Electrochemical nitrogen fixation (EN2F) is emerging as a promising technology. This review summarizes recent advances in both nitrogen reduction reaction (N2RR) and nitrogen oxidation reaction (N2OR) pathways for ammonia synthesis under mild conditions. We begin by contrasting the fundamental reactions and mechanisms of N2RR and N2OR, emphasizing the impact of competing hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) on EN2F selectivity. A detailed analysis of various N2RR mechanisms, including dissociative, associative, Mars-van Krevelen, surface-hydrogenation, and dual-site pathways, is presented, highlighting their respective advantages and limitations. For N2OR, we focus on O-preferred and N-preferred reaction routes, emphasizing the role of hydroxyl species in N2 activation. Progress in catalyst development for both N2RR and N2OR is summarized, with particular attention to surface-hydrogenation or dual-site mechanisms, as well as strong/weak N-bonding bimetallic catalysts. Strategies for promoting N2OR by modulating the OER are also discussed. Furthermore, we analyze the kinetic steps of EN2F, identifying nitrogen dissolution and mass transfer as potential rate-limiting factors, and propose strategies for enhancing EN2F selectivity through electrolyte optimization, electrolyzer design, and catalyst modification. Finally, we offer a perspective on the future directions of EN2F, acknowledging the current challenges of active nitrogen pollution and suggesting avenues for technological advancement through refined experimental protocols, innovative catalyst design, and thermochemical analysis.
- Open Access
- Review
Electrochemical N2 Conversion: Reduction and Oxidation Pathways under Mild Conditions
- Xingyu Ma 1,
- Xuanhe Wang 2,3,
- Jingwen Liu 2,3,
- Yibo Ma 2,3,*
Author Information
Received: 10 Dec 2025 | Revised: 01 Jan 2026 | Accepted: 05 Feb 2026 | Published: 02 Mar 2026
Abstract
Graphical Abstract
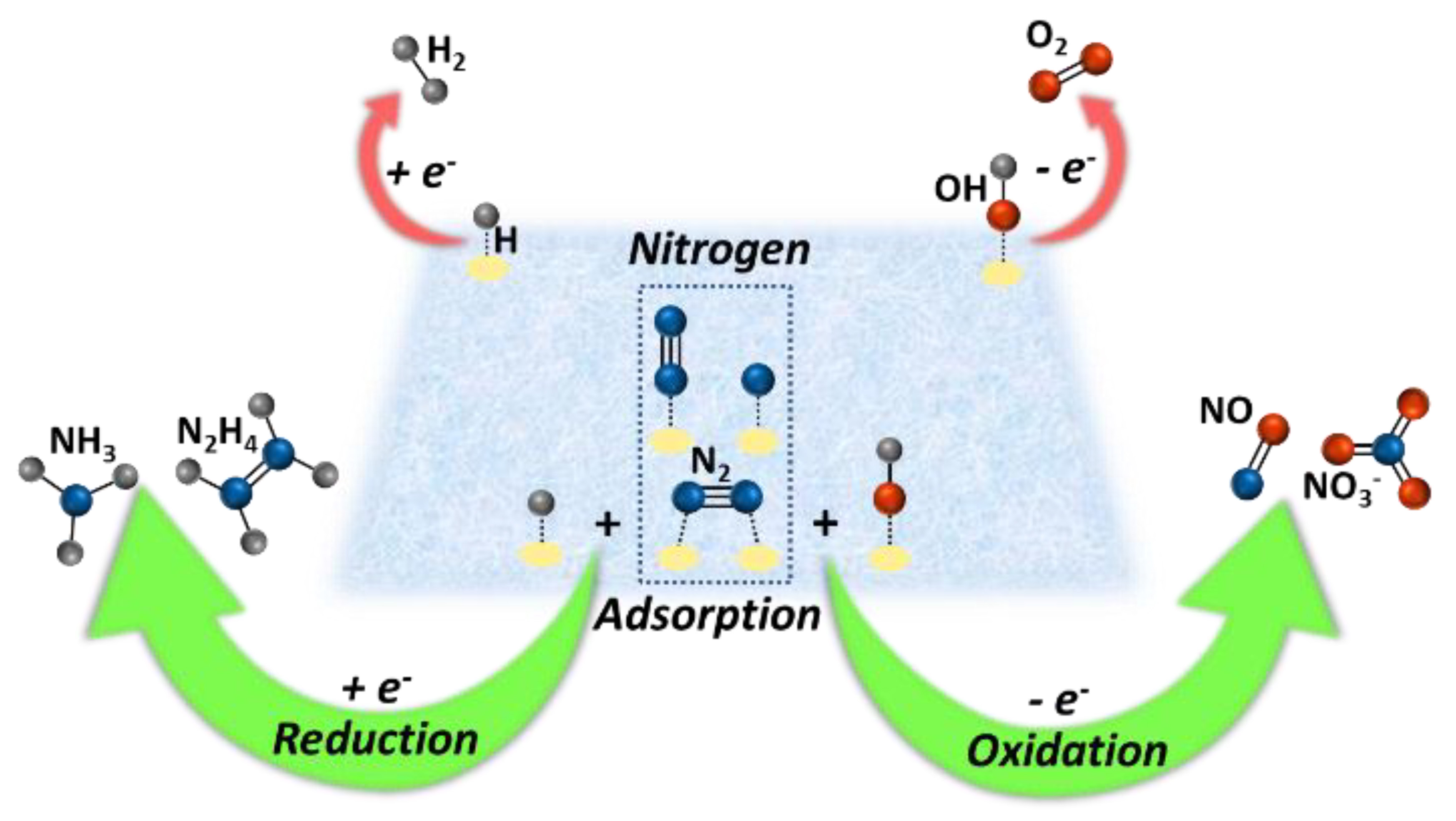
Keywords
electrochemical nitrogen fixation | nitrogen reduction | nitrogen oxidation | catalyst mechanisms | selectivity
References
- 1.
Ye, L.; Nayak-Luke, R.; Bañares-Alcántara, R.; et al. Reaction: “Green” Ammonia Production. Chem 2017, 3, 712–714. https://doi.org/10.1016/j.chempr.2017.10.016.
- 2.
Heffer, P.; Prud’homme, M. Global Nitrogen Fertiliser Demand and Supply Trend, Current Level and Outlook. In Proceedings of the 2016 International Nitrogen Initiative Conference, “Solutions to Improve Nitrogen Use Efficiency for the World”, Melbourne, VIC, Australia, 4–8 December 2016.
- 3.
Smil, V. Detonator of the Population Explosion. Nature 1999, 400, 415. https://doi.org/10.1038/22672.
- 4.
Yu, Y.-H.; Yuan, Y.-F.; Li, W.; et al. Unveiling Cutting-Edge Developments on d-Block Iron-Based Materials for Electrocatalytic Ammonia Synthesis. Rare Met. 2025, 44, 9785–9798. https://doi.org/10.1007/s12598-025-03592-5.
- 5.
Guo, J.; Chen, P. Catalyst: NH3 as an Energy Carrier. Chem 2017, 3, 709–712. https://doi.org/10.1016/j.chempr.2017.10.004.
- 6.
Giddey, S.; Badwal, S.P.S.; Munnings, C.; et al. Ammonia as a Renewable Energy Transportation Media. ACS Sustain. Chem. Eng. 2017, 5, 10231–10239. https://doi.org/10.1021/acssuschemeng.7b02219.
- 7.
Li, R.; Bao, L.; Li, J.; et al. Construction of Multiple Active Sites for Energy-Efficient Ammonia Production by Nitrate Reduction Coupling with Urea Oxidation. Chem. Eng. J. 2025, 524, 169332. https://doi.org/10.1016/j.cej.2025.169332.
- 8.
Groves, M.C.E. Nitric Acid. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons: Hoboken, NJ, USA, 2020; pp. 1–37.
- 9.
Yu, Y.; Li, J.; Yue, Z.; et al. Advances in Multiple Active Site Catalysts for Urea Synthesis via Electrocatalytic C–N Coupling. Trans. Tianjin Univ. 2025, 31, 478–497. https://doi.org/10.1007/s12209-025-00444-2.
- 10.
Kyriakou, V.; Garagounis, I.; Vourros, A.; et al. An Electrochemical Haber-Bosch Process. Joule 2020, 4, 142–158. https://doi.org/10.1016/j.joule.2019.10.006.
- 11.
Jiang, L.; Liu, F. Carbon Dioxide and Nitrate Electrocatalytic C–N Coupling for Sustainable Production of Urea. Sci. Energy Environ. 2024, 1, 2. https://doi.org/10.53941/see.2024.100002.
