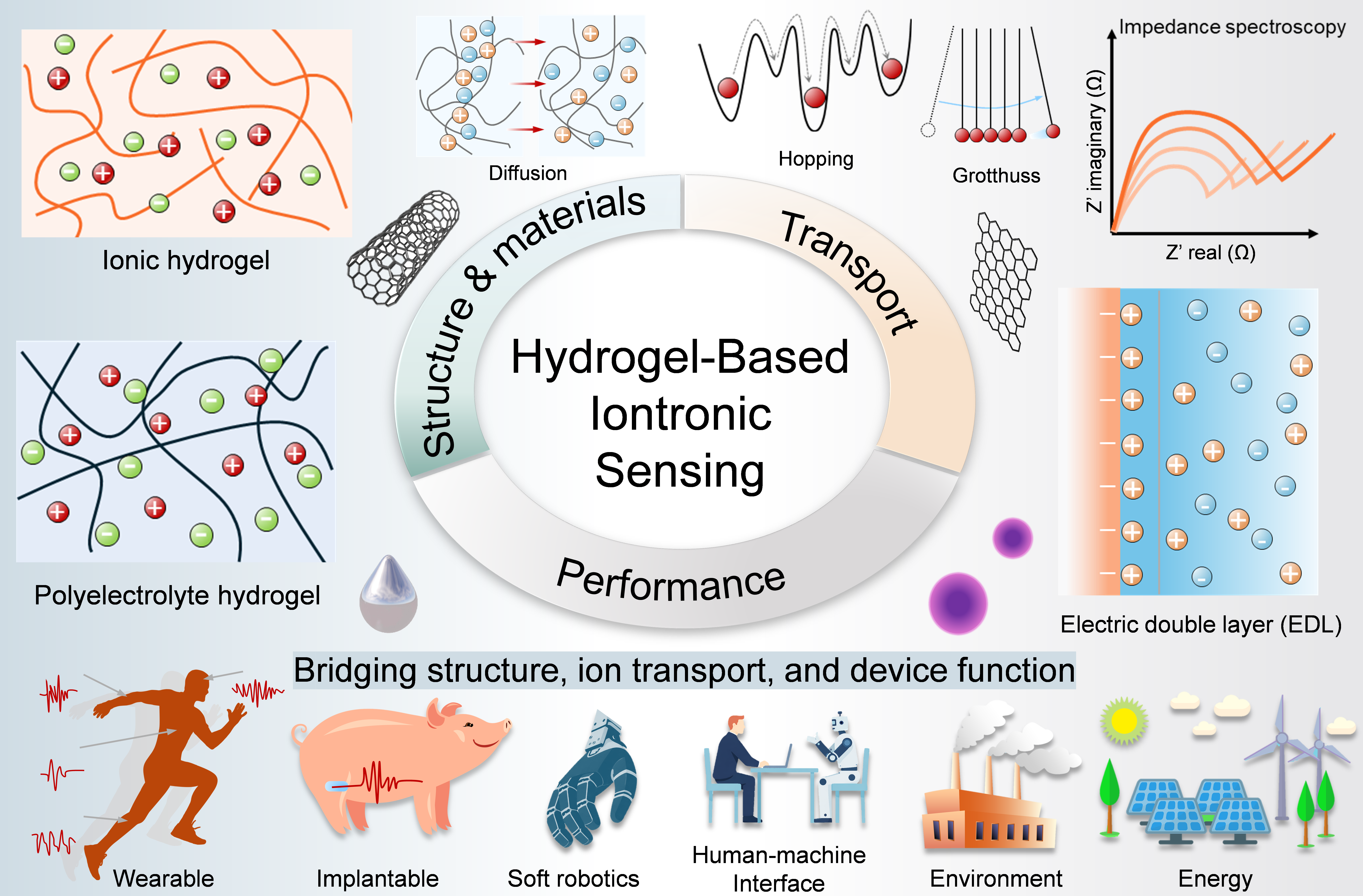
Hydrogel-based iontronic sensing (HBIS) is emerging as a compelling frontier at the interface of soft matter, electrochemistry, and bioelectronics, driven by the unique ability of hydrogels to communicate with living systems through hydrated ionic networks rather than electronic conduction in rigid solids. This ionic mode of signal transduction enables intrinsically compliant interactions with biological tissues, electrolytes, and dynamic fluidic microenvironments, making HBIS highly attractive for flexible bioelectronics. As the field evolves from material optimization toward integrated and intelligent systems, a unifying understanding of material design, ion transport, and device function remains lacking. Here, a multiscale quantitative framework is established to bridge structure, transport, and sensing performance in HBIS. Representative material platforms, including polyelectrolytes, ionogels, and nanocomposite hydrogels, are examined alongside key transport mechanisms, such as diffusion, electromigration, convection, electroosmotic flow, adsorption-site hopping, and Grotthuss-type conduction. Quantitative modeling approaches based on electrochemical impedance spectroscopy and Poisson-Nernst-Planck theory are further highlighted, together with emerging applications in iontronic skins, soft robotics, human-machine interfaces, and energy conversion. This review provides a design blueprint for next-generation hydrogel iontronic sensors with improved predictability, adaptability, and operational stability across biological interfaces.
- Open Access
- Review
Hydrogel-Based Iontronic Sensing
Author Information
Received: 30 Jan 2026 | Revised: 08 Apr 2026 | Accepted: 14 Apr 2026 | Published: 28 Apr 2026
Abstract
Graphical Abstract
Keywords
hydrogels | iontronics | ion transport | wearable devices | bioelectronic interfaces
References
- 1.
Zhang, J.; Wang, M. Alternative Micro/Nanofabrication Approaches for Wearable Electronics. Chem. Rev. 2026, 126, 1686–1762. https://doi.org/10.1021/acs.chemrev.5c00801.
- 2.
Luo, Y.; Abidian, M.R.; Ahn, J.-H.; et al. Technology roadmap for flexible sensors. ACS Nano 2023, 17, 5211–5295. https://doi.org/10.1021/acsnano.2c12606.
- 3.
Tang, W.; Sun, Q.; Wang, Z.L. Self-powered sensing in wearable electronics—A paradigm shift technology. Chem. Rev. 2023, 123, 12105–12134. https://doi.org/10.1021/acs.chemrev.3c00305.
- 4.
Niu, Y.; Liu, H.; He, R.; et al. The new generation of soft and wearable electronics for health monitoring in varying environment: From normal to extreme conditions. Mater. Today 2020, 41, 219–242. https://doi.org/10.1016/j.mattod.2020.10.004.
- 5.
Zhao, C.; Park, J.; Root, S.E.; et al. Skin-inspired soft bioelectronic materials, devices and systems. Nat. Rev. Bioeng. 2024, 2, 671–690. https://doi.org/10.1038/s44222-024-00194-1.
- 6.
Wang, Y.; Haick, H.; Guo, S.; et al. Skin bioelectronics towards long-term, continuous health monitoring. Chem. Soc. Rev. 2022, 51, 3759–3793. https://doi.org/10.1039/d2cs00207h.
- 7.
