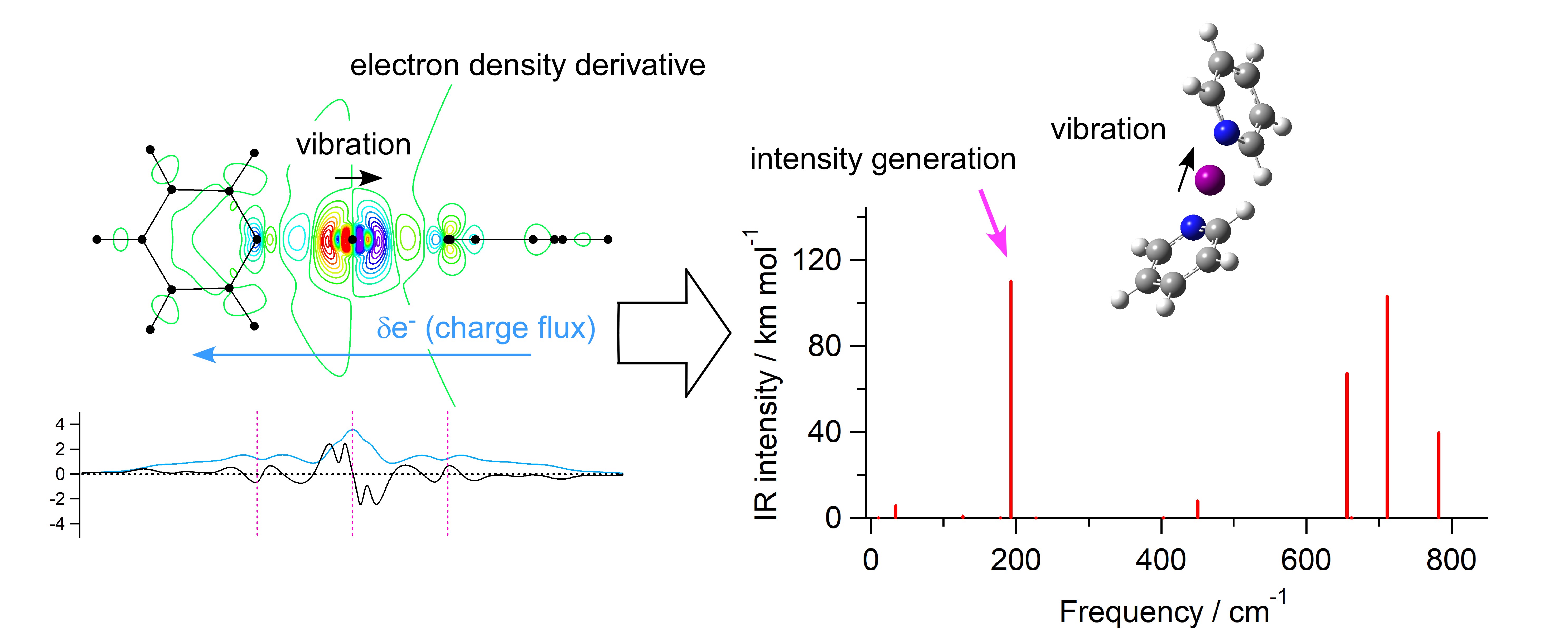
Behavior of electrons is often essentially important for infrared intensities of strongly infrared-active vibrational modes, and elucidating its features is helpful in deepening our understanding on the electronic structural properties of the concerned molecular systems. Here, such an analysis based on electron density features is carried out for the bis(pyridine)iodonium cation (the pyridine‧‧‧I+‧‧‧pyridine complex), a representative system containing a three-center four-electron (3c4e) halogen bond. It is shown that, for the infrared intensity generation and enhancement of two vibrational modes that occur upon formation of this complex, vibration-induced partial transfer of electron density (called charge flux) plays an essential role. It is also shown that the effective electric charge of I+ in this complex is 0.6–0.7 e, which is significantly less than 1 e of an isolated I+ ion, suggesting that the 3c4e halogen bond of the N‧‧‧I+‧‧‧N part has a partially covalent character. The extent of anisotropy in the occupancies of electrons in the p orbitals of the outer-most shell of I+ is ~0.8 in the mixing ratio of px2py2pz0. Similarities and differences with the case of the protonated complex are also discussed.
- Open Access
- Article
Electronic Structural Analysis of the Infrared Intensity Generation and Enhancement Occurring upon Formation of the Bis(Pyridine)Iodonium Cation †
Author Information
Received: 06 Dec 2025 | Revised: 16 Mar 2026 | Accepted: 19 Mar 2026 | Published: 09 May 2026
Abstract
Graphical Abstract
Keywords
infrared intensity | charge flux | electron density | halogen bond | iodonium
References
- 1.