- 12.
Smith, C.; Hill, A.K.; Torrente-Murciano, L. Current and Future Role of Haber-Bosch Ammonia in a Carbon-Free Energy Landscape. Energy Environ. Sci. 2020, 13, 331–344. https://doi.org/10.1039/c9ee02873k.
- 13.
Wang, Q.; Guo, J.; Chen, P. Recent Progress Towards Mild-Condition Ammonia Synthesis. J. Energy Chem. 2019, 36, 25–36. https://doi.org/10.1016/j.jechem.2019.01.027.
- 14.
Pérez-Ramírez, J.; Kapteijn, F.; Schöffel, K.; et al. Formation and Control of N2O in Nitric Acid Production. Appl. Catal. B 2003, 44, 117–151. https://doi.org/10.1016/s0926-3373(03)00026-2.
- 15.
Schiffer, Z.J.; Manthiram, K. Electrification and Decarbonization of the Chemical Industry. Joule 2017, 1, 10–14. https://doi.org/10.1016/j.joule.2017.07.008.
- 16.
MacFarlane, D.R.; Cherepanov, P.V.; Choi, J.; et al. A Roadmap to the Ammonia Economy. Joule 2020, 4, 1186–1205. https://doi.org/10.1016/j.joule.2020.04.004.
- 17.
Stevens, C.J. Nitrogen in the Environment. Science 2019, 363, 578–580. https://doi.org/10.1126/science.aav8215.
- 18.
Chen, J.G.; Crooks, R.M.; Seefeldt, L.C.; et al. Beyond Fossil Fuel-Driven Nitrogen Transformations. Science 2018, 360, 873–880. https://doi.org/10.1126/science.aar6611.
- 19.
Cui, X.; Tang, C.; Zhang, Q. A Review of Electrocatalytic Reduction of Dinitrogen to Ammonia under Ambient Conditions. Adv. Energy Mater. 2018, 8, 1800369–1800394. https://doi.org/10.1002/aenm.201800369.
- 20.
Soloveichik, G. Electrochemical Synthesis of Ammonia as a Potential Alternative to the Haber-Bosch Process. Nat. Catal. 2019, 2, 377–380. https://doi.org/10.1038/s41929-019-0280-0.
- 21.
Qing, G.; Ghazfar, R.; Jackowski, S.T.; et al. Recent Advances and Challenges of Electrocatalytic N2 Reduction to Ammonia. Chem. Rev. 2020, 120, 5437–5516. https://doi.org/10.1021/acs.chemrev.9b00659.
- 22.
Yang, C.; Zhu, Y.; Liu, J.; et al. Defect Engineering for Electrochemical Nitrogen Reduction Reaction to Ammonia. Nano Energy 2020, 77, 105126. https://doi.org/10.1016/j.nanoen.2020.105126.
- 23.
Liu, J.; Wang, G.; Zhou, S.; et al. CeO2-Supported Monodispersed MoO3 Clusters for High-Efficiency Electrochemical Nitrogen Reduction under Ambient Condition. J. Energy Chem. 2021, 56, 186–192. https://doi.org/10.1016/j.jechem.2020.07.042.
- 24.
Lv, X.-W.; Liu, Y.; Wang, Y.-S.; et al. Encapsulating Vanadium Nitride Nanodots into N,S-Codoped Graphitized Carbon for Synergistic Electrocatalytic Nitrogen Reduction and Aqueous Zn-N2 Battery. Appl. Catal. B 2021, 280, 119434–119442. https://doi.org/10.1016/j.apcatb.2020.119434.
- 25.
Wang, Y.; Yu, Y.; Jia, R.; et al. Electrochemical Synthesis of Nitric Acid from Air and Ammonia through Waste Utilization. Natl. Sci. Rev. 2019, 6, 730–738. https://doi.org/10.1093/nsr/nwz019.
- 26.
Peng, X.; Mi, Y.; Bao, H.; et al. Ambient Electrosynthesis of Ammonia with Efficient Denitration. Nano Energy 2020, 78, 105321. https://doi.org/10.1016/j.nanoen.2020.105321.
- 27.
Long, J.; Chen, S.; Zhang, Y.; et al. Direct Electrochemical Ammonia Synthesis from Nitric Oxide. Angew. Chem. Int. Ed. 2020, 59, 9711–9718. https://doi.org/10.1002/anie.202002337.
- 28.
Li, J.; Zhan, G.; Yang, J.; et al. Efficient Ammonia Electrosynthesis from Nitrate on Strained Ruthenium Nanoclusters. J. Am. Chem. Soc. 2020, 142, 7036–7046. https://doi.org/10.1021/jacs.0c00418.
- 29.
Chen, G.-F.; Yuan, Y.; Jiang, H.; et al. Electrochemical Reduction of Nitrate to Ammonia via Direct Eight-Electron Transfer Using a Copper–Molecular Solid Catalyst. Nat. Energy 2020, 5, 605–613. https://doi.org/10.1038/s41560-020-0654-1.
- 30.
Wang, Y.; Zhou, W.; Jia, R.; et al. Unveiling the Activity Origin of a Copper-Based Electrocatalyst for Selective Nitrate Reduction to Ammonia. Angew. Chem. Int. Ed. 2020, 59, 5350–5354. https://doi.org/10.1002/anie.201915992.
- 31.
Rod, T.H.; Logadottir, A.; Nørskov, J.K. Ammonia Synthesis at Low Temperatures. J. Chem. Phys. 2000, 112, 5343–5347. https://doi.org/10.1063/1.481103.
- 32.
van der Ham, C.J.; Koper, M.T.; Hetterscheid, D.G. Challenges in Reduction of Dinitrogen by Proton and Electron Transfer. Chem. Soc. Rev. 2014, 43, 5183–5191. https://doi.org/10.1039/c4cs00085d.
- 33.
Tang, C.; Qiao, S.Z. How to Explore Ambient Electrocatalytic Nitrogen Reduction Reliably and Insightfully. Chem. Soc. Rev. 2019, 48, 3166–3180. https://doi.org/10.1039/c9cs00280d.
- 34.
Fang, W.; Du, C.; Kuang, M.; et al. Boosting Efficient Ambient Nitrogen Oxidation by a Well-Dispersed Pd on MXene Electrocatalyst. Chem. Commun. 2020, 56, 5779–5782. https://doi.org/10.1039/d0cc01759k.
- 35.
Koper, M.T.M. Theory of Multiple Proton-Electron Transfer Reactions and Its Implications for Electrocatalysis. Chem. Sci. 2013, 4, 2710–2723. https://doi.org/10.1039/c3sc50205h.
- 36.
Cheng, H.; Ding, L.X.; Chen, G.F.; et al. Molybdenum Carbide Nanodots Enable Efficient Electrocatalytic Nitrogen Fixation under Ambient Conditions. Adv. Mater. 2018, 30, 1803694. https://doi.org/10.1002/adma.201803694.
- 37.
Honkala, K.; Hellman, A.; Remediakis, I.N.; et al. Ammonia Synthesis from First-Principles Calculations. Science 2005, 307, 555–558. https://doi.org/10.1126/science.1106435.
- 38.
Montoya, J.H.; Tsai, C.; Vojvodic, A.; et al. The Challenge of Electrochemical Ammonia Synthesis: A New Perspective on the Role of Nitrogen Scaling Relations. ChemSusChem 2015, 8, 2180–2186. https://doi.org/10.1002/cssc.201500322.
- 39.
Garden, A.L.; Skúlason, E. The Mechanism of Industrial Ammonia Synthesis Revisited: Calculations of the Role of the Associative Mechanism. J. Phys. Chem. C 2015, 119, 26554–26559. https://doi.org/10.1021/acs.jpcc.5b08508.
- 40.
Singh, A.R.; Montoya, J.H.; Rohr, B.A.; et al. Computational Design of Active Site Structures with Improved Transition-State Scaling for Ammonia Synthesis. ACS Catal. 2018, 8, 4017–4024. https://doi.org/10.1021/acscatal.8b00106.
- 41.
Kitano, M.; Kanbara, S.; Inoue, Y.; et al. Electride Support Boosts Nitrogen Dissociation over Ruthenium Catalyst and Shifts the Bottleneck in Ammonia Synthesis. Nat. Commun. 2015, 6, 6731–6740. https://doi.org/10.1038/ncomms7731.
- 42.
Song, T.; Hu, P. Insight into the Adsorption Competition and the Relationship between Dissociation and Association Reactions in Ammonia Synthesis. J. Chem. Phys. 2007, 127, 234706–234715. https://doi.org/10.1063/1.2799984.