Park, J.; Lee, Y.; Cho, S.; et al. Soft sensors and actuators for wearable human–machine interfaces. Chem. Rev. 2024, 124, 1464–1534. https://doi.org/10.1021/acs.chemrev.3c00356.
- 8.
Hou, Y.; Hou, X. Bioinspired nanofluidic iontronics. Science 2021, 373, 628–629. https://doi.org/10.1126/science.abj0437.
- 9.
Ro, Y.G.; Na, S.; Kim, J.; et al. Iontronics: Neuromorphic Sensing and Energy Harvesting. ACS Nano 2025, 19, 24425–24507. https://doi.org/10.1021/acsnano.5c04885.
- 10.
Yoo, H.; Lee, Y.H.; Lee, M.-G.; et al. Gel-Based Ionic Circuits. Chem. Rev. 2025, 125, 8956–9011. https://doi.org/10.1021/acs.chemrev.5c00245.
- 11.
Yuan, H.; Zhang, Q.; Cheng, Y.; et al. Double-sided microstructured flexible iontronic pressure sensor with wide linear sensing range. J. Colloid Interface Sci. 2024, 670, 41–49. https://doi.org/10.1016/j.jcis.2024.05.054.
- 12.
Zhu, J.; Tao, J.; Yan, W.; et al. Pathways toward wearable and high-performance sensors based on hydrogels: Toughening networks and conductive networks. Natl. Sci. Rev. 2023, 10, nwad180. https://doi.org/10.1093/nsr/nwad180.
- 13.
Roy, A.; Afshari, R.; Jain, S.; et al. Advances in conducting nanocomposite hydrogels for wearable biomonitoring. Chem. Soc. Rev. 2025, 54, 2595–2652. https://doi.org/10.1039/d4cs00220b.
- 14.
Li, W.; Zhou, R.; Ouyang, Y.; et al. Harnessing biomimicry for controlled adhesion on material surfaces. Small 2024, 20, 2401859. https://doi.org/10.1002/smll.202401859.
- 15.
Li, Y.; Bai, N.; Chang, Y.; et al. Flexible iontronic sensing. Chem. Soc. Rev. 2025, 54, 4651–4700. https://doi.org/10.1039/d4cs00870g.
- 16.
Wang, Z.; Wei, H.; Huang, Y.; et al. Naturally sourced hydrogels: Emerging fundamental materials for next-generation healthcare sensing. Chem. Soc. Rev. 2023, 52, 2992–3034. https://doi.org/10.1039/d2cs00813k.
- 17.
Li, W.; Guan, Q.; Li, M.; et al. Nature-inspired strategies for the synthesis of hydrogel actuators and their applications. Prog. Polym. Sci. 2023, 140, 101665. https://doi.org/10.1016/j.progpolymsci.2023.101665.
- 18.
Choi, S.-G.; Kang, S.-H.; Lee, J.-Y.; et al. Recent advances in wearable iontronic sensors for healthcare applications. Front. Bioeng. Biotechnol. 2023, 11, 1335188. https://doi.org/10.3389/fbioe.2023.1335188.
- 19.
Mo, F.; Lin, Y.; Liu, Y.; et al. Advances in ionic conductive hydrogels for skin sensor applications. Mater. Sci. Eng. R Rep. 2025, 165, 100989. https://doi.org/10.1016/j.mser.2025.100989.
- 20.
Chen, W.; Zhai, L.; Zhang, S.; et al. Cascade-heterogated biphasic gel iontronics for electronic-to-multi-ionic signal transmission. Science 2023, 382, 559–565. https://doi.org/10.1126/science.adg0059.
- 21.
Gao, N.; Pan, C. Intelligent ion gels: Design, performance, and applications. SmartMat 2024, 5, e1215. https://doi.org/10.1002/smm2.1215.
- 22.
Chen, X.; Xia, X.; Guo, C.F. Flexible iontronic sensing: Ionic materials, electrodes, and encapsulation. Adv. Funct. Mater. 2025, 36, e12920. https://doi.org/10.1002/adfm.202512920.
- 23.
Liu, Q.; Liu, Z.; Li, C.; et al. Highly transparent and flexible iontronic pressure sensors based on an opaque to transparent transition. Adv. Sci. 2020, 7, 2000348. https://doi.org/10.1002/advs.202000348.
- 24.
Boateng, D.; Li, X.; Zhu, Y.; et al. Recent advances in flexible hydrogel sensors: Enhancing data processing and machine learning for intelligent perception. Biosens. Bioelectron. 2024, 261, 116499. https://doi.org/10.1016/j.bios.2024.116499.
- 25.
Lee, J.; Ihle, S.J.; Pellegrino, G.S.; et al. Stretchable and suturable fibre sensors for wireless monitoring of connective tissue strain. Nat. Electron. 2021, 4, 291–301. https://doi.org/10.1038/s41928-021-00557-1.
- 26.
Zhao, C.; Wang, Y.; Tang, G.; et al. Ionic flexible sensors: Mechanisms, materials, structures, and applications. Adv. Funct. Mater. 2022, 32, 2110417. https://doi.org/10.1002/adfm.202110417.
- 27.
Chen, J.; Zhu, Y.; Chang, X.; et al. Recent progress in essential functions of soft electronic skin. Adv. Funct. Mater. 2021, 31, 2104686. https://doi.org/10.1002/adfm.202104686.
- 28.
Schroeder, T.B.; Guha, A.; Lamoureux, A.; et al. An electric-eel-inspired soft power source from stacked hydrogels. Nature 2017, 552, 214–218. https://doi.org/10.1038/nature24670.
- 29.
Choi, K.; Lee, G.; Lee, M.-G.; et al. Bio-Inspired Ionic Sensors: Transforming Natural Mechanisms into Sensory Technologies. Nano-Micro Lett. 2025, 17, 180. https://doi.org/10.1007/s40820-025-01692-6.
- 30.
Lu, Q.; Li, H.; Cai, C.; et al. Hydrophobic deep eutectic solvent-based eutectogels for long-term humidity resistance and multifunctional sensing. Mater. Horiz. 2025, 12, 9801–9807. https://doi.org/10.1039/d5mh01293g.
- 31.
Cui, X.; Xi, Y.; Tu, S.; et al. An overview of flexible sensors from ionic liquid-based gels. TrAC Trends Anal. Chem. 2024, 174, 117662. https://doi.org/10.1016/j.trac.2024.117662.
- 32.
Shi, J.; Xie, S.; Liu, Z.; et al. Non-hygroscopic ionogel-based humidity-insensitive iontronic sensor arrays for intra-articular pressure sensing. Natl. Sci. Rev. 2024, 11, nwae351. https://doi.org/10.1093/nsr/nwae351.
- 33.
Balakrishnan, G.; Song, J.; Khair, A.S.; et al. Poisson–Nernst–Planck framework for modelling ionic strain and temperature sensors. J. Mater. Chem. B 2023, 11, 5544–5551. https://doi.org/10.1039/d2tb02819k.
- 34.
Lu, D.; Chen, H. Solid-state organic electrochemical transistors (OECTs) based on gel electrolytes for biosensors and bioelectronics. J. Mater. Chem. A 2025, 13, 136–157. https://doi.org/10.1039/d4ta05288a.
- 35.