Desiraju, G.R.; Ho, P.S.; Kloo, L.; et al. Definition of the Halogen Bond (IUPAC Recommendations 2013). Pure Appl. Chem. 2013, 85, 1711–1713.
- 2.
Cavallo, G.; Metrangolo, P.; Milani, R.; et al. The Halogen Bond. Chem. Rev. 2016, 116, 2478–2601.
- 3.
Metrangolo, P.; Neukirch, H.; Pilati, T.; et al. Halogen Bonding Based Recognition Processes: A World Parallel to Hydrogen Bonding. Acc. Chem. Res. 2005, 38, 386–395.
- 4.
Costa, P.J. The Halogen Bond: Nature and Applications. Phys. Sci. Rev. 2017, 2, 20170136.
- 5.
Scheiner, S. Understanding Noncovalent Bonds and Their Controlling Forces. J. Chem. Phys. 2020, 153, 140901.
- 6.
Brammer, L.; Peuronen, A.; Roseveare, T.M. Halogen Bonds, Chalcogen Bonds, Pnictogen Bonds, Tetrel Bonds and Other σ-Hole Interactions: A Snapshot of Current Progress. Acta Crystallogr. C 2023, 79, 204–216.
- 7.
Auffinger, P.; Hays, F.A.; Westhof, E.; et al. Halogen Bonds in Biological Molecules. Proc. Natl. Acad. Sci. USA 2004, 101, 16789–16794.
- 8.
Lu, Y.; Wang, Y.; Zhu, W. Nonbonding Interactions of Organic Halogens in Biological Systems: Implications for Drug Discovery and Biomolecular Design. Phys. Chem. Chem. Phys. 2010, 12, 4543–4551.
- 9.
Wilcken, R.; Zimmermann, M.O.; Lange, A.; et al. Principles and Applications of Halogen Bonding in Medicinal Chemistry and Chemical Biology. J. Med. Chem. 2013, 56, 1363–1388.
- 10.
Mukherjee, A.; Tothadi, S.; Desiraju, G.R. Halogen Bonds in Crystal Engineering: Like Hydrogen Bonds Yet Different. Acc. Chem. Res. 2014, 47, 2514–2524.
- 11.
Gilday, L.C.; Robinson, S.W.; Barendt, T.A.; et al. Halogen Bonding in Supramolecular Chemistry. Chem. Rev. 2015, 115, 7118–7195.
- 12.
Bulfield, D.; Huber, S.M. Halogen Bonding in Organic Synthesis and Organocatalysis. Chem. Eur. J. 2016, 22, 14434–14450.
- 13.
Řezáč, J.; de la Lande, A. On the Role of Charge Transfer in Halogen Bonding. Phys. Chem. Chem. Phys. 2017, 19, 791–803.
- 14.
Ramasubbu, N.; Parthasarathy, R.; Murray-Rust, P. Angular Preferences of Intermolecular Forces around Halogen Centers: Preferred Directions of Approach of Electrophiles and Nucleophiles around the Carbon-Halogen Bond. J. Am. Chem. Soc. 1986, 108, 4308–4314.
- 15.
Lommerse, J.P.M.; Stone, A.J.; Taylor, R.; et al. The Nature and Geometry of Intermolecular Interactions between Halogens and Oxygen or Nitrogen. J. Am. Chem. Soc. 1996, 118, 3108–3116.
- 16.
Politzer, P.; Murray, J.S.; Clark, T. Halogen Bonding: An Electrostatically-Driven Highly Directional Noncovalent Interaction. Phys. Chem. Chem. Phys. 2010, 12, 7748–7757.
- 17.
Shields, Z.P.; Murray, J.S.; Politzer, P. Directional Tendencies of Halogen and Hydrogen Bonds. Int. J. Quantum Chem. 2010, 110, 2823–2832.
- 18.
Tsuzuki, S.; Wakisaka, A.; Ono, T.; et al. Magnitude and Origin of the Attraction and Directionality of the Halogen Bonds of the Complexes of C6F5X and C6H5X (X = I, Br, Cl and F) with Pyridine. Chem. Eur. J. 2012, 18, 951–960.
- 19.
Huber, S.M.; Scanlon, J.D.; Jimenez-Izal, E.; et al. On the Directionality of Halogen Bonding. Phys. Chem. Chem. Phys. 2013, 15, 10350–10357.
- 20.