- 43.
Buscagan, T.M.; Rees, D.C. Rethinking the Nitrogenase Mechanism: Activating the Active Site. Joule 2019, 3, 2662–2678. https://doi.org/10.1016/j.joule.2019.09.004.
- 44.
Kobayashi, Y.; Kitano, M.; Kawamura, S.; et al. Kinetic Evidence: The Rate-Determining Step for Ammonia Synthesis over Electride-Supported Ru Catalysts Is No Longer the Nitrogen Dissociation Step. Catal. Sci. Technol. 2017, 7, 47–50. https://doi.org/10.1039/c6cy01962e.
- 45.
Matsuishi, S.; Toda, Y.; Miyakawa, M.; et al. High-Density Electron Anions in a Nanoporous Single Crystal: [Ca24Al28O64]4+(4e−). Science 2003, 301, 626–629. https://doi.org/10.1126/science.1083842.
- 46.
Kitano, M.; Inoue, Y.; Yamazaki, Y.; et al. Ammonia Synthesis Using a Stable Electride as an Electron Donor and Reversible Hydrogen Store. Nat. Chem. 2012, 4, 934–940. https://doi.org/10.1038/nchem.1476.
- 47.
Kanbara, S.; Kitano, M.; Inoue, Y.; et al. Mechanism Switching of Ammonia Synthesis Over Ru-Loaded Electride Catalyst at Metal-Insulator Transition. J. Am. Chem. Soc. 2015, 137, 14517–14524. https://doi.org/10.1021/jacs.5b10145.
- 48.
Kitano, M.; Inoue, Y.; Ishikawa, H.; et al. Essential Role of Hydride Ion in Ruthenium-Based Ammonia Synthesis Catalysts. Chem. Sci. 2016, 7, 4036–4043. https://doi.org/10.1039/c6sc00767h.
- 49.
Wu, J.; Gong, Y.; Inoshita, T.; et al. Tiered Electron Anions in Multiple Voids of LaScSi and Their Applications to Ammonia Synthesis. Adv. Mater. 2017, 29, 1700924–1700931. https://doi.org/10.1002/adma.201700924.
- 50.
Wu, J.; Li, J.; Gong, Y.; et al. Intermetallic Electride Catalyst as a Platform for Ammonia Synthesis. Angew. Chem. Int. Ed. 2019, 58, 825–829. https://doi.org/10.1002/anie.201812131.
- 51.
Hattori, M.; Mori, T.; Arai, T.; et al. Enhanced Catalytic Ammonia Synthesis with Transformed BaO. ACS Catal. 2018, 8, 10977–10984. https://doi.org/10.1021/acscatal.8b02839.
- 52.
Nakao, T.; Tada, T.; Hosono, H. Transition Metal-Doped Ru Nanoparticles Loaded on Metal Hydrides for Efficient Ammonia Synthesis from First Principles. J. Phys. Chem. C 2019, 124, 1529–1534. https://doi.org/10.1021/acs.jpcc.9b10544.
- 53.
Kitano, M.; Kujirai, J.; Ogasawara, K.; et al. Low-Temperature Synthesis of Perovskite Oxynitride-Hydrides as Ammonia Synthesis Catalysts. J. Am. Chem. Soc. 2019, 141, 20344–20353. https://doi.org/10.1021/jacs.9b10726.
- 54.
Hattori, M.; Iijima, S.; Nakao, T.; et al. Solid Solution for Catalytic Ammonia Synthesis from Nitrogen and Hydrogen Gases at 50 ℃. Nat. Commun. 2020, 11, 2001–2009. https://doi.org/10.1038/s41467-020-15868-8.
- 55.
Ye, T.N.; Park, S.W.; Lu, Y.; et al. Vacancy-Enabled N2 Activation for Ammonia Synthesis on an Ni-Loaded Catalyst. Nature 2020, 583, 391–395. https://doi.org/10.1038/s41586-020-2464-9.
- 56.
Ma, X.L.; Liu, J.C.; Xiao, H.; et al. Surface Single-Cluster Catalyst for N2-to-NH3 Thermal Conversion. J. Am. Chem. Soc. 2018, 140, 46–49. https://doi.org/10.1021/jacs.7b10354.
- 57.
Liu, J.C.; Ma, X.L.; Li, Y.; et al. Heterogeneous Fe3 Single-Cluster Catalyst for Ammonia Synthesis via an Associative Mechanism. Nat. Commun. 2018, 9, 1610–1619. https://doi.org/10.1038/s41467-018-03795-8.
- 58.
Rostamikia, G.; Maheshwari, S.; Janik, M.J. Elementary Kinetics of Nitrogen Electroreduction to Ammonia on Late Transition Metals. Catal. Sci. Technol. 2019, 9, 174–181. https://doi.org/10.1039/c8cy01845f.
- 59.
Jasniewski, A.J.; Lee, C.C.; Ribbe, M.W.; et al. Reactivity, Mechanism, and Assembly of the Alternative Nitrogenases. Chem. Rev. 2020, 120, 5107–5157. https://doi.org/10.1021/acs.chemrev.9b00704.
- 60.
Cao, L.; Ryde, U. Putative Reaction Mechanism of Nitrogenase after Dissociation of a Sulfide Ligand. J. Catal. 2020, 391, 247–259. https://doi.org/10.1016/j.jcat.2020.08.028.
- 61.
Zhao, J.; Chen, Z. Single Mo Atom Supported on Defective Boron Nitride Monolayer as an Efficient Electrocatalyst for Nitrogen Fixation: A Computational Study. J. Am. Chem. Soc. 2017, 139, 12480–12487. https://doi.org/10.1021/jacs.7b05213.
- 62.
He, T.; Puente Santiago, A.R.; Du, A. Atomically Embedded Asymmetrical Dual-Metal Dimers on N-Doped Graphene for Ultra-Efficient Nitrogen Reduction Reaction. J. Catal. 2020, 388, 77–83. https://doi.org/10.1016/j.jcat.2020.05.009.
- 63.
Ling, C.; Zhang, Y.; Li, Q.; et al. New Mechanism for N2 Reduction: The Essential Role of Surface Hydrogenation. J. Am. Chem. Soc. 2019, 141, 18264–18270. https://doi.org/10.1021/jacs.9b09232.
- 64.
Siegbahn, P.E.M. The Mechanism for Nitrogenase Including All Steps. Phys. Chem. Chem. Phys. 2019, 21, 15747–15759. https://doi.org/10.1039/c9cp02073j.
- 65.
Raugei, S.; Seefeldt, L.C.; Hoffman, B.M. Critical Computational Analysis Illuminates the Reductive-Elimination Mechanism That Activates Nitrogenase for N2 Reduction. Proc. Natl. Acad. Sci. USA 2018, 115, E10521–E10530. https://doi.org/10.1073/pnas.1810211115.
- 66.
Varley, J.B.; Wang, Y.; Chan, K.; et al. Mechanistic Insights into Nitrogen Fixation by Nitrogenase Enzymes. Phys. Chem. Chem. Phys. 2015, 17, 29541–29547. https://doi.org/10.1039/c5cp04034e.
- 67.
Kim, M.-C.; Nam, H.; Choi, J.; et al. Hydrogen Bonding-Mediated Enhancement of Bioinspired Electrochemical Nitrogen Reduction on Cu2−xS Catalysts. ACS Catal. 2020, 10, 10577–10584. https://doi.org/10.1021/acscatal.0c01730.
- 68.
Zhang, J.; Tian, X.; Liu, M.; et al. Cobalt-Modulated Molybdenum-Dinitrogen Interaction in MoS2 for Catalyzing Ammonia Synthesis. J. Am. Chem. Soc. 2019, 141, 19269–19275. https://doi.org/10.1021/jacs.9b02501.
- 69.
Mao, C.; Wang, J.; Zou, Y.; et al. Hydrogen Spillover to Oxygen Vacancy of TiO2−xHγ/Fe: Breaking the Scaling Relationship of Ammonia Synthesis. J. Am. Chem. Soc. 2020, 142, 17403–17412. https://doi.org/10.1021/jacs.0c06118.
- 70.
Marnellos, G.; Stoukides, M. Ammonia Synthesis at Atmospheric Pressure. Science 1998, 282, 98–100. https://doi.org/10.1126/science.282.5386.98.
- 71.
Marnellos, G.; Zisekas, S.; Stoukides, M. Synthesis of Ammonia at Atmospheric Pressure with the Use of Solid State Proton Conductors. J. Catal. 2000, 193, 80–87. https://doi.org/10.1006/jcat.2000.2877.