Yang, C.; Suo, Z. Hydrogel ionotronics. Nat. Rev. Mater. 2018, 3, 125–142. https://doi.org/10.1038/s41578-018-0018-7.
- 36.
Yiming, B.; Guo, X.; Ali, N.; et al. Ambiently and mechanically stable ionogels for soft ionotronics. Adv. Funct. Mater. 2021, 31, 2102773. https://doi.org/10.1002/adfm.202102773.
- 37.
Yuk, H.; Lu, B.; Zhao, X. Hydrogel bioelectronics. Chem. Soc. Rev. 2019, 48, 1642–1667. https://doi.org/10.1039/c8cs00595h.
- 38.
Liu, X.; Liu, J.; Lin, S.; et al. Hydrogel machines. Mater. Today 2020, 36, 102–124. https://doi.org/10.1016/j.mattod.2019.12.026.
- 39.
Li, G.; Li, C.; Li, G.; et al. Development of conductive hydrogels for fabricating flexible strain sensors. Small 2022, 18, 2101518. https://doi.org/10.1002/smll.202101518.
- 40.
Zhou, C.; Wu, T.; Xie, X.; et al. Advances and challenges in conductive hydrogels: From properties to applications. Eur. Polym. J. 2022, 177, 111454. https://doi.org/10.1016/j.eurpolymj.2022.111454.
- 41.
Hou, W.; Sheng, N.; Zhang, X.; et al. Design of injectable agar/NaCl/polyacrylamide ionic hydrogels for high performance strain sensors. Carbohydr. Polym. 2019, 211, 322–328. https://doi.org/10.1016/j.carbpol.2019.01.094.
- 42.
Zhang, D.; Chen, H.; Zhang, Y.; et al. Antifreezing hydrogels: From mechanisms and strategies to applications. Chem. Soc. Rev. 2025, 54, 5292–5341. https://doi.org/10.1039/d4cs00718b.
- 43.
Liza, L.; Kabir, M.H.; Kabir, M.; et al. Flexible fabric-integrated PAAm–LiCl hydrogel pressure sensor for wearable and soft robotics applications. Mater. Adv. 2025, 6, 9590–9601. https://doi.org/10.1039/d5ma00942a.
- 44.
Zheng, B.; Zhou, H.; Zhao, G.; et al. Bioinspired electrically conductive hydrogels: Rational engineering for next-generation flexible mechanosensors. Mater. Sci. Eng. R Rep. 2025, 166, 101080. https://doi.org/10.1016/j.mser.2025.101080.
- 45.
Hu, L.; Chee, P.L.; Sugiarto, S.; et al. Hydrogel-based flexible electronics. Adv. Mater. 2023, 35, 2205326. https://doi.org/10.1002/adma.202205326.
- 46.
Wang, J.; Chen, Y.; Tu, S.; et al. Recent advances in flexible iontronic pressure sensors: Materials, microstructure designs, applications, and opportunities. J. Mater. Chem. C 2024, 12, 14202–14221. https://doi.org/10.1039/d4tc03226h.
- 47.
He, Y.; Cheng, Y.; Yang, C.; et al. Creep-free polyelectrolyte elastomer for drift-free iontronic sensing. Nat. Mater. 2024, 23, 1107–1114. https://doi.org/10.1038/s41563-024-01848-6.
- 48.
Song, J.; Yang, R.; Shi, J.; et al. Polyelectrolyte-based wireless and drift-free iontronic sensors for orthodontic sensing. Sci. Adv. 2025, 11, eadu6086. https://doi.org/10.1126/sciadv.adu6086.
- 49.
Fan, X.; Chen, Z.; Sun, H.; et al. Polyelectrolyte-based conductive hydrogels: From theory to applications. Soft Sci. 2022, 2, 10. https://doi.org/10.20517/ss.2022.09.
- 50.
Pan, X.; Wang, Q.; Benetti, D.; et al. Polyelectrolyte hydrogel: A versatile platform for mechanical-electric conversion and self-powered sensing. Nano Energy 2022, 103, 107718. https://doi.org/10.1016/j.nanoen.2022.107718.
- 51.
Wang, Z.; Si, Y.; Zhao, C.; et al. Flexible and washable poly (ionic liquid) nanofibrous membrane with moisture proof pressure sensing for real-life wearable electronics. ACS Appl. Mater. Interfaces 2019, 11, 27200–27209. https://doi.org/10.1021/acsami.9b07786.
- 52.
Chen, S.; Zhang, M.; Zou, P.; et al. Historical development and novel concepts on electrolytes for aqueous rechargeable batteries. Energy Environ. Sci. 2022, 15, 1805–1839. https://doi.org/10.1039/d2ee00004k.
- 53.
Liu, J.; Liu, X.; Chen, J.; et al. Drying-enhanced polyvinyl alcohol-polyacrylic acid double-network hydrogel and its application in flexible strain sensors. Chem. Eng. Sci. 2022, 264, 118120. https://doi.org/10.1016/j.ces.2022.118120.
- 54.
Liu, Z.; Wang, Y.; Ren, Y.; et al. Poly (ionic liquid) hydrogel-based anti-freezing ionic skin for a soft robotic gripper. Mater. Horiz. 2020, 7, 919–927. https://doi.org/10.1039/c9mh01688k.
- 55.
Ji, R.; Yan, S.; Zhu, Z.; et al. Ureido-Ionic Liquid Mediated Conductive Hydrogel: Superior Integrated Properties for Advanced Biosensing Applications. Adv. Sci. 2024, 11, 2401869. https://doi.org/10.1002/advs.202401869.
- 56.
Fan, X.; Liu, S.; Jia, Z.; et al. Ionogels: Recent advances in design, material properties and emerging biomedical applications. Chem. Soc. Rev. 2023, 52, 2497–2527. https://doi.org/10.1039/d2cs00652a.
- 57.
Nie, B.; Li, R.; Cao, J.; et al. Flexible transparent iontronic film for interfacial capacitive pressure sensing. Adv. Mater. 2015, 27, 6055–6062. https://doi.org/10.1002/adma.201502556.
- 58.
Wang, X.; Guo, C.; Su, Z.; et al. Flexible iontronic pressure sensing technology: Advanced structural ionic layer. Iontronics 2026, 2, 1. https://dx.doi.org/10.20517/iontronics.2026.01.
- 59.
Zhang, Y.-A.; Ma, K.; Chen, K.-Z.; et al. Flexible wearable ionogels: Classification, fabrication, properties and applications. Sens. Actuators A Phys. 2024, 372, 115325. https://doi.org/10.1016/j.sna.2024.115325.
- 60.
Wen, J.; Zhou, L.; Ye, T. Polymer ionogels and their application in flexible ionic devices. SmartMat 2024, 5, e1253. https://doi.org/10.1002/smm2.1253.
- 61.
Xiong, Y.; Han, J.; Wang, Y.; et al. Emerging iontronic sensing: Materials, mechanisms, and applications. Research 2022, 2022, 9867378. https://doi.org/10.34133/2022/9867378.
- 62.
Hu, X.; Zhao, Y.; Pu, L.; et al. Stretchable anti-freeze deep eutectic solvent (DES) gels for low-temperature wearable soft sensors. New J. Chem. 2024, 48, 11003–11013. https://doi.org/10.1039/d4nj00452c.