Torii, H.; Yoshida, M. Properties of Halogen Atoms for Representing Intermolecular Electrostatic Interactions Related to Halogen Bonding and Their Substituent Effects. J. Comput. Chem. 2010, 31, 107–116.
- 21.
Torii, H. Properties of Halogen Atoms Related to the Electrostatic Origin of Halogen Bonding: Basic Aspects and Some Applications. AIP Conf. Proc. 2012, 1504, 228–239.
- 22.
Clark, T.; Hennemann, M.; Murray, J.S.; et al. Halogen Bonding: The σ-Hole. J. Mol. Model. 2007, 13, 291–296.
- 23.
Kolář, M.H.; Hobza, P. Computer Modeling of Halogen Bonds and Other σ-Hole Interactions. Chem. Rev. 2016, 116, 5155–5187.
- 24.
Torii, H. The Role of Atomic Quadrupoles in Intermolecular Electrostatic Interactions of Polar and Nonpolar Molecules. J. Chem. Phys. 2003, 119, 2192–2198.
- 25.
Jahromi, H.J.; Eskandari, K. Halogen Bonding: A Theoretical Study Based on Atomic Multipoles Derived from Quantum Theory of Atoms in Molecules. Struct. Chem. 2013, 24, 1281–1287.
- 26.
Cardamone, S.; Hughes, T.J.; Popelier, P.L.A. Multipolar Electrostatics. Phys. Chem. Chem. Phys. 2014, 16, 10367–10387.
- 27.
Tsuzuki, S. Origin of Attraction and Directionality of Hydrogen Bond and Halogen Bond: Analysis by ab initio MO Calculations. AIP Conf. Proc. 2015, 1702, 090044.
- 28.
Ibrahim, M.A.A. Molecular Mechanical Study of Halogen Bonding in Drug Discovery. J. Comput. Chem. 2011, 32, 2564–2574.
- 29.
Gutiérrez, I.S.; Lin, F.-Y.; Vanommeslaeghe, K.; et al. Parametrization of Halogen Bonds in the CHARMM General Force Field: Improved Treatment of Ligand–Protein Interactions. Bioorg. Med. Chem. 2016, 24, 4812–4825.
- 30.
Jorgensen, W.L.; Schyman, P. Treatment of Halogen Bonding in the OPLS-AA Force Field: Application to Potent Anti-HIV Agents. J. Chem. Theory Comput. 2012, 8, 3895–3901.
- 31.
Kolář, M.; Hobza, P. On Extension of the Current Biomolecular Empirical Force Field for the Description of Halogen Bonds. J. Chem. Theory Comput. 2012, 8, 1325–1333.
- 32.
Nunes, R.; Vila-Viçosa, D.; Costa, P.J. Tackling Halogenated Species with PBSA: Effect of Emulating the σ‑Hole. J. Chem. Theory Comput. 2019, 15, 4241–4251.
- 33.
Saito, K.; Izumi, R.; Torii, H. Dissecting the Electric Quadrupolar and Polarization Effects Operating in Halogen Bonding through Electron Density Analysis with a Focus on Bromine. J. Chem. Phys. 2020, 153, 174302.
- 34.
Sakai, T.; Torii, H. Substituent Effect and Its Halogen-Atom Dependence of Halogen Bonding Viewed through Electron Density Changes. Chem. Asian J. 2023, 18, e202201196.
- 35.
Wieske, L.H.E.; Erdélyi, M. Halogen Bonds of Halogen(I) Ions─Where Are We and Where to Go? J. Am. Chem. Soc. 2024, 146, 3–18.
- 36.
Turunen, L.; Erdélyi, M. Halogen Bonds of Halonium Ions. Chem. Soc. Rev. 2020, 49, 2688–2700.
- 37.
Hakkert, S.B.; Erdélyi, M. Halogen Bond Symmetry: The N–X–N Bond. J. Phys. Org. Chem. 2015, 28, 226–233.
- 38.
Carlsson, A.-C.C.; Gräfenstein, J.; Laurila, J.L.; et al. Symmetry of [N–X–N]+ Halogen Bonds in Solution. Chem. Commun. 2012, 48, 1458–1460.
- 39.
Karim, A.; Reitti, M.; Carlsson, A.-C.C.; et al. The Nature of [N–Cl–N]+ and [N–F–N]+ Halogen Bonds in Solution. Chem. Sci. 2014, 5, 3226–3233.
- 40.
Pröhm, P.; Berg, W.; Rupf, S.M.; et al. On Pyridine Chloronium Cations. Chem. Sci. 2023, 14, 2325–2329.