- 72.
Ouzounidou, M.; Skodra, A.; Kokkofitis, C.; et al. Catalytic and Electrocatalytic Synthesis of NH3 in a H+ Conducting Cell by Using an Industrial Fe Catalyst. Solid State Ionics 2007, 178, 153–159. https://doi.org/10.1016/j.ssi.2006.11.019.
- 73.
Skodra, A.; Stoukides, M. Electrocatalytic Synthesis of Ammonia from Steam and Nitrogen at Atmospheric Pressure. Solid State Ionics 2009, 180, 1332–1336. https://doi.org/10.1016/j.ssi.2009.08.001.
- 74.
Vasileiou, E.; Kyriakou, V.; Garagounis, I.; et al. Electrochemical Enhancement of Ammonia Synthesis in a BaZr0.7Ce0.2Y0.1O2.9 Solid Electrolyte Cell. Solid State Ionics 2016, 288, 357–362. https://doi.org/10.1016/j.ssi.2015.12.022.
- 75.
Hirakawa, H.; Hashimoto, M.; Shiraishi, Y.; et al. Photocatalytic Conversion of Nitrogen to Ammonia with Water on Surface Oxygen Vacancies of Titanium Dioxide. J. Am. Chem. Soc. 2017, 139, 10929–10936. https://doi.org/10.1021/jacs.7b06634.
- 76.
Xie, X.-Y.; Xiao, P.; Fang, W.-H.; et al. Probing Photocatalytic Nitrogen Reduction to Ammonia with Water on the Rutile TiO2 (110) Surface by First-Principles Calculations. ACS Catal. 2019, 9, 9178–9187. https://doi.org/10.1021/acscatal.9b01551.
- 77.
Kuang, M.; Wang, Y.; Fang, W.; et al. Efficient Nitrate Synthesis via Ambient Nitrogen Oxidation with Ru-Doped TiO2/RuO2 Electrocatalysts. Adv. Mater. 2020, 32, 2002189. https://doi.org/10.1002/adma.202002189.
- 78.
Yuan, S.J.; Chen, J.J.; Lin, Z.Q.; et al. Nitrate Formation from Atmospheric Nitrogen and Oxygen Photocatalysed by Nano-Sized Titanium Dioxide. Nat. Commun. 2013, 4, 2249. https://doi.org/10.1038/ncomms3249.
- 79.
Comer, B.M.; Medford, A.J. Analysis of Photocatalytic Nitrogen Fixation on Rutile TiO2(110). ACS Sustainable Chem. Eng. 2018, 6, 4648–4660. https://doi.org/10.1021/acssuschemeng.7b03652.
- 80.
Zhang, L.; Cong, M.; Ding, X.; et al. A Janus Fe-SnO2 Catalyst That Enables Bifunctional Electrochemical Nitrogen Fixation. Angew. Chem. Int. Ed. 2020, 59, 10888–10893. https://doi.org/10.1002/anie.202003518.
- 81.
Liu, Y.; Cheng, M.; He, Z.; et al. Pothole-Rich Ultrathin WO3 Nanosheets that Trigger N≡N Bond Activation of Nitrogen for Direct Nitrate Photosynthesis. Angew. Chem. Int. Ed. 2019, 58, 731–735. https://doi.org/10.1002/anie.201808177.
- 82.
Vasileff, A.; Xu, C.; Jiao, Y.; et al. Surface and Interface Engineering in Copper-Based Bimetallic Materials for Selective CO2 Electroreduction. Chem 2018, 4, 1809–1831. https://doi.org/10.1016/j.chempr.2018.05.001.
- 83.
Andersen, M.; Medford, A.J.; Nørskov, J.K.; et al. Scaling-Relation-Based Analysis of Bifunctional Catalysis: The Case for Homogeneous Bimetallic Alloys. ACS Catal. 2017, 7, 3960–3967. https://doi.org/10.1021/acscatal.7b00482.
- 84.
Andersen, M.; Medford, A.J.; Nørskov, J.K.; et al. Analyzing the Case for Bifunctional Catalysis. Angew. Chem. Int. Ed. 2016, 55, 5210–5214. https://doi.org/10.1002/anie.201601049.
- 85.
Shi, L.; Yin, Y.; Wang, S.; et al. Rational Catalyst Design for N2 Reduction under Ambient Conditions: Strategies toward Enhanced Conversion Efficiency. ACS Catal. 2020, 10, 6870–6899. https://doi.org/10.1021/acscatal.0c01081.
- 86.
Medford, A.J.; Vojvodic, A.; Hummelshøj, J.S.; et al. From the Sabatier Principle to a Predictive Theory of Transition-Metal Heterogeneous Catalysis. J. Catal. 2015, 328, 36–42. https://doi.org/10.1016/j.jcat.2014.12.033.
- 87.
Vojvodic, A.; Nørskov, J.K. New Design Paradigm for Heterogeneous Catalysts. Natl. Sci. Rev. 2015, 2, 140–149. https://doi.org/10.1093/nsr/nwv023.
- 88.
Suryanto, B.H.R.; Wang, D.; Azofra, L.M.; et al. MoS2 Polymorphic Engineering Enhances Selectivity in the Electrochemical Reduction of Nitrogen to Ammonia. ACS Energy Lett. 2018, 4, 430–435. https://doi.org/10.1021/acsenergylett.8b02257.
- 89.
Wang, J.; Yu, L.; Hu, L.; et al. Ambient Ammonia Synthesis via Palladium-Catalyzed Electrohydrogenation of Dinitrogen at Low Overpotential. Nat. Commun. 2018, 9, 1795. https://doi.org/10.1038/s41467-018-04213-9.
- 90.
Lai, F.; Chen, N.; Ye, X.; et al. Refining Energy Levels in ReS2 Nanosheets by Low-Valent Transition-Metal Doping for Dual-Boosted Electrochemical Ammonia/Hydrogen Production. Adv. Funct. Mater. 2020, 30, 1907376. https://doi.org/10.1002/adfm.201907376.
- 91.
Li, J.; Yu, Y.; Jiang, S.P.; et al. Electronic Engineering of Spinels for Advanced Electrocatalysis. Nanoscale Horiz. 2026, 11, 357–374. https://doi.org/10.1039/D5NH00539F.
- 92.
Xu, W.; Fan, G.; Chen, J.; et al. Nanoporous Palladium Hydride for Electrocatalytic N2 Reduction under Ambient Conditions. Angew. Chem. Int. Ed. 2020, 59, 3511–3516. https://doi.org/10.1002/anie.201914335.
- 93.
Fukui, K.; Iimura, S.; Tada, T.; et al. Characteristic Fast H− Ion Conduction in Oxygen-Substituted Lanthanum Hydride. Nat. Commun. 2019, 10, 2578. https://doi.org/10.1038/s41467-019-10492-7.
- 94.
Li, L.; Zhang, T.; Cai, J.; et al. Operando Spectroscopic and Isotopic-Label Directed Observation of LaN-Promoted Ru/ZrH2 Catalyst for Ammonia Synthesis via Associative and Chemical Looping Route. J. Catal. 2020, 389, 218–228. https://doi.org/10.1016/j.jcat.2020.05.039.
- 95.
Lu, F.; Yi, D.; Liu, S.; et al. Engineering Platinum-Oxygen Dual Catalytic Sites via Charge Transfer towards High-Efficient Hydrogen Evolution. Angew. Chem. Int. Ed. 2020, 59, 19460–19467. https://doi.org/10.1002/anie.202008117.
- 96.
Wang, J.; Liu, W.; Luo, G.; et al. Synergistic Effect of Well-Defined Dual Sites Boosting the Oxygen Reduction Reaction. Energy Environ. Sci. 2018, 11, 3375–3379. https://doi.org/10.1039/c8ee02656d.
- 97.
Chen, C.; Yan, X.; Liu, S.; et al. Highly Efficient Electroreduction of CO2 to C2+ Alcohols on Heterogeneous Dual Active Sites. Angew. Chem. Int. Ed. 2020, 59, 16459–16464. https://doi.org/10.1002/anie.202006847.
- 98.
Xiao, B.B.; Yang, L.; Yu, L.B.; et al. The VN3 Embedded Graphane with the Improved Selectivity for Nitrogen Fixation. Appl. Surf. Sci. 2020, 513, 145855. https://doi.org/10.1016/j.apsusc.2020.145855.
- 99.
Zeinalipour-Yazdi, C.D.; Hargreaves, J.S.J.; Catlow, C.R.A. DFT-D3 Study of Molecular N2 and H2 Activation on Co3Mo3N Surfaces. J. Phys. Chem. C 2016, 120, 21390–21398. https://doi.org/10.1021/acs.jpcc.6b04748.