- 63.
Htwe, Y.Z.N.; Pawłowska, S.; Jaafar, M. Emerging Strategies for the Fabrication of Conductive Hydrogels from Conductive Polymers and Their Composites for Wearable Sensors, Energy Storage, and Biosensor Applications: Methods, Mechanisms, and Future Perspectives. Adv. Mater. Technol. 2025, 11, e01845. https://doi.org/10.1002/admt.202501845.
- 64.
Zhu, T.; Ni, Y.; Biesold, G.M.; et al. Recent advances in conductive hydrogels: Classifications, properties, and applications. Chem. Soc. Rev. 2023, 52, 473–509. https://doi.org/10.1039/d2cs00173j.
- 65.
Ren, J.; Wang, Y.; Liu, Z.; et al. Balancing stretchability and conductivity: Carbon nanotube layer-enhanced non-ionic conductive hydrogels with a sandwich structure. Chem. Eng. J. 2024, 500, 156641. https://doi.org/10.1016/j.cej.2024.156641.
- 66.
Hauck, M.; Saure, L.M.; Zeller-Plumhoff, B.; et al. Overcoming water diffusion limitations in hydrogels via microtubular graphene networks for soft actuators. Adv. Mater. 2023, 35, 2302816. https://doi.org/10.1002/adma.202370292.
- 67.
Lin, Y.; Wu, A.; Zhang, Y.; et al. Recent progress of nanomaterials-based composite hydrogel sensors for human–machine interactions. Discov. Nano 2025, 20, 1–30. https://doi.org/10.1186/s11671-025-04240-8.
- 68.
Li, W.; Liu, J.; Wei, J.; et al. Recent progress of conductive hydrogel fibers for flexible electronics: Fabrications, applications, and perspectives. Adv. Funct. Mater. 2023, 33, 2213485. https://doi.org/10.1002/adfm.202213485.
- 69.
Peng, Q.; Chen, J.; Wang, T.; et al. Recent advances in designing conductive hydrogels for flexible electronics. InfoMat 2020, 2, 843–865. https://doi.org/10.1002/inf2.12113.
- 70.
Guan, Z.; Jiang, Y.; Zhou, Y.; et al. Liquid metal-based electrodes for flexible electronics: Z. Guan et al. Rare Met. 2025, 44, 6897–6923. https://doi.org/10.1007/s12598-025-03466-w.
- 71.
Li, Y.; Zhao, Y.; Yang, R.; et al. Flexible electrodes with enhancement of electronic-ionic conductivity for electrophysiological signal monitoring. Wearable Electron. 2024, 1, 228–235. https://doi.org/10.1016/j.wees.2024.09.005.
- 72.
Liu, K.; Duan, T.; Zhang, F.; et al. Flexible electrode materials for emerging electronics: Materials, fabrication and applications. J. Mater. Chem. A 2024, 12, 20606–20637. https://doi.org/10.1039/d4ta01960a.
- 73.
Yan, N.; Sujanani, R.; Kamcev, J.; et al. Salt and ion transport in a series of crosslinked AMPS/PEGDA hydrogel membranes. J. Membr. Sci. 2022, 653, 120549. https://doi.org/10.1016/j.memsci.2022.120549.
- 74.
Dai, Q.; Liao, W.; Liu, J.; et al. Microfluidic bubble-templating 3D printing of ordered macroporous hydrogels. Compos. Part B Eng. 2024, 284, 111725. https://doi.org/10.1016/j.compositesb.2024.111725.
- 75.
Yang, Z.; Wang, J.; Wan, X.; et al. Microbubble-based fabrication of resilient porous ionogels for high-sensitivity pressure sensors. Microsyst. Nanoeng. 2024, 10, 177. https://doi.org/10.1038/s41378-024-00780-8.
- 76.
Adelnia, H.; Ensandoost, R.; Moonshi, S.S.; et al. Freeze/thawed polyvinyl alcohol hydrogels: Present, past and future. Eur. Polym. J. 2022, 164, 110974. https://doi.org/10.1016/j.eurpolymj.2021.110974.
- 77.
Nordness, O.; Moon, J.D.; Marioni, N.; et al. Probing water and ion diffusion in functional hydrogel membranes by PFG-NMR. Macromolecules 2023, 56, 4669–4680. https://doi.org/10.1021/acs.macromol.3c00306.
- 78.
Yu, Y.Y.; Xiang, H.P.; Fan, L.F.; et al. Shape Memory Elastomers: A Review of Molecular Structures, Stimulus Mechanisms, and Emerging Applications. Polym. Sci. Technol. 2025, 1, 271–298. https://doi.org/10.1021/polymscitech.4c00035.
- 79.
Fong, K.D.; Self, J.; McCloskey, B.D.; et al. Ion correlations and their impact on transport in polymer-based electrolytes. Macromolecules 2021, 54, 2575–2591. https://doi.org/10.1021/acs.macromol.0c02545.
- 80.
Shao, Y.; Gudla, H.; Mindemark, J.; et al. Ion transport in polymer electrolytes: Building new bridges between experiment and molecular simulation. Acc. Chem. Res. 2024, 57, 1123–1134. https://doi.org/10.1021/acs.accounts.3c00791.
- 81.
Kiyohara, K.; Tamura, M. Transport coefficients of gel electrolytes: A molecular dynamics simulation study. J. Chem. Phys. 2022, 156, 084905. https://doi.org/10.1063/5.0081118.
- 82.
Lu, M.; Lian, W.Z.; Xiao, Z.; et al. Interplay of chain dynamics and ion transport on mechanical behavior and conductivity in ionogels. Soft Matter 2025, 21, 435–447. https://doi.org/10.1039/d4sm01251h.
- 83.
Klika, V.; Gaffney, E.A. Upscaling the Poisson–Nernst–Planck equations for ion transport in weakly heterogeneous charged porous media. Appl. Math. Lett. 2023, 137, 108482. https://doi.org/10.1016/j.aml.2022.108482.
- 84.
Appel, E.A.; Tibbitt, M.W.; Greer, J.M.; et al. Exploiting electrostatic interactions in polymer–nanoparticle hydrogels. ACS Macro Lett. 2015, 4, 848–852. https://doi.org/10.1021/acsmacrolett.5b00416.
- 85.
Appel, E.A.; Tibbitt, M.W.; Webber, M.J.; et al. Self-assembled hydrogels utilizing polymer–nanoparticle interactions. Nat. Commun. 2015, 6, 6295. https://doi.org/10.1038/ncomms7295.
- 86.
Ouyang, Y.; Wang, Z.L.; Wei, D. Ionic rectification via electrical double layer modulation at hydrogel interfaces. RSC Appl. Interfaces 2025, 2, 873–896. https://doi.org/10.1039/d5lf00098j.
- 87.
Liu, Z.; Zheng, S.; Li, Z.; et al. Multiscale modeling of hydrogels. In The Mechanics of Hydrogels, Elsevier: Amsterdam, The Netherlands, 2022; pp. 187–222. http://dx.doi.org/10.1016/b978-0-08-102862-9.00012-9.