- 41.
Ramasami, P.; Murray, J.S. Anisotropies in Electronic Densities and Electrostatic Potentials of Halonium Ions: Focus on Chlorine, Bromine and Iodine. J. Mol. Model. 2024, 30, 81.
- 42.
Carlsson, A.-C.C.; Gräfenstein, J.; Budnjo, A.; et al. Symmetric Halogen Bonding Is Preferred in Solution. J. Am. Chem. Soc. 2012, 134, 5706–5715.
- 43.
Georgiou, D.C.; Butler, P.; Browne, E.C.; et al. On the Bonding in Bis-pyridine Iodonium Cations. Aust. J. Chem. 2013, 66, 1179–1188.
- 44.
Razmazma, H.; Ebrahimi, A. The Effects of Cation-π and Anion-π Interactions on Halogen Bonds in the [N···X···N]+ Complexes: A Comprehensive Theoretical Study. J. Mol. Graph. Model. 2018, 84, 134–144.
- 45.
Velasquez, J.D.; Echeverría, J.; Alvarez, S. Structure and Bonding of Halonium Compounds. Inorg. Chem. 2023, 62, 8980–8992.
- 46.
Barluenga, J.; Gonzalez, J.M.; Garcia-Martin, M.A.; et al. Acid-Mediated Reaction of Bis(Pyridine)Iodonium(I) Tetrafluoroborate with Aromatic Compounds. A Selective and General Iodination Method. J. Org. Chem. 1993, 58, 2058–2060.
- 47.
Guha, S.; Kazi, I.; Nandy, A.; et al. Role of Lewis-Base-Coordinated Halogen(I) Intermediates in Organic Synthesis: The Journey from Unstable Intermediates to Versatile Reagents. Eur. J. Org. Chem. 2017, 2017, 5497–5518.
- 48.
Oishi, S.; Fujinami, T.; Masui, Y.; et al. Three-Center-Four-Electron Halogen Bond Enables Non-Metallic Complex Catalysis for Mukaiyama-Mannich-Type Reaction. iScience 2022, 25, 105220.
- 49.
Turunen, L.; Peuronen, A.; Forsblom, S.; et al. Tetrameric and Dimeric [N···I+···N] Halogen-Bonded Supramolecular Cages. Chem. Eur. J. 2017, 23, 11714–11718.
- 50.
Warzok, U.; Marianski, M.; Hoffmann, W.; et al. Surprising Solvent-Induced Structural Rearrangements in Large [N⋯I+⋯N] Halogen-Bonded Supramolecular Capsules: An Ion Mobility-Mass Spectrometry Study. Chem. Sci. 2018, 9, 8343–8351.
- 51.
Vanderkooy, A.; Gupta, A.K.; Földes, T.; et al. Halogen Bonding Helicates Encompassing Iodonium Cations. Angew. Chem. Int. Ed. 2019, 58, 9012–9016.
- 52.
An, S.; Hao, A.; Xing, P. Supramolecular Axial Chirality in [N–I–N]+-Type Halogen Bonded Dimers. Chem. Sci. 2023, 14, 10194–10202.
- 53.
Haque, I.; Wood, J.L. The Vibrational Spectra and Structure of the Bis(Pyridine)Iodine(I), Bis(Pyridine)Bromine(I), Bis(γ-Picoline)Iodine(I) and Bis(γ-Picoline)Bromine(I) Cations. J. Mol. Struct. 1968, 2, 217–238.
- 54.
Torii, H. Intermolecular Charge Flux as the Origin of Infrared Intensity Enhancement upon Halogen-Bond Formation of the Peptide Group. J. Chem. Phys. 2010, 133, 034504.
- 55.
Torii, H. Correlation of the Partial Charge-Transfer and Covalent Nature of Halogen Bonding with the THz and IR Spectral Changes. Phys. Chem. Chem. Phys. 2019, 21, 17118–17125.
- 56.
Ino, K.; Torii, H. Relative Importance of Electrostatic and Intermolecular Charge-Transfer Interactions in Halogen Bonding Depending on the Properties Analyzed. Phys. Chem. Chem. Phys. 2025, 27, 19295–19303.
- 57.
Decius, J.C. An effective Atomic Charge Model for Infrared Intensities. J. Mol. Spectrosc. 1975, 57, 348–362.
- 58.
van Straten, A.J.; Smit, W.M.A. Bond Charge Parameters from Integrated Infrared Intensities. J. Mol. Spectrosc. 1976, 62, 297–312.
- 59.