- 100.
Chen, Z.W.; Yan, J.M.; Jiang, Q. Single or Double: Which Is the Altar of Atomic Catalysts for Nitrogen Reduction Reaction? Small Methods 2018, 3, 1800291. https://doi.org/10.1002/smtd.201800291.
- 101.
Wang, S.; Shi, L.; Bai, X.; et al. Highly Efficient Photo-/Electrocatalytic Reduction of Nitrogen into Ammonia by Dual-Metal Sites. ACS Cent. Sci. 2020, 6, 1762–1773. https://doi.org/10.1021/acscentsci.0c00552.
- 102.
Jacobsen, C.J.H.; Dahl, S.; Clausen, B.S.; et al. Catalyst Design by Interpolation in the Periodic Table: Bimetallic Ammonia Synthesis Catalysts. J. Am. Chem. Soc. 2001, 123, 8404–8405. https://doi.org/10.1021/ja010963d.
- 103.
Xue, Z.H.; Zhang, S.N.; Lin, Y.X.; et al. Electrochemical Reduction of N2 into NH3 by Donor-Acceptor Couples of Ni and Au Nanoparticles with a 67.8% Faradaic Efficiency. J. Am. Chem. Soc. 2019, 141, 14976–14980. https://doi.org/10.1021/jacs.9b07963.
- 104.
Han, L.; Ren, Z.; Ou, P.; et al. Modulating Single-Atom Pd Sites with Cu for Enhanced Ambient Ammonia Electrosynthesis. Angew. Chem. Int. Ed. 2021, 60, 345–350. https://doi.org/10.1002/anie.202010159.
- 105.
Liu, W.; Han, L.; Wang, H.-T.; et al. FeMo Sub-Nanoclusters/Single Atoms for Neutral Ammonia Electrosynthesis. Nano Energy 2020, 77, 105078. https://doi.org/10.1016/j.nanoen.2020.105078.
- 106.
Wang, X.; Qiu, S.; Feng, J.; et al. Confined Fe-Cu Clusters as Sub-Nanometer Reactors for Efficiently Regulating the Electrochemical Nitrogen Reduction Reaction. Adv. Mater. 2020, 32, 2004382. https://doi.org/10.1002/adma.202004382.
- 107.
Liu, Y.; Zhang, X.; Chen, Z.; et al. Electrocatalytic Reduction of Nitrogen on FeAg/Si for Ammonia Synthesis: A Simple Strategy for Continuous Regulation of Faradaic Efficiency by Controlling H+ Ions Transfer Rate. Appl. Catal. B 2021, 280, 119606. https://doi.org/10.1016/j.apcatb.2020.119606.
- 108.
Cromer, D.T.; Mills, R.L.; Schiferi, D.; et al. The Structure of N2 at 49 Kbar and 299 K. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1980, B37, 8–11. https://doi.org/10.1107/S0567740881002070.
- 109.
Mitchell, S.; Perez-Ramirez, J. Single Atom Catalysis: A Decade of Stunning Progress and the Promise for a Bright Future. Nat. Commun. 2020, 11, 4302. https://doi.org/10.1038/s41467-020-18182-5.
- 110.
Guo, Y.; Yao, Z.; Zhan, S.; et al. Molybdenum and Boron Synergistically Boosting Efficient Electrochemical Nitrogen Fixation. Nano Energy 2020, 78, 105391. https://doi.org/10.1016/j.nanoen.2020.105391.
- 111.
Dai, C.; Sun, Y.; Chen, G.; et al. Electrochemical Oxidation of Nitrogen towards Direct Nitrate Production on Spinel Oxides. Angew. Chem. Int. Ed. 2020, 59, 9418–9422. https://doi.org/10.1002/anie.202002923.
- 112.
Han, S.; Wang, C.; Wang, Y.; et al. Electrosynthesis of Nitrate via the Oxidation of Nitrogen on Tensile Strained Palladium Porous Nanosheets. Angew. Chem. Int. Ed. 2021, 60, 4474–4478. https://doi.org/10.1002/anie.202014017.
- 113.
Jia, H.P.; Quadrelli, E.A. Mechanistic Aspects of Dinitrogen Cleavage and Hydrogenation to Produce Ammonia in Catalysis and Organometallic Chemistry: Relevance of Metal Hydride Bonds and Dihydrogen. Chem. Soc. Rev. 2014, 43, 547–564. https://doi.org/10.1039/c3cs60206k.
- 114.
Li, P.; Jin, Z.; Fang, Z.; et al. A Surface Strained and Geometry Tailored Nanoreactor Promotes the Ammonia Electrosynthesis. Angew. Chem. Int. Ed. 2020, 59, 22610–22616. https://doi.org/10.1002/anie.202011596.
- 115.
Gevantman, L.H. Solubility of Selected Gases in Water. In CRC Handbook of Chemistry and Physics, 97th ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 5–134.
- 116.
Li, X.; Sun, X.; Zhang, L.; et al. Efficient Photocatalytic Fixation of N2 by KOH-Treated g-C3N4. J. Mater. Chem. A 2018, 6, 3005–3011. https://doi.org/10.1039/c7ta09762j.
- 117.
Singh, A.R.; Rohr, B.A.; Schwalbe, J.A.; et al. Electrochemical Ammonia Synthesis—The Selectivity Challenge. ACS Catal. 2016, 7, 706–709. https://doi.org/10.1021/acscatal.6b03035.
- 118.
Suryanto, B.H.R.; Kang, C.S.M.; Wang, D.; et al. Rational Electrode–Electrolyte Design for Efficient Ammonia Electrosynthesis under Ambient Conditions. ACS Energy Lett. 2018, 3, 1219–1224. https://doi.org/10.1021/acsenergylett.8b00487.
- 119.
Tian, Y.; Liu, Y.; Wang, H.; et al. Electrocatalytic Reduction of Nitrogen to Ammonia in Ionic Liquids. ACS Sustainable Chem. Eng. 2022, 10, 4345–4358. https://doi.org/10.1021/acssuschemeng.2c00018.
- 120.
Zhou, F.; Azofra, L.M.; Ali, M.; et al. Electro-Synthesis of Ammonia from Nitrogen at Ambient Temperature and Pressure in Ionic Liquids. Energy Environ. Sci. 2017, 10, 2516–2520. https://doi.org/10.1039/c7ee02716h.
- 121.
Kang, C.S.M.; Zhang, X.; MacFarlane, D.R. High Nitrogen Gas Solubility and Physicochemical Properties of [C4mpyr][eFAP]—Fluorinated Solvent Mixtures. J. Phys. Chem. C 2019, 123, 21376–21385. https://doi.org/10.1021/acs.jpcc.9b06000.
- 122.
Ortuno, M.A.; Holloczki, O.; Kirchner, B.; et al. Selective Electrochemical Nitrogen Reduction Driven by Hydrogen Bond Interactions at Metal-Ionic Liquid Interfaces. J. Phys. Chem. Lett. 2019, 10, 513–517. https://doi.org/10.1021/acs.jpclett.8b03409.
- 123.
Zou, H.; Rong, W.; Wei, S.; et al. Regulating Kinetics and Thermodynamics of Electrochemical Nitrogen Reduction with Metal Single-Atom Catalysts in a Pressurized Electrolyser. Proc. Natl. Acad. Sci. USA 2020, 117, 29462–29468. https://doi.org/10.1073/pnas.2015108117.
- 124.
Cheng, H.; Cui, P.; Wang, F.; et al. High Efficiency Electrochemical Nitrogen Fixation Achieved with a Lower Pressure Reaction System by Changing the Chemical Equilibrium. Angew. Chem. Int. Ed. 2019, 58, 15541–15547. https://doi.org/10.1002/anie.201910658.
- 125.
Zhang, S.; Duan, G.; Qiao, L.; et al. Electrochemical Ammonia Synthesis from N2 and H2O Catalyzed by Doped LaFeO3 Perovskite under Mild Conditions. Ind. Eng. Chem. Res. 2019, 58, 14602–14607. https://doi.org/10.1021/acs.iecr.9b00833.
- 126.
Qiao, L.; Duan, G.; Zhang, S.; et al. Electrochemical Ammonia Synthesis Catalyzed with a CoFe Layered Double Hydroxide—A New Initiative in Clean Fuel Synthesis. J. Cleaner Prod. 2020, 250, 119525. https://doi.org/10.1016/j.jclepro.2019.119525.
- 127.
