Transcranial direct current stimulation (tDCS) has emerged as a versatile non-invasive neuromodulation approach that can alter cortical excitability and affect network plasticity. Recent advances in machine learning (ML) offer an opportunity to transform tDCS from largely heuristic practice into a quantitatively informed, adaptive intervention paradigm. Here, we synthesize developments from 2020 to 2025 at the intersection of tDCS and ML. Search results from structured PubMed and Google Scholar queries were screened for eligibility based on predefined inclusion criteria, retaining peer-reviewed studies that applied ML techniques to tDCS related studies. Eligible studies were evaluated for data integrity, and ML model validation methodology. Sixteen studies met inclusion criteria. Across these studies, ML was applied to heterogeneous datasets, including electroencephalography, neuroimaging, and clinico–demographic features, to predict stimulation outcomes, characterize neural responses, and identify biomarkers of tDCS sensitivity. Support vector machines and random forests remain prevalent, reflecting the modest scale and exploratory nature of current datasets; most studies rely on early-stage clinical or preclinical cohorts, resulting in promising yet fragmented evidence. Nevertheless, emerging results illustrate how ML can reveal latent physiological structure, guide dose–response optimization, and support the translation of tDCS toward precision neuromodulation. Drawing on this integrated analysis, we highlight key directions for the field: multimodal integration that unifies electrophysiological, structural, and behavioral signatures; incorporation of biophysically grounded forward models and pretrained deep-learning architectures; and development of adaptive, closed-loop control strategies capable of personalizing stimulation in real time. Together, these advances chart a pathway toward ML-guided tDCS systems that are mechanistically informed, clinically actionable, and scalable for widespread application.
- Open Access
- Review
Recent Advancements of Transcranial Direct Current Stimulation and Machine Learning: Methods, Challenges, and Opportunities
- Junfu Cheng 1,
- Tara Sahni 2,
- Zeyun Zhao 3,
- Skylar E. Stolte 4,5,
- Chenyu You 6,7,
- Adam J. Woods 8,
