Q.; Wang, Q.; Shi, J.; et al. PUMA Reduces FASN Ubiquitination to Promote Lipid Accumulation and Tumor Progression in Human Clear Cell Renal Cell Carcinoma. Cell Death Dis. 2025, 16, 460. https://doi.org/10.1038/s41419-025-07782-y.
- 45.
Ferrer, C.M.; Lynch, T.P.; Sodi, V.L.; et al. O-GlcNAcylation Regulates Cancer Metabolism and Survival Stress Signaling via Regulation of the HIF-1 Pathway. Mol. Cell 2014, 54, 820–831. https://doi.org/10.1016/j.molcel.2014.04.026.
- 46.
Spada, S.; Tocci, A.; Di Modugno, F.; et al. Fibronectin as a Multiregulatory Molecule Crucial in Tumor Matrisome: From Structural and Functional Features to Clinical Practice in Oncology. J. Exp. Clin. Cancer Res. 2021, 40, 102. https://doi.org/10.1186/s13046-021-01908-8.
- 47.
Wicks, E.E.; Semenza, G.L. Hypoxia-Inducible Factors: Cancer Progression and Clinical Translation. J. Clin. Investig. 2022, 132, e159839. https://doi.org/10.1172/JCI159839.
- 48.
Nowak-Stępniowska, A.; Osuchowska, P.N.; Fiedorowicz, H.; et al. Hypoxia-Induced Extracellular Matrix Deposition in Human Mesenchymal Stem Cells: Insights from Atomic Force, Scanning Electron, and Confocal Laser Microscopy. Appl. Sci. 2025, 15, 10701. https://doi.org/10.3390/app151910701.
- 49.
Gilkes, D.M.; Semenza, G.L.; Wirtz, D. Hypoxia and the Extracellular Matrix: Drivers of Tumour Metastasis. Nat. Rev. Cancer 2014, 14, 430–439. https://doi.org/10.1038/nrc3726.
- 50.
Magar, A.G.; Morya, V.K.; Kwak, M.K.; et al. A Molecular Perspective on HIF-1α and Angiogenic Stimulator Networks and Their Role in Solid Tumors: An Update. Int. J. Mol. Sci. 2024, 25, 3313. https://doi.org/10.3390/ijms25063313.
- 51.
Yuan, Z.; Li, Y.; Zhang, S.; et al. Extracellular Matrix Remodeling in Tumor Progression and Immune Escape: From Mechanisms to Treatments. Mol. Cancer 2023, 22, 48. https://doi.org/10.1186/s12943-023-01744-8.
- 52.
Junhom, C.; Weerapreeyakul, N.; Tanthanuch, W.; et al. FTIR Microspectroscopy Defines Early Drug Resistant Human Hepatocellular Carcinoma (HepG2) Cells. Exp. Cell Res. 2016, 340, 71–80. https://doi.org/10.1016/j.yexcr.2015.12.007.
- 53.
Maurotti, S.; Scopacasa, B.; Scionti, F.; et al. Raman Spectroscopic Characterization of Liver Steatosis and Fibrosis in a 2D and 3D in Vitro Thyroxine-Treated Hypothyroid Cellular Model. Mol. Cell Endocrinol. 2026, 611, 112679. https://doi.org/10.1016/j.mce.2025.112679.

This work is licensed under a Creative Commons Attribution 4.0 International License.