Ba, K.; Wang, G.K.; Ye, T.; et al. Single Faceted Two-Dimensional Mo2C Electrocatalyst for Highly Efficient Nitrogen Fixation. ACS Catal. 2020, 10, 7864–7870. https://doi.org/10.1021/acscatal.0c01127.
- 128.
Ji, S.G.; Kim, H.; Park, C.; et al. Underestimation of Platinum Electrocatalysis Induced by Carbon Monoxide Evolved from Graphite Counter Electrodes. ACS Catal. 2020, 10, 10773–10783. https://doi.org/10.1021/acscatal.0c01783.
- 129.
Hanifpour, F.; Sveinbjornson, A.; Canales, C.P.; et al. Preparation of Nafion® Membranes for Reliable Ammonia Quantification in Nitrogen Reduction Reaction Experiments. Angew. Chem. Int. Ed. 2020, 59, 23138–23142. https://doi.org/10.1002/anie.202007998.
- 130.
Liu, H.; Zhang, Y.; Luo, J. The Removal of Inevitable NO Species in Catalysts and the Selection of Appropriate Membrane for Measuring Electrocatalytic Ammonia Synthesis Accurately. J. Energy Chem. 2020, 49, 51–58. https://doi.org/10.1016/j.jechem.2020.01.029.
- 131.
Wang, H.; Chen, Y.; Fan, R.; et al. Selective Electrochemical Reduction of Nitrogen to Ammonia by Adjusting the Three-Phase Interface. Research 2019, 2019, 1401209. https://doi.org/10.34133/2019/1401209.
- 132.
Weekes, D.M.; Salvatore, D.A.; Reyes, A.; et al. Electrolytic CO2 Reduction in a Flow Cell. Acc. Chem. Res. 2018, 51, 910–918. https://doi.org/10.1021/acs.accounts.8b00010.
- 133.
Li, J.; Chen, S.; Quan, F.; et al. Accelerated Dinitrogen Electroreduction to Ammonia via Interfacial Polarization Triggered by Single-Atom Protrusions. Chem 2020, 6, 885–901. https://doi.org/10.1016/j.chempr.2020.01.013.
- 134.
Chen, S.; Perathoner, S.; Ampelli, C.; et al. Electrocatalytic Synthesis of Ammonia at Room Temperature and Atmospheric Pressure from Water and Nitrogen on a Carbon-Nanotube-Based Electrocatalyst. Angew. Chem. Int. Ed. 2017, 56, 2699–2703. https://doi.org/10.1002/anie.201609533.
- 135.
Chen, S.; Perathoner, S.; Ampelli, C.; et al. Room-Temperature Electrocatalytic Synthesis of NH3 from H2O and N2 in a Gas–Liquid–Solid Three-Phase Reactor. ACS Sustain. Chem. Eng. 2017, 5, 7393–7400. https://doi.org/10.1021/acssuschemeng.7b01742.
- 136.
Chen, S.; Perathoner, S.; Ampelli, C.; et al. Enhanced Performance in the Direct Electrocatalytic Synthesis of Ammonia from N2 and H2O by an In-Situ Electrochemical Activation of CNT-Supported Iron Oxide Nanoparticles. J. Energy Chem. 2020, 49, 22–32. https://doi.org/10.1016/j.jechem.2020.01.011.
- 137.
Chen, S.; Perathoner, S.; Ampelli, C.; et al. Direct Synthesis of Ammonia from N2 and H2O on Different Iron Species Supported on Carbon Nanotubes Using a Gas-Phase Electrocatalytic Flow Reactor. ChemElectroChem 2020, 7, 3028–3037. https://doi.org/10.1002/celc.202000514.
- 138.
Cui, X.; Tang, C.; Liu, X.M.; et al. Highly Selective Electrochemical Reduction of Dinitrogen to Ammonia at Ambient Temperature and Pressure over Iron Oxide Catalysts. Chem. Eur. J. 2018, 24, 18494–18501. https://doi.org/10.1002/chem.201800535.
- 139.
Wei, X.; Vogel, D.; Keller, L.; et al. Microtubular Gas Diffusion Electrode Based on Ruthenium-Carbon Nanotubes for Ambient Electrochemical Nitrogen Reduction to Ammonia. ChemElectroChem 2020, 7, 4679–4684. https://doi.org/10.1002/celc.202001370.
- 140.
Liu, Y.; Huang, B.; Chen, X.; et al. Electrocatalytic Production of Ammonia: Biomimetic Electrode–Electrolyte Design for Efficient Electrocatalytic Nitrogen Fixation under Ambient Conditions. Appl. Catal. B 2020, 271, 118919. https://doi.org/10.1016/j.apcatb.2020.118919.
- 141.
Salvatore, D.; Berlinguette, C.P. Voltage Matters When Reducing CO2 in an Electrochemical Flow Cell. ACS Energy Lett. 2019, 5, 215–220. https://doi.org/10.1021/acsenergylett.9b02356.
- 142.
Zhang, Z.; Melo, L.; Jansonius, R.P.; et al. pH Matters When Reducing CO2 in an Electrochemical Flow Cell. ACS Energy Lett. 2020, 5, 3101–3107. https://doi.org/10.1021/acsenergylett.0c01606.
- 143.
Kani, N.C.; Prajapati, A.; Collins, B.A.; et al. Competing Effects of pH, Cation Identity, H2O Saturation, and N2 Concentration on the Activity and Selectivity of Electrochemical Reduction of N2 to NH3 on Electrodeposited Cu at Ambient Conditions. ACS Catal. 2020, 10, 14592–14603. https://doi.org/10.1021/acscatal.0c04864.
- 144.
Back, S.; Jung, Y. On the Mechanism of Electrochemical Ammonia Synthesis on the Ru Catalyst. Phys. Chem. Chem. Phys. 2016, 18, 9161–9166. https://doi.org/10.1039/c5cp07363d.
- 145.
Li, Q.; Qiu, S.; He, L.; et al. Impact of H-Termination on the Nitrogen Reduction Reaction of Molybdenum Carbide as an Electrochemical Catalyst. Phys. Chem. Chem. Phys. 2018, 20, 23338–23343. https://doi.org/10.1039/c8cp04474k.
- 146.
Zheng, J.; Lyu, Y.; Qiao, M.; et al. Photoelectrochemical Synthesis of Ammonia on the Aerophilic-Hydrophilic Heterostructure with 37.8% Efficiency. Chem 2019, 5, 617–633. https://doi.org/10.1016/j.chempr.2018.12.003.
- 147.
Guan, R.; Wang, D.; Zhang, Y.; et al. Enhanced Photocatalytic N2 Fixation via Defective and Fluoride Modified TiO2 Surface. Appl. Catal. B 2021, 282, 119580. https://doi.org/10.1016/j.apcatb.2020.119580.
- 148.
Sun, Y.; Jiang, T.; Duan, J.; et al. Two-Dimensional Nanomesh Arrays as Bifunctional Catalysts for N2 Electrolysis. ACS Catal. 2020, 10, 11371–11379. https://doi.org/10.1021/acscatal.0c02745.
- 149.
Qin, M.; Li, X.; Gan, G.; et al. Boosting Electrocatalytic Nitrogen Fixation with Co–N3 Site-Decorated Porous Carbon. ACS Sustainable Chem. Eng. 2020, 8, 13430–13439. https://doi.org/10.1021/acssuschemeng.0c04021.
- 150.
Wang, H.; Wang, L.; Wang, Q.; et al. Ambient Electrosynthesis of Ammonia: Electrode Porosity and Composition Engineering. Angew. Chem. Int. Ed. 2018, 57, 12360–12364. https://doi.org/10.1002/anie.201805514.
- 151.
Li, H.; Shang, J.; Ai, Z.; et al. Efficient Visible Light Nitrogen Fixation with BiOBr Nanosheets of Oxygen Vacancies on the Exposed {001} Facets. J. Am. Chem. Soc. 2015, 137, 6393–6399. https://doi.org/10.1021/jacs.5b03105.
- 152.
Ma, X.; Liu, Z.; Sun, H.; et al. Cu(N2)-Li Battery for Ammonia Synthesis. J. Phys. Chem. Lett. 2024, 15, 6435–6442. https://doi.org/10.1021/acs.jpclett.4c01328.
- 153.
Ma, X.; Li, J.; Zhou, H.; et al. Li-N2 Battery for Ammonia Synthesis and Computational Insight. ACS Appl. Mater. Interfaces 2023, 15, 19032–19042. https://doi.org/10.1021/acsami.3c01929.
- 154.