- 88.
Shen, K.-H.; Fan, M.; Hall, L.M. Molecular dynamics simulations of ion-containing polymers using generic coarse-grained models. Macromolecules 2021, 54, 2031–2052. https://doi.org/10.1021/acs.macromol.0c02557.
- 89.
Singhal, A.; Schneible, J.D.; Lilova, R.L.; et al. A multiscale coarse-grained model to predict the molecular architecture and drug transport properties of modified chitosan hydrogels. Soft Matter 2020, 16, 10591–10610. https://doi.org/10.1039/d0sm01243b.
- 90.
Wang, M. Ionogels: From Properties and Synthesis to Toughening, Patterning, and Applications. Chem. Rev. 2025, 125, 11815–11839. https://doi.org/10.1021/acs.chemrev.5c00370.
- 91.
Ouyang, Y.; Li, X.; Du, Y.; et al. Mechano-Driven Neuromimetic Logic Gates Established by Geometrically Asymmetric Hydrogel Iontronics. Small 2025, 21, 2409998. https://doi.org/10.1002/smll.202409998.
- 92.
Daso, R.E.; Posey, R.; Garza, H.; et al. Standardized Electrochemical Characterization of Conductive Hydrogels. Adv. Funct. Mater. 2025, 35, e08859. https://doi.org/10.1002/adfm.71976.
- 93.
Sun, W.; Xu, Z.; Qiao, C.; et al. Antifreezing proton zwitterionic hydrogel electrolyte via ionic hopping and grotthuss transport mechanism toward solid supercapacitor working at −50° C. Adv. Sci. 2022, 9, 2201679. https://doi.org/10.1002/advs.202201679.
- 94.
Guo, Y.; Bae, J.; Fang, Z.; et al. Hydrogels and hydrogel-derived materials for energy and water sustainability. Chem. Rev. 2020, 120, 7642–7707. https://doi.org/10.1021/acs.chemrev.0c00345.
- 95.
Zhang, S.; Song, C.; Ji, Z.; et al. Synergistic vehicular-grotthuss conduction in double-network hydrogel electrolytes for zinc dendrite suppression in zinc-air batteries. J. Colloid Interface Sci. 2026, 703, 139064. https://doi.org/10.1016/j.jcis.2025.139064.
- 96.
Colla, T.; Telles, I.M.; Arfan, M.; et al. Spiers Memorial Lecture: Towards understanding of iontronic systems: Electroosmotic flow of monovalent and divalent electrolyte through charged cylindrical nanopores. Faraday Discuss. 2023, 246, 11–46. https://doi.org/10.1039/d3fd00062a.
- 97.
Schuszter, G.; Gehér-Herczegh, T.; Szűcs, Á.; et al. Determination of the diffusion coefficient of hydrogen ion in hydrogels. Phys. Chem. Chem. Phys. 2017, 19, 12136–12143. https://doi.org/10.1039/c7cp00986k.
- 98.
Barbero, G.; Lelidis, I. Analysis of Warburg’s impedance and its equivalent electric circuits. Phys. Chem. Chem. Phys. 2017, 19, 24934–24944. https://doi.org/10.1039/c7cp04032f.
- 99.
Henrique, F.; Gupta, A. Parallel Warburg Elements Describe Ionic Transport in Nanopores. PRX Energy 2025, 4, 023009. https://doi.org/10.1103/prxenergy.4.023009.
- 100.
Xu, Z.; Yue, P.; Feng, J.J. Hystereses in flow-induced compression of a poroelastic hydrogel. Soft Matter 2024, 20, 6940–6951. https://doi.org/10.1039/d4sm00678j.
- 101.
Biutty, M.N.; Kim, H.; Handayani, P.L.; et al. Self-powered smart pressure sensors by stimuli-responsive ion transport within layered hydrogels. Chem. Eng. J. 2024, 495, 153565. https://doi.org/10.1016/j.cej.2024.153565.
- 102.
Zhang, W.; Zhang, X.; Dutta, A.; et al. Hydrogel-based sweat chloride sensor with high sensitivity and low hysteresis. Biosens. Bioelectron. 2025, 288, 117805. https://doi.org/10.1016/j.bios.2025.117805.
- 103.
Liu, C.; Liu, Z. Micro-Gas Flow Sensor Utilizing Surface Network Density Regulation for Humidity-Modulated Ion Transport. Gels 2025, 11, 570. https://doi.org/10.3390/gels11080570.
- 104.
Wu, M.; Liu, Z.; Gao, Y. Design and Fabrication of Microelectrodes for Dielectrophoresis and Electroosmosis in Microsystems for Bio-Applications. Micromachines 2025, 16, 190. https://doi.org/10.3390/mi16020190.
- 105.
Wang, G.; Kato, K.; Aoki, I.; et al. Transdermal drug delivery using a porous microneedle device driven by a hydrogel electroosmotic pump. J. Mater. Chem. B 2024, 12, 1490–1494. https://doi.org/10.1039/d3tb02208k.
- 106.
Kusama, S.; Sato, K.; Matsui, Y.; et al. Transdermal electroosmotic flow generated by a porous microneedle array patch. Nat. Commun. 2021, 12, 658. https://doi.org/10.1038/s41467-021-20948-4.
- 107.
Zhu, K.; Luo, J.; Zhang, D.; et al. Molecular engineering enables hydrogel electrolyte with ionic hopping migration and self-healability toward dendrite-free zinc-metal anodes. Adv. Mater. 2024, 36, 2311082. https://doi.org/10.1002/adma.202311082.
- 108.
Popov, I.; Zhu, Z.; Singh, H.; et al. Mechanisms of proton transport in aqueous acid solutions. Cell Rep. Phys. Sci. 2024, 5, 102294. https://doi.org/10.1016/j.xcrp.2024.102294.
- 109.
Li, Z.; Yun, H.; Yan, Y.; et al. Boosting Electronic Charge Transport in Conductive Hydrogels via Rapid Ion-Electron Transduction. Angew. Chem. Int. Ed. 2025, 64, e202506560. https://doi.org/10.1002/ange.202506560.
- 110.
Popov, I.; Zhu, Z.; Young-Gonzales, A.R.; et al. Search for a Grotthuss mechanism through the observation of proton transfer. Commun. Chem. 2023, 6, 77. https://doi.org/10.1038/s42004-023-00878-6.
- 111.
Ichikawa, T.; Yamada, T.; Aoki, N.; et al. Surface proton hopping conduction mechanism dominant polymer electrolytes created by self-assembly of bicontinuous cubic liquid crystals. Chem. Sci. 2024, 15, 7034–7040. https://doi.org/10.1039/d4sc01211a.
- 112.
Wang, Z.D.; Yang, Y.Q.; Chen, J.H.; et al. Proton Superhighways Enabled by Hofmeister-Electrostatic Synergy in All-Inorganic Polyoxometalate Hydrogels for Electronics. Adv. Mater. 2025, 38, e15892. https://doi.org/10.1002/adma.202515892.
- 113.
Yang, H.; Sun, X.; Li, X.; et al. Janus Grotthuss-Vehicle Mechanism Enhances Fast OH− Transport for Ultralong Lifetime Flexible Zinc–Air Battery. Adv. Funct. Mater. 2024, 34, 2409695. https://doi.org/10.1002/adfm.202409695.