- Aprinda Indahlastari 4,5,
- Ruogu Fang 1,3,4,*
Author Information
Received: 13 Jan 2026 | Revised: 05 Feb 2026 | Accepted: 25 Feb 2026 | Published: 05 Mar 2026
Abstract
Graphical Abstract
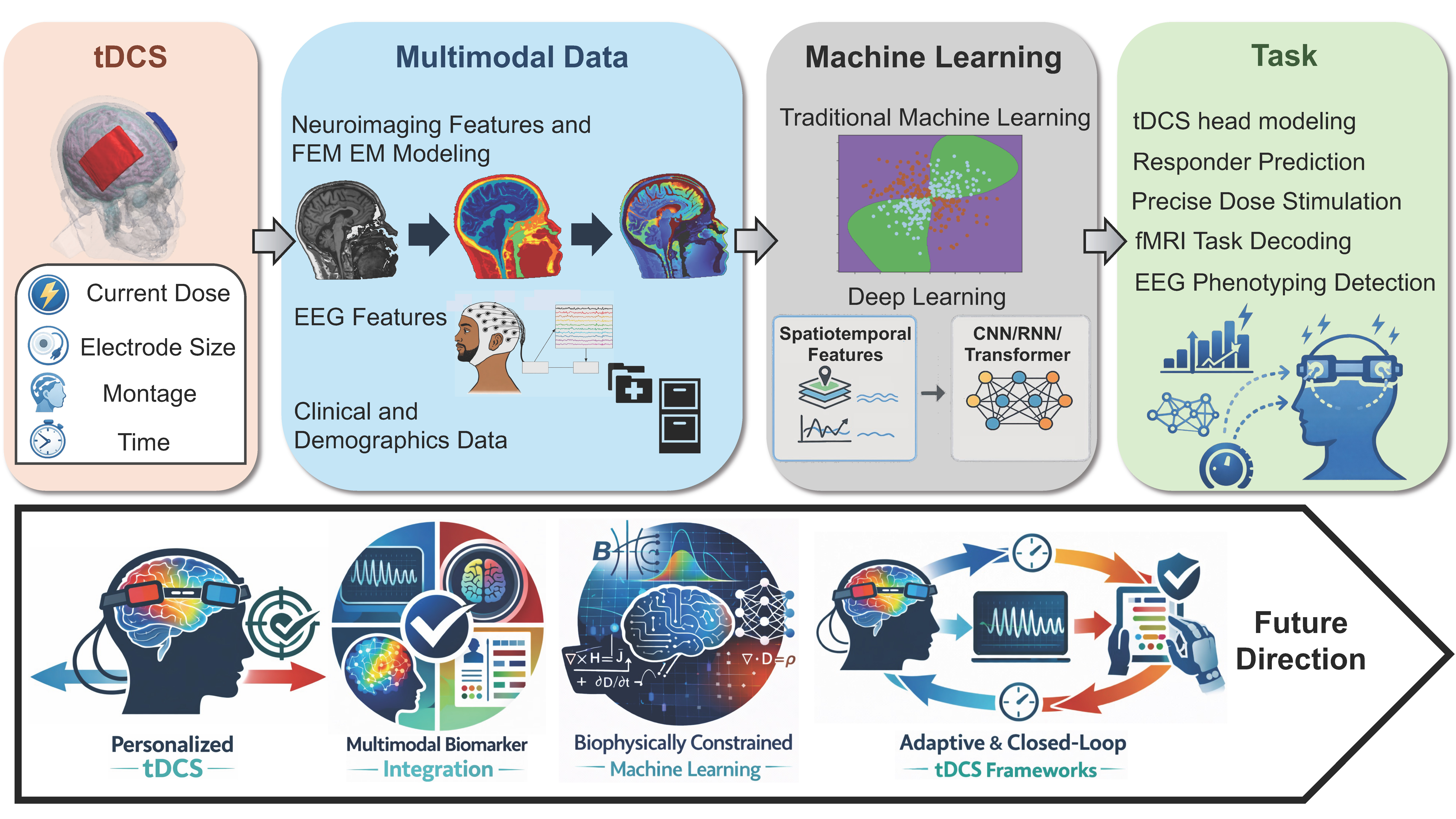
Keywords
transcranial direct current stimulation (tDCS) | machine learning | precision neuromodulation
References
- 1.
Guleyupoglu, B.; Schestatsky, P.; Edwards, D.; et al. Classification of methods in transcranial Electrical Stimulation (tES) and evolving strategy from historical approaches to contemporary innovations. J. Neurosci. Methods 2013, 219, 297–311. https://doi.org/10.1016/j.jneumeth.2013.07.016.
- 2.
Chase, H.W.; Boudewyn, M.A.; Carter, C.S.; et al. Transcranial direct current stimulation: A roadmap for research, from mechanism of action to clinical implementation. Mol. Psychiatry 2020, 25, 397–407. https://doi.org/10.1038/s41380-019-0499-9.
- 3.
Indahlastari, A.; Hardcastle, C.; Albizu, A.; et al. A Systematic Review and Meta-Analysis of Transcranial Direct Current Stimulation to Remediate Age-Related Cognitive Decline in Healthy Older Adults. Neuropsychiatr. Dis. Treat. 2021, 17, 971–990. https://doi.org/10.2147/NDT.S259499.
- 4.
Hausman, H.K.; Alexander, G.E.; Cohen, R.; et al. Primary outcome from the augmenting cognitive training in older adults study (ACT): A tDCS and cognitive training randomized clinical trial. Brain Stimul. 2023, 16, 904–917. https://doi.org/10.1016/j.brs.2023.05.021.
- 5.
Hsu, C.W.; Chou, P.H.; Brunoni, A.R.; et al. Comparing different non-invasive brain stimulation interventions for bipolar depression treatment: A network meta-analysis of randomized controlled trials. Neurosci. Biobehav. Rev. 2024, 156, 105483. https://doi.org/10.1016/j.neubiorev.2023.105483.
- 6.
Salehinejad, M.A.; Ghanavati, E.; Glinski, B.; et al. A systematic review of randomized controlled trials on efficacy and safety of transcranial direct current stimulation in major neurodevelopmental disorders: ADHD, autism, and dyslexia. Brain Behav. 2022, 12, e2724. https://doi.org/10.1002/brb3.2724.
- 7.
Begemann, M.J.; Brand, B.A.; Ćurčić-Blake, B.; et al. Efficacy of non-invasive brain stimulation on cognitive functioning in brain disorders: A meta-analysis. Psychol. Med. 2020, 50, 2465–2486. https://doi.org/10.1017/S0033291720003670.