Ma, X.; Li, J.; Zhou, H.; et al. Continuous Ammonia Synthesis Using Ru Nanoparticles Based on Li-N2 Battery. Mater. Today Energy 2022, 29, 101113–101122. https://doi.org/10.1016/j.mtener.2022.101113.
- 155.
Ma, X.; Sun, H.; Li, J.; et al. “Continuous” Nitrogen Reduction Synthesis of Ammonia Based on Li-N2 Battery System. Acta Chim. Sinica 2022, 80, 861–866. https://doi.org/10.6023/A22020088.
- 156.
Ma, X.; Ma, Y.; Sun, H.; et al. Cycle Issue and Failure Analysis of Li–N2 Batteries. ACS Sustain. Chem. Eng. 2025, 13, 2521–2528. https://doi.org/10.1021/acssuschemeng.4c09349.
- 157.
Mangini, A.; Garbujo, A.; Biasi, P.; et al. Debunking Pitfalls of Li–N2 Cells for Ammonia Electroproduction: Is This Setup Affordable to Prove Nitro-Fixation before Lithium Plating? ACS Electrochem. 2025, 1, 2866–2877. https://doi.org/10.1021/acselectrochem.5c00402.
- 158.
Liu, C.; Li, Q.; Wu, C.; et al. Single-Boron Catalysts for Nitrogen Reduction Reaction. J. Am. Chem. Soc. 2019, 141, 2884–2888. https://doi.org/10.1021/jacs.8b13165.
- 159.
Ling, C.; Niu, X.; Li, Q.; et al. Metal-Free Single Atom Catalyst for N2 Fixation Driven by Visible Light. J. Am. Chem. Soc. 2018, 140, 14161–14168. https://doi.org/10.1021/jacs.8b07472.
- 160.
Hao, Q.; Liu, C.; Jia, G.; et al. Catalytic Reduction of Nitrogen to Produce Ammonia by Bismuth-Based Catalysts: State of the Art and Future Prospects. Mater. Horiz. 2020, 7, 1014–1029. https://doi.org/10.1039/c9mh01668f.
- 161.
Choi, J.; Du, H.-L.; Nguyen, C.K.; et al. Electroreduction of Nitrates, Nitrites, and Gaseous Nitrogen Oxides: A Potential Source of Ammonia in Dinitrogen Reduction Studies. ACS Energy Lett. 2020, 5, 2095–2097. https://doi.org/10.1021/acsenergylett.0c00924.
- 162.
MacFarlane, D.; Simonov, A.; Suryanto, B.H.R.; et al. Promoting Nitrogen Electroreduction to Ammonia with Bismuth Nanocrystals and Potassium Cations in Water. ChemRxiv 2020. https://doi.org/10.26434/chemrxiv.11768814.v1.
- 163.
Mao, C.; Wang, J.; Zou, Y.; et al. Anion (O, N, C, and S) Vacancies Promoted Photocatalytic Nitrogen Fixation. Green Chem. 2019, 21, 2852–2867. https://doi.org/10.1039/c9gc01010f.
- 164.
Liu, Y.; Kong, X.; Guo, X.; et al. Enhanced N2 Electroreduction over LaCoO3 by Introducing Oxygen Vacancies. ACS Catal. 2019, 10, 1077–1085. https://doi.org/10.1021/acscatal.9b03864.
- 165.
Yang, X.; Ling, F.; Su, J.; et al. Insights into the Role of Cation Vacancy for Significantly Enhanced Electrochemical Nitrogen Reduction. Appl. Catal. B 2020, 264, 118477. https://doi.org/10.1016/j.apcatb.2019.118477.
- 166.
Peng, W.; Luo, M.; Xu, X.; et al. Spontaneous Atomic Ruthenium Doping in Mo2CTX MXene Defects Enhances Electrocatalytic Activity for the Nitrogen Reduction Reaction. Adv. Energy Mater. 2020, 10, 2001364. https://doi.org/10.1002/aenm.202001364.
- 167.
Yao, D.; Tang, C.; Li, L.; et al. In Situ Fragmented Bismuth Nanoparticles for Electrocatalytic Nitrogen Reduction. Adv. Energy Mater. 2020, 10, 2001289. https://doi.org/10.1002/aenm.202001289.
- 168.
Liu, S.; Wang, M.; Qian, T.; et al. Facilitating Nitrogen Accessibility to Boron-Rich Covalent Organic Frameworks via Electrochemical Excitation for Efficient Nitrogen Fixation. Nat. Commun. 2019, 10, 3898. https://doi.org/10.1038/s41467-019-11846-x.
- 169.
Manjunatha, R.; Schechter, A. Electrochemical Synthesis of Ammonia Using Ruthenium–Platinum Alloy at Ambient Pressure and Low Temperature. Electrochem. Commun. 2018, 90, 96–100. https://doi.org/10.1016/j.elecom.2018.04.008.
- 170.
de Araujo, R.G.; Perez, J. Nitrogen Electrochemical Reduction Reaction Pathways Evidenced by Online Electrochemical Mass Spectrometry and Isotope Labeling on the MoS2 Surface. ACS Electrochem. 2025, 1, 294–302. https://doi.org/10.1021/acselectrochem.4c00135.
- 171.
Yao, Y.; Wang, H.; Yuan, X.-Z.; et al. Electrochemical Nitrogen Reduction Reaction on Ruthenium. ACS Energy Lett. 2019, 4, 1336–1341. https://doi.org/10.1021/acsenergylett.9b00699.
- 172.
Yao, Y.; Zhu, S.; Wang, H.; et al. A Spectroscopic Study on the Nitrogen Electrochemical Reduction Reaction on Gold and Platinum Surfaces. J. Am. Chem. Soc. 2018, 140, 1496–1501. https://doi.org/10.1021/jacs.7b12101.
- 173.
Yao, Y.; Zhu, S.; Wang, H.; et al. A Spectroscopic Study of Electrochemical Nitrogen and Nitrate Reduction on Rhodium Surfaces. Angew. Chem. Int. Ed. 2020, 59, 10479–10483. https://doi.org/10.1002/anie.202003071.
- 174.
Qu, X.; Shen, L.; Mao, Y.; et al. Facile Preparation of Carbon Shells-Coated O-Doped Molybdenum Carbide Nanoparticles as High Selective Electrocatalysts for Nitrogen Reduction Reaction under Ambient Conditions. ACS Appl. Mater. Interfaces 2019, 11, 31869–31877. https://doi.org/10.1021/acsami.9b09007.
- 175.
Yang, X.; Nash, J.; Anibal, J.; et al. Mechanistic Insights into Electrochemical Nitrogen Reduction Reaction on Vanadium Nitride Nanoparticles. J. Am. Chem. Soc. 2018, 140, 13387–13391. https://doi.org/10.1021/jacs.8b08379.
- 176.
Luo, S.; Li, X.; Wang, M.; et al. Long-Term Electrocatalytic N2 Fixation by MOF-Derived Y-Stabilized ZrO2: Insight into the Deactivation Mechanism. J. Mater. Chem. A 2020, 8, 5647–5654. https://doi.org/10.1039/d0ta01154a.
- 177.
Chen, G.F.; Ren, S.; Zhang, L.; et al. Advances in Electrocatalytic N2 Reduction-Strategies to Tackle the Selectivity Challenge. Small Methods 2018, 3, 1800337–1800357. https://doi.org/10.1002/smtd.201800337.
- 178.
Singh, A.R.; Rohr, B.A.; Statt, M.J.; et al. Strategies toward Selective Electrochemical Ammonia Synthesis. ACS Catal. 2019, 9, 8316–8324. https://doi.org/10.1021/acscatal.9b02245.
- 179.
Su, H.; Chen, L.; Chen, Y.; et al. Single Atoms of Iron on MoS2 Nanosheets for N2 Electroreduction into Ammonia. Angew. Chem. Int. Ed. 2020, 59, 20411–20416. https://doi.org/10.1002/anie.202009217.
- 180.
Sim, H.Y.F.; Chen, J.R.T.; Koh, C.S.L.; et al. ZIF-Induced d-Band Modification in a Bimetallic Nanocatalyst: Achieving Over 44% Efficiency in the Ambient Nitrogen Reduction Reaction. Angew. Chem. Int. Ed. 2020, 59, 16997–17003. https://doi.org/10.1002/anie.202006071.
- 181.
Mukherjee, S.; Yang, X.; Shan, W.; et al. Atomically Dispersed Single Ni Site Catalysts for Nitrogen Reduction toward Electrochemical Ammonia Synthesis Using N2 and H2O. Small Methods 2020, 4, 1900821. https://doi.org/10.1002/smtd.201900821.