- 114.
Chen, K.; Hu, H.; Song, I.; et al. Organic optoelectronic synapse based on photon-modulated electrochemical doping. Nat. Photonics 2023, 17, 629–637. https://doi.org/10.1038/s41566-023-01232-x.
- 115.
Lazanas, A.; Simón, B.P. A guide to recognizing your electrochemical impedance spectra: Revisions of the randles circuit in (bio) sensing. Sensors 2025, 25, 6260. https://doi.org/10.3390/s25196260.
- 116.
Plank, C.; Rüther, T.; Jahn, L.; et al. A review on the distribution of relaxation times analysis: A powerful tool for process identification of electrochemical systems. J. Power Sources 2024, 594, 233845. https://doi.org/10.1016/j.jpowsour.2023.233845.
- 117.
Huang, Y.; Hu, S.; Li, Y.; et al. Programmable high-sensitivity iontronic pressure sensors support broad human-interactive perception and identification. npj Flex. Electron. 2025, 9, 41. https://doi.org/10.1038/s41528-025-00420-9.
- 118.
Greco, A.; Imoto, S.; Backus, E.H.; et al. Ultrafast aqueous electric double layer dynamics. Science 2025, 388, 405–410. https://doi.org/10.1126/science.adu5781.
- 119.
Yan, H.; Qi, R.; Liu, Z.; et al. Unlocking the potential of hydrogel-electrode electrical double layer for high-performance moisture-enabled electric generators. Device 2025, 3, 100568. https://doi.org/10.1016/j.device.2024.100568.
- 120.
Gateman, S.M.; Gharbi, O.; De Melo, H.G.; et al. On the use of a constant phase element (CPE) in electrochemistry. Curr. Opin. Electrochem. 2022, 36, 101133. https://doi.org/10.1016/j.coelec.2022.101133.
- 121.
Gamry Instruments. Two-, Three-, and Four-Electrode Experiments; Gamry Instruments: Warminster, PA, USA, 2021.
- 122.
Jjagwe, J.; Olupot, P.W.; Kulabako, R.; et al. Electrochemical sensors modified with iron oxide nanoparticles/nanocomposites for voltammetric detection of Pb (II) in water: A review. Heliyon 2024, 10, e29743. https://doi.org/10.1016/j.heliyon.2024.e29743.
- 123.
Lazanas, A.C.; Prodromidis, M.I. Electrochemical impedance spectroscopy—A tutorial. ACS Meas. Sci. Au 2023, 3, 162–193. https://doi.org/10.1021/acsmeasuresciau.2c00070.
- 124.
Jiang, Y.; Han, Y.; Gao, Z.; et al. Frequency-dependent electrochemical breakdown of hydrogel ionotronics. Extrem. Mech. Lett. 2024, 71, 102210. https://doi.org/10.1016/j.eml.2024.102210.
- 125.
Li, S.; Gao, L.; Liu, C.; et al. Biomimetic Neuromorphic Sensory System via Electrolyte Gated Transistors. Sensors 2024, 24, 4915. https://doi.org/10.3390/s24154915.
- 126.
Huang, Y.J.; Di, J.K.; Li, Y.; et al. Polyvinyl alcohol electrolyte-gated oxide transistors with tetanization activities for neuromorphic computing. J. Mater. Chem. C 2024, 12, 5166–5174. https://doi.org/10.1039/d4tc00526k.
- 127.
Owyeung, R.E.; Zeng, W.; Panzer, M.J.; et al. Free form three dimensional integrated circuits and wearables on a thread using organic eutectogel gated electrochemical transistors. arXiv 2023, arXiv:2303.02447.
- 128.
Wu, B.; Peng, Y.; Gao, L.; et al. Eutectogel-gated ultra-flexible organic electrochemical transistors for humidity sensing and neuromorphic computing. Sens. Actuators B Chem. 2025, 450, 139268. https://doi.org/10.1016/j.snb.2025.139268.
- 129.
Tian, Z.; Zhao, Z.; Yan, F. Organic electrochemical transistor in wearable bioelectronics: Profiles, applications, and integration. Wearable Electron. 2024, 1, 1–25. https://doi.org/10.1016/j.wees.2024.03.002.
- 130.
Tang, L.; Zheng, X.; Sun, M.; et al. Photopatternable gel electrolytes for stretchable solid-state organic electrochemical transistors. Sci. China Mater. 2025, 68, 3212–3218. https://doi.org/10.1007/s40843-025-3429-x.
- 131.
Tian, X.; Bai, J.; Liu, D.; et al. A fully-integrated flexible in-sensor computing circuit based on gel-gated organic electrochemical transistors. npj Flex. Electron. 2025, 9, 90. https://doi.org/10.1038/s41528-025-00472-x.
- 132.
Zhong, Y.; Lopez-Larrea, N.; Alvarez-Tirado, M.; et al. Eutectogels as a semisolid electrolyte for organic electrochemical transistors. Chem. Mater. 2024, 36, 1841–1854. https://doi.org/10.1021/acs.chemmater.3c02385.
- 133.
Li, Y.; Cheng, Q.; Deng, Z.; et al. Recent progress of anti-freezing, anti-drying, and anti-swelling conductive hydrogels and their applications. Polymers 2024, 16, 971. https://doi.org/10.3390/polym16070971.
- 134.
Fan, Z.; Ji, D.; Kim, J. Congelation-and dehydration-tolerant, mechanically robust hydrogel electrolyte for durable iontronic sensors operating in open air and freezing temperatures over wide strain and pressure ranges. Chem. Eng. J. 2024, 499, 156677. https://doi.org/10.1016/j.cej.2024.156677.
- 135.
Lei, T.; Wang, Y.; Feng, Y.; et al. PNIPAAm-based temperature responsive ionic conductive hydrogels for flexible strain and temperature sensing. J. Colloid Interface Sci. 2025, 678, 726–741. https://doi.org/10.1016/j.jcis.2024.09.131.
- 136.
Zhen, E.; Chen, Y.; Huang, J. Double-layer capacitance peaks: Origins, ion dependence, and temperature effects. J. Chem. Phys. 2025, 162, 144702. https://doi.org/10.1063/5.0251548.
- 137.
De Bortoli, A.; Negro, E.; Di Noto, V. Generalized modified Poisson–Nernst–Planck model for electrical double layer with steric, correlation and thermal effects applied to fuel cells. Electrochim. Acta 2025, 525, 146070. https://doi.org/10.1016/j.electacta.2025.146070.
- 138.
Chen, K.; Lai, W.; Xiao, W.; et al. Low-temperature adaptive dual-network mxene nanocomposite hydrogel as flexible wearable strain sensors. Micromachines 2023, 14, 1563. https://doi.org/10.3390/mi14081563.
- 139.
Song, Z.; Han, R.; Yu, K.; et al. Antifouling strategies for electrochemical sensing in complex biological media. Microchim. Acta 2024, 191, 138. https://doi.org/10.1007/s00604-024-06218-2.
- 140.