Gussoni, M.; Castiglioni, C.; Zerbi, G. Physical Meaning of Electrooptical Parameters Derived from Infrared Intensities. J. Phys. Chem. 1984, 88, 600–604.
- 60.
Gussoni, M.; Ramos, M.N.; Castiglioni, C.; et al. Ab Initio Counterpart of Infrared Atomic Charges: Charge Fluxes. Chem. Phys. Lett. 1989, 160, 200–205.
- 61.
Torii, H.; Tasumi, M. Infrared Intensities of Vibrational Modes of an α-Helical Polypeptide: Calculations Based on the Equilibrium Charge/Charge Flux (ECCF) Model. J. Mol. Struct. 1993, 300, 171–179.
- 62.
Torii, H. Cooperative Contributions of the Intermolecular Charge Fluxes and Intramolecular Polarizations in the Far-Infrared Spectral Intensities of Liquid Water. J. Chem. Theory Comput. 2014, 10, 1219–1227.
- 63.
Corrsin, L.; Fax, B.J.; Lord, R.C. The Vibrational Spectra of Pyridine and Pyridine-d5. J. Chem. Phys. 1953, 21, 1170–1176.
- 64.
Partal Ureña, F.; Fernández Gómez, M.; López González, J.J.; et al. A New Insight into the Vibrational Analysis of Pyridine. Spectrochim. Acta Part A 2003, 59, 2815–2839.
- 65.
Torii, H.; Ukawa, R. Role of Intermolecular Charge Fluxes in the Hydrogen-Bond-Induced Frequency Shifts of the OH Stretching Mode of Water. J. Phys. Chem. B 2021, 125, 1468–1475.
- 66.
Torii, H. Roles of Electrostatics and Intermolecular Electronic Motions in the Structural and Spectroscopic Features of Hydrogen- and Halogen-Bonded Systems. Pure Appl. Chem. 2024, 96, 579–595.
- 67.
Chai, J.-D.; Head-Gordon, M. Long-Range Corrected Hybrid Density Functionals with Damped Atom–Atom Dispersion Corrections. Phys. Chem. Chem. Phys. 2008, 10, 6615–6620.
- 68.
Weigend, F.; Ahlrichs, R. Balanced Basis Sets of Split Valence, Triple Zeta Valence and Quadruple Zeta Valence Quality for H to Rn: Design and Assessment of Accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305.
- 69.
Weigend, F. Accurate Coulomb-fitting Basis Sets for H to Rn. Phys. Chem. Chem. Phys. 2006, 8, 1057–1065.
- 70.
Del Bene, J.E.; Elguero, J. Systematic ab Initio Study of 15N−15N and 15N−1H Spin−Spin Coupling Constants Across N−H+−N Hydrogen Bonds: Predicting N−N and N−H Coupling Constants and Relating Them to Hydrogen Bond Type. J. Phys. Chem. A 2006, 110, 7496–7502.
- 71.
Tayyari, S.F.; Mahdizadeh, S.J.; Holakoei, S.; et al. Vibrational Assignment and Proton Tunneling in Pyridine–Pyridinium Complexes. J. Mol. Struct. 2010, 971, 39–46.
- 72.
Melikova, S.M.; Rutkowski, K.S.; Gurinov, A.A.; et al. FTIR Study of the Hydrogen Bond Symmetry in Protonated Homodimers of Pyridine and Collidine in Solution. J. Mol. Struct. 2012, 1018, 39–44.
- 73.
Attah, I.K.; Platt, S.P.; Meot-Ner (Mautner), M.; et al. Proton-Bound Dimers of Nitrogen Heterocyclic Molecules: Substituent Effects on the Structures and Binding Energies of Homodimers of Diazine, Triazine, and Fluoropyridine. J. Chem. Phys. 2014, 140, 114313.
- 74.
Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; et al. Gaussian 09, Revision D.01; Gaussian, Inc.: Wallingford, CT, USA, 2013.
- 75.
Agmon, N.; Bakker, H.J.; Campen, R.K.; et al. Protons and Hydroxide Ions in Aqueous Systems. Chem. Rev. 2016, 116, 7642–7672.
- 76.
Biswas, R.; Carpenter, W.; Fournier, J.A.; et al. IR Spectral Assignments for the Hydrated Excess Proton in Liquid Water. J. Chem. Phys. 2017, 146, 154507.
- 77.
Zundel, G. Hydration Structure and Intermolecular Interaction in Polyelectrolytes. Angew. Chem. Int. Ed. 1969, 8, 499–509.

This work is licensed under a Creative Commons Attribution 4.0 International License.