- 182.
Zhao, H.; Zhang, D.; Li, H.; et al. Exposure of Definite Palladium Facets Boosts Electrocatalytic Nitrogen Fixation at Low Overpotential. Adv. Energy Mater. 2020, 10, 2002131. https://doi.org/10.1002/aenm.202002131.
- 183.
Bohra, D.; Chaudhry, J.H.; Burdyny, T.; et al. Modeling the Electrical Double Layer to Understand the Reaction Environment in a CO2 Electrocatalytic System. Energy Environ. Sci. 2019, 12, 3380–3389. https://doi.org/10.1039/c9ee02485a.
- 184.
Xue, S.; Garlyyev, B.; Watzele, S.; et al. Influence of Alkali Metal Cations on the Hydrogen Evolution Reaction Activity of Pt, Ir, Au, and Ag Electrodes in Alkaline Electrolytes. ChemElectroChem 2018, 5, 2326–2329. https://doi.org/10.1002/celc.201800690.
- 185.
Guha, A.; Narayanaru, S.; Narayanan, T.N. Tuning the Hydrogen Evolution Reaction on Metals by Lithium Salt. ACS Appl. Energy Mater. 2018, 1, 7116–7122. https://doi.org/10.1021/acsaem.8b01546.
- 186.
Perini, N.; Ticianelli, E.A. Oxygen Evolution on Gold: The Effects of Alkali-Metal Cations and Iron Impurities from Alkaline Electrolytes. J. Catal. 2019, 378, 277–282. https://doi.org/10.1016/j.jcat.2019.09.003.
- 187.
Garcia, A.C.; Touzalin, T.; Nieuwland, C.; et al. Enhancement of Oxygen Evolution Activity of Nickel Oxyhydroxide by Electrolyte Alkali Cations. Angew. Chem. Int. Ed. 2019, 58, 12999–13003. https://doi.org/10.1002/anie.201905501.
- 188.
Guha, A.; Narayanaru, S.; Kaley, N.M.; et al. Mechanistic Insight into High Yield Electrochemical Nitrogen Reduction to Ammonia Using Lithium Ions. Mater. Today Commun. 2019, 21, 100700. https://doi.org/10.1016/j.mtcomm.2019.100700.
- 189.
Liu, K.W.; Ma, Y.B.; Guo, Y.; et al. Functional Binder with Enhanced Chemical Adsorption for Black Phosphorus Anode in Lithium-Ion Capacitors. Adv. Funct. Mater. 2024, 34, 2410451. https://doi.org/10.1002/adfm.202410451.
- 190.
Hao, Y.-C.; Guo, Y.; Chen, L.-W.; et al. Promoting Nitrogen Electroreduction to Ammonia with Bismuth Nanocrystals and Potassium Cations in Water. Nat. Catal. 2019, 2, 448–456. https://doi.org/10.1038/s41929-019-0241-7.
- 191.
Song, Y.; Johnson, D.; Peng, R.; et al. A Physical Catalyst for the Electrolysis of Nitrogen to Ammonia. Sci. Adv. 2018, 4, e1700336. https://doi.org/10.1126/sciadv.1700336.
- 192.
Lindley, B.M.; Appel, A.M.; Krogh-Jespersen, K.; et al. Evaluating the Thermodynamics of Electrocatalytic N2 Reduction in Acetonitrile. ACS Energy Lett. 2016, 1, 698–704. https://doi.org/10.1021/acsenergylett.6b00319.
- 193.
Zhang, L.; Sharada, S.M.; Singh, A.R.; et al. A Theoretical Study of the Effect of a Non-Aqueous Proton Donor on Electrochemical Ammonia Synthesis. Phys. Chem. Chem. Phys. 2018, 20, 4982–4989. https://doi.org/10.1039/c7cp05484j.
- 194.
Kim, K.; Lee, N.; Yoo, C.-Y.; et al. Communication—Electrochemical Reduction of Nitrogen to Ammonia in 2-Propanol under Ambient Temperature and Pressure. J. Electrochem. Soc. 2016, 163, F610–F612. https://doi.org/10.1149/2.0231607jes.
- 195.
Köleli, F.; Röpke, T. Electrochemical Hydrogenation of Dinitrogen to Ammonia on a Polyaniline Electrode. Appl. Catal. B 2006, 62, 306–310. https://doi.org/10.1016/j.apcatb.2005.08.006.
- 196.
Kim, K.; Yoo, C.-Y.; Kim, J.-N.; et al. Electrochemical Synthesis of Ammonia from Water and Nitrogen in Ethylenediamine under Ambient Temperature and Pressure. J. Electrochem. Soc. 2016, 163, F1523–F1526. https://doi.org/10.1149/2.0741614jes.
- 197.
Sheets, B.L.; Botte, G.G. Electrochemical Nitrogen Reduction to Ammonia under Mild Conditions Enabled by a Polymer Gel Electrolyte. Chem. Commun. 2018, 54, 4250–4253. https://doi.org/10.1039/C8CC00657A.
- 198.
Koh, C.S.L.; Lee, H.K.; Sim, H.Y.F.; et al. Turning Water from a Hindrance to the Promotor of Preferential Electrochemical Nitrogen Reduction. Chem. Mater. 2020, 32, 1674–1683. https://doi.org/10.1021/acs.chemmater.9b05313.
- 199.
Lee, H.K.; Koh, C.S.L.; Lee, Y.H.; et al. Favoring the Unfavored: Selective Electrochemical Nitrogen Fixation Using a Reticular Chemistry Approach. Sci. Adv. 2018, 4, eaar3208. https://doi.org/10.1126/sciadv.aar3208.
- 200.
Yang, Y.; Wang, S.Q.; Wen, H.; et al. Nanoporous Gold Embedded ZIF Composite for Enhanced Electrochemical Nitrogen Fixation. Angew. Chem. Int. Ed. 2019, 58, 15362–15366. https://doi.org/10.1002/anie.201909770.
- 201.
Du, C.; Gao, Y.; Wang, J.; et al. Achieving 59% Faradaic Efficiency of the N2 Electroreduction Reaction in an Aqueous Zn-N2 Battery by Facilely Regulating the Surface Mass Transport on Metallic Copper. Chem. Commun. 2019, 55, 12801–12804. https://doi.org/10.1039/c9cc05978d.
- 202.
Chen, Y.; Liu, H.; Ha, N.; et al. Revealing Nitrogen-Containing Species in Commercial Catalysts Used for Ammonia Electrosynthesis. Nat. Catal. 2020, 3, 1055–1061. https://doi.org/10.1038/s41929-020-00527-4.
- 203.
Andersen, S.Z.; Čolić, V.; Yang, S.; et al. A Rigorous Electrochemical Ammonia Synthesis Protocol with Quantitative Isotope Measurements. Nature 2019, 570, 504–508. https://doi.org/10.1038/s41586-019-1260-x.
- 204.
Choi, J.; Suryanto, B.H.R.; Wang, D.; et al. Identification and Elimination of False Positives in Electrochemical Nitrogen Reduction Studies. Nat. Commun. 2020, 11, 5546–5556. https://doi.org/10.1038/s41467-020-19130-z.
- 205.
Dahl, S.; Logadottir, A.; Jacobsen, C.J.H.; et al. Electronic Factors in Catalysis: The Volcano Curve and the Effect of Promotion in Catalytic Ammonia Synthesis. Appl. Catal. A 2001, 222, 19–29. https://doi.org/10.1016/S0926-860X(01)00826-2.
- 206.
Kibsgaard, J.; Nørskov, J.K.; Chorkendorff, I. The Difficulty of Proving Electrochemical Ammonia Synthesis. ACS Energy Lett. 2019, 4, 2986–2988. https://doi.org/10.1021/acsenergylett.9b02286.
- 207.
Medford, A.J.; Hatzell, M.C. Photon-Driven Nitrogen Fixation: Current Progress, Thermodynamic Considerations, and Future Outlook. ACS Catal. 2017, 7, 2624–2643. https://doi.org/10.1021/acscatal.7b00439.
- 208.
Rouwenhorst, K.H.R.; Engelmann, Y.; van ‘t Veer, K.; et al. Plasma-Driven Catalysis: Green Ammonia Synthesis with Intermittent Electricity. Green Chem. 2020, 22, 6258–6287. https://doi.org/10.1039/d0gc02058c.
- 209.
NIST-JANAF Thermochemical Tables. Available online: https://janaf.nist.gov/ (accessed on 20 March 2025).

This work is licensed under a Creative Commons Attribution 4.0 International License.