Wang, Z.; Liu, X.; Zhu, M.; et al. Recent advances in zwitterionic hydrogels: Structure, applications and challenges. J. Mater. Chem. A 2025, 13, 13693–13705. https://doi.org/10.1039/d5ta01111f.
- 141.
Han, X.; Su, J.; Li, J.; et al. Bio-Inspired and Protein-Based Elastomeric Materials. Polym. Sci. Technol. 2025, 2, 6–21. https://doi.org/10.1021/polymscitech.5c00054.
- 142.
Zhang, T.; Liang, X.; Wang, L.; et al. An ionic liquid assisted hydrogel functionalized silica stationary phase for mixed-mode liquid chromatography. Chin. Chem. Lett. 2025, 36, 109889. https://doi.org/10.1016/j.cclet.2024.109889.
- 143.
Liang, C.; Liu, J.; Zhang, H.; et al. Mitigating lipid biofouling in wearable sweat sensors: A study on conductive MOF-based electrodes with tuned hydrophilicity. Chem. Eng. J. 2025, 518, 164477. https://doi.org/10.1016/j.cej.2025.164477.
- 144.
Wareham-Mathiassen, S.; Jolly, P.; Radha Shanmugam, N.; et al. An Antimicrobial and Antifibrotic Coating for Implantable Biosensors. Biosensors 2025, 15, 171. https://doi.org/10.3390/bios15030171.
- 145.
Kang, M.; Park, J.; Kim, S.A.; et al. Modulus-tunable multifunctional hydrogel ink with nanofillers for 3D-Printed soft electronics. Biosens. Bioelectron. 2024, 255, 116257. https://doi.org/10.1016/j.bios.2024.116257.
- 146.
Pan, M.; Shui, T.; Zhao, Z.; et al. Engineered Janus hydrogels: Biomimetic surface engineering and biomedical applications. Natl. Sci. Rev. 2024, 11, nwae316. https://doi.org/10.1093/nsr/nwae316.
- 147.
Xu, Y.; Sun, K.; Huang, L.; et al. Magneto-induced Janus adhesive-tough hydrogels for wearable human motion sensing and enhanced low-grade heat harvesting. ACS Appl. Mater. Interfaces 2024, 16, 10556–10564. https://doi.org/10.1021/acsami.3c19373.
- 148.
Zhang, C.W.; Chen, C.; Duan, S.; et al. Hydrogel-based soft bioelectronics for personalized healthcare. Med-X 2024, 2, 20. https://doi.org/10.1007/s44258-024-00036-0.
- 149.
Sun, Z.; Cao, X.; Wang, S.; et al. Designing polydopamine embedded hydrogel with high-stable adhesion and long-term stability at high temperature. Polymer 2025, 317, 127914. https://doi.org/10.1016/j.polymer.2024.127914.
- 150.
Lian, Z.; Wang, L.; Jiang, Y.; et al. 3D-printed octopus-inspired PAM/CS hydrogels with excellent adhesion for high-performance ECG sensors. Chem. Eng. J. 2025, 508, 161043. https://doi.org/10.1016/j.cej.2025.161043.
- 151.
Li, W.; Liu, H.; Mi, Y.; et al. Robust and conductive hydrogel based on mussel adhesive chemistry for remote monitoring of body signals. Friction 2022, 10, 80–93. https://doi.org/10.1007/s40544-020-0416-x.
- 152.
Shin, Y.; Lee, H.S.; Kim, J.-U.; et al. Functional-hydrogel-based electronic-skin patch for accelerated healing and monitoring of skin wounds. Biomaterials 2025, 314, 122802. https://doi.org/10.1016/j.biomaterials.2024.122802.
- 153.
Ren, N.; Zheng, G.; Cui, M.; et al. Integrally formed Janus adhesive conductive hydrogel with excellent interfacial stability and fatigue resistance for robust flexible sensing. Cell Rep. Phys. Sci. 2025, 6, 102916. https://doi.org/10.1016/j.xcrp.2025.102916.
- 154.
Yao, B.; Wu, S.; Wang, R.; et al. Hydrogel ionotronics with ultra-low impedance and high signal fidelity across broad frequency and temperature ranges. Adv. Funct. Mater. 2022, 32, 2109506. https://doi.org/10.1002/adfm.202109506.
- 155.
Jayakumar, K.; Lielpetere, A.; Domingo-Lopez, D.A.; et al. Tethering zwitterionic polymer coatings to mediated glucose biosensor enzyme electrodes can decrease sensor foreign body response yet retain sensor sensitivity to glucose. Biosens. Bioelectron. 2023, 219, 114815. https://doi.org/10.1016/j.bios.2022.114815.
- 156.
Zhao, B.; Li, Z.; Zheng, L.; et al. Recent progress in the biomedical application of PEDOT: PSS hydrogels. Chin. Chem. Lett. 2024, 35, 109810. https://doi.org/10.1016/j.cclet.2024.109810.
- 157.
Conti, S. Robust pure PEDOT: PSS hydrogels for bioelectronic interfaces. Nat. Rev. Electr. Eng. 2024, 1, 356. https://doi.org/10.1038/s44287-024-00066-1.
- 158.
Wen, J.; Huang, S.; Hu, Q.; et al. Recent advances in zwitterionic polymers-based non-fouling coating strategies for biomedical applications. Mater. Today Chem. 2024, 40, 102232. https://doi.org/10.1016/j.mtchem.2024.102232.
- 159.
Yadav, S.; Rani, N.; Saini, K.; et al. Biological evaluation of medical devices. In Medical Devices in Modern Healthcare, Elsevier: Amsterdam, The Netherlands, 2026; pp. 475–498. https://doi.org/10.1016/b978-0-443-32828-2.00010-7.
- 160.
Stewart, E.M.; Narayan, S.; Anand, L. An electro-chemo-mechanical theory for hydrogel ionotronics: Application to modeling a capacitive strain sensor and a dynamic large strain actuator. J. Mech. Phys. Solids 2023, 173, 105196. https://doi.org/10.1016/j.jmps.2022.105196.
- 161.
Castillo-Acuna, R.; Kochmann, D.M. Multiphysics modeling of hydrogels with a maximum-entropy-based meshless framework. Comput. Methods Appl. Mech. Eng. 2025, 446, 118293. https://doi.org/10.1016/j.cma.2025.118293.
- 162.
Xu, Z.; Yue, P.; Feng, J.J. A theory of hydrogel mechanics that couples swelling and external flow. Soft Matter 2024, 20, 5389–5406. https://doi.org/10.1039/d4sm00424h.
- 163.
Lyu, Z.; Sciazko, A.; Shikazono, N.; et al. Enhanced temporal prediction of electrochemical impedance spectroscopy using long short-term memory neural networks. Electrochim. Acta 2024, 508, 145227. https://doi.org/10.1016/j.electacta.2024.145227.
- 164.
Wang, Z.; Wang, Y.; Py, B.; et al. DRTtools: Freely Accessible Distribution of Relaxation Times Analysis for Electrochemical Impedance Spectroscopy. ACS Electrochem. 2025, 1, 2680–2689. https://doi.org/10.1021/acselectrochem.5c00334.
- 165.
Zhu, S.; Jia, G.; Sui, S.; et al. Integrating large language and multimodal models with machine learning for equivalent circuit analysis of electrochemical impedance spectroscopy. J. Mater. Sci. 2025, 60, 22189–22202. https://doi.org/10.1007/s10853-025-11692-x.
- 166.
Ansari, M.; Darvishi, A. A review of the current state of natural biomaterials in wound healing applications. Front. Bioeng. Biotechnol. 2024, 12, 1309541. https://doi.org/10.3389/fbioe.2024.1309541.
- 167.
Yuan, W.; Qu, X.; Lu, Y.; et al. MXene-composited highly stretchable, sensitive and durable hydrogel for flexible strain sensors. Chin. Chem. Lett. 2021, 32, 2021–2026. https://doi.org/10.1016/j.cclet.2020.12.003.
- 168.
Isik, M.; Lonjaret, T.; Sardon, H.; et al. Cholinium-based ion gels as solid electrolytes for long-term cutaneous electrophysiology. J. Mater. Chem. C 2015, 3, 8942–8948. https://doi.org/10.1039/c5tc01888a.
- 169.
De la Cruz, L.G.; Abt, T.; León, N.; et al. Ice-template crosslinked PVA aerogels modified with tannic acid and sodium alginate. Gels 2022, 8, 419. https://doi.org/10.3390/gels8070419.
- 170.
Li, M.; Li, W.; Guan, Q.; et al. Sweat-resistant bioelectronic skin sensor. Device 2023, 1, 100006. https://doi.org/10.1016/j.device.2023.100006.
- 171.
Liu, L.; Feng, X.; Du, J.; et al. Sweat-activated conductive hydrogel nanomesh for breathable, long-term electrophysiological monitoring and human-centric interfaces. Matter 2026, 9, 102428. https://doi.org/10.1016/j.matt.2025.102428.
- 172.
Zhu, Y.; Chen, B.; Liu, Y.; et al. Recent advances in conductive hydrogels for electronic skin and healthcare monitoring. Biosensors 2025, 15, 463. https://doi.org/10.3390/bios15070463.
- 173.
Tian, Y.; Yang, Y.; Tang, H.; et al. An implantable hydrogel-based phononic crystal for continuous and wireless monitoring of internal tissue strains. Nat. Biomed. Eng. 2025, 9, 1335–1348. https://doi.org/10.1038/s41551-025-01374-z.
- 174.
Ding, M.; Xie, P.; Wang, J.; et al. Biomimetic microstructure design for ultrasensitive piezoionic mechanoreceptors in multimodal object recognition. Nat. Commun. 2025, 16, 8129. https://doi.org/10.1038/s41467-025-63115-9.
- 175.
Ni, Y.; Zang, X.; Yang, Y.; et al. Environmental stability stretchable organic hydrogel humidity sensor for respiratory monitoring with ultrahigh sensitivity. Adv. Funct. Mater. 2024, 34, 2402853. https://doi.org/10.1002/adfm.202402853.
- 176.
Oh, B.; Lim, Y.-S.; Ko, K.W.; et al. Ultra-soft and highly stretchable tissue-adhesive hydrogel based multifunctional implantable sensor for monitoring of overactive bladder. Biosens. Bioelectron. 2023, 225, 115060. https://doi.org/10.1016/j.bios.2023.115060.
- 177.
Wang, Y.; Ge, M.; Wang, R.; et al. Injectable ultrasonic metagels for intracranial monitoring. npj Biosensing 2025, 2, 38. https://doi.org/10.1038/s44328-025-00058-7.
- 178.
Huang, X.; Yang, N.; Sun, S.; et al. Recent progress of hydrogel-based bioelectronics for mechanophysiological signal sensing. Mater. Sci. Eng. R Rep. 2025, 162, 100888. https://doi.org/10.1016/j.mser.2024.100888.
- 179.
Sagdic, K.; Fernandez-Lavado, E.; Mariello, M.; et al. Hydrogels and conductive hydrogels for implantable bioelectronics. MRS Bull. 2023, 48, 495–505. https://doi.org/10.1557/s43577-023-00536-1.
- 180.
Amoli, V.; Kim, J.S.; Jee, E.; et al. A bioinspired hydrogen bond-triggered ultrasensitive ionic mechanoreceptor skin. Nat. Commun. 2019, 10, 4019. https://doi.org/10.1038/s41467-019-11973-5.
- 181.
Zheng, Y.; Wang, J.; Cui, T.; et al. Transparent ionogel balancing rigidity and flexibility with prolonged stability for ultra-high sensitivity temperature sensing. Chem. Eng. J. 2024, 494, 152695. https://doi.org/10.1016/j.cej.2024.152695.
- 182.
Zheng, Y.; Liu, H.; Wang, J.; et al. Unlocking intrinsic conductive dynamics of ionogel microneedle arrays as wearable electronics for intelligent fire safety. Adv. Fiber Mater. 2024, 6, 195–213. https://doi.org/10.1007/s42765-023-00344-x.
- 183.
Huang, W.; Ding, Q.; Wang, H.; et al. Design of stretchable and self-powered sensing device for portable and remote trace biomarkers detection. Nat. Commun. 2023, 14, 5221. https://doi.org/10.1038/s41467-023-40953-z.
- 184.
Yang, M.; Hu, Y.; Wang, X.; et al. Chaotropic Effect-Boosted Thermogalvanic Ionogel Thermocells for All-Weather Power Generation. Adv. Mater. 2024, 36, 2312249. https://doi.org/10.1002/adma.202312249.
- 185.
Li, M.; Guan, Q.; Li, C.; et al. Self-powered hydrogel sensors. Device 2023, 1, 100007. https://doi.org/10.1016/j.device.2023.100007.
- 186.
Ou, K.; Wang, M.; Meng, C.; et al. Enhanced mechanical strength and stretchable ionic conductive hydrogel with double-network structure for wearable strain sensing and energy harvesting. Compos. Sci. Technol. 2024, 255, 110732. https://doi.org/10.1016/j.compscitech.2024.110732.
- 187.
Zhang, Y.; Wang, H.; Khan, S.A.; et al. Deep-learning-assisted thermogalvanic hydrogel fiber sensor for self-powered in-nostril respiratory monitoring. J. Colloid Interface Sci. 2025, 678, 143–149. https://doi.org/10.1016/j.jcis.2024.09.132.
- 188.
Wang, X.; Yuan, G.; Zhou, H.; et al. Composite laminar membranes for electricity generation from water evaporation. Nano Res. 2024, 17, 307–311. https://doi.org/10.1007/s12274-023-5906-5.
- 189.
Chen, J.; Zhang, X.; Cheng, M.; et al. A self-sustained moist-electric generator with enhanced energy density and longevity through a bilayer approach. Mater. Horiz. 2025, 12, 2309–2318. https://doi.org/10.1039/d4mh01642d.
- 190.
Chang, K.; Zhang, C.; Liu, T. A comprehensive review on fabrication and structural design of polymer composites for wearable pressure sensors. Polym. Sci. Technol. 2025, 1, 3–24. https://doi.org/10.1021/polymscitech.4c00047.

This work is licensed under a Creative Commons Attribution 4.0 International License.