- 8.
Sun, J.; Yan, F.; Liu, A.; et al. Electrical Stimulation of the Motor Cortex or Paretic Muscles Improves Strength Production in Stroke Patients: A Systematic Review and Meta-Analysis. PMR 2021, 13, 171–179. https://doi.org/10.1002/pmrj.12399.
- 9.
Heiland, L.D.; Owen, J.M., III; Nguyen, S.A.; et al. Neuromodulation for Treatment of Tinnitus: A Systematic Review and Meta-Analysis. Otolaryngol. Head Neck Surg. 2024, 170, 1234–1245. https://doi.org/10.1002/ohn.671.
- 10.
Palm, U.; Hasan, A.; Strube, W.; et al. tDCS for the treatment of depression: A comprehensive review. Eur. Arch. Psychiatry Clin. Neurosci. 2016, 266, 681–694. https://doi.org/10.1007/s00406-016-0674-9.
- 11.
Clark, V.P.; Coffman, B.A.; Trumbo, M.C.; et al. Transcranial direct current stimulation (tDCS) produces localized and specific alterations in neurochemistry: A 1H magnetic resonance spectroscopy study. Neurosci. Lett. 2011, 500, 67–71. https://doi.org/10.1016/j.neulet.2011.05.244.
- 12.
Fritsch, B.; Reis, J.; Martinowich, K.; et al. Direct current stimulation promotes BDNF-dependent synaptic plasticity: Potential implications for motor learning. Neuron 2010, 66, 198–204. https://doi.org/10.1016/j.neuron.2010.03.035.
- 13.
Giordano, J.; Bikson, M.; Kappenman, E.S.; et al. Mechanisms and Effects of Transcranial Direct Current Stimulation. Dose-Response 2017, 15, 1559325816685467. https://doi.org/10.1177/1559325816685467.
- 14.
Solomons, C.D.; Shanmugasundaram, V. Transcranial direct current stimulation: A review of electrode characteristics and materials. Med. Eng. Phys. 2020, 85, 63–74. https://doi.org/10.1016/j.medengphy.2020.09.015.
- 15.
Li, L.M.; Uehara, K.; Hanakawa, T. The contribution of interindividual factors to variability of response in transcranial direct current stimulation studies. Front. Cell. Neurosci. 2015, 9, 181. https://doi.org/10.3389/fncel.2015.00181.
- 16.
Gurr, C.; Splittgerber, M.; Puonti, O.; et al. Neuroanatomical Predictors of Transcranial Direct Current Stimulation (tDCS)-Induced Modifications in Neurocognitive Task Performance in Typically Developing Individuals. J. Neurosci. 2024, 44, e1372232024. https://doi.org/10.1523/JNEUROSCI.1372-23.2024.
- 17.
dos Santos Moraes, W.A.; Poyares, D.R.; Guilleminault, C.; et al. The Effect of Donepezil on Sleep and REM Sleep EEG in Patients with Alzheimer Disease: A Double-Blind Placebo-Controlled Study. Sleep 2006, 29, 199–205. https://doi.org/10.1093/sleep/29.2.199.
- 18.
Babiloni, C.; Carducci, F.; Lizio, R.; et al. Resting state cortical electroencephalographic rhythms are related to gray matter volume in subjects with mild cognitive impairment and Alzheimer’s disease. Hum. Brain Mapp. 2013, 34, 1427–1446. https://doi.org/10.1002/hbm.22005.
- 19.
Andrade, S.M.; da Silva Machado, D.G.; da Silva-Sauerc, L.; et al. Effects of multisite anodal transcranial direct current stimulation combined with cognitive stimulation in patients with Alzheimer’s disease and its neurophysiological correlates: A double-blind randomized clinical trial. Neurophysiol. Clin. 2022, 52, 117–127. https://doi.org/10.1016/j.neucli.2022.02.003.
- 20.
Schmidt, C.F.; Boesiger, P.; Ishai, A. Comparison of fMRI activation as measured with gradient- and spin-echo EPI during visual perception. NeuroImage 2005, 26, 852–859. https://doi.org/10.1016/j.neuroimage.2005.02.043.
- 21.
Schirrmeister, R.T.; Springenberg, J.T.; Fiederer, L.D.J.; et al. Deep learning with convolutional neural networks for EEG decoding and visualization. Hum. Brain Mapp. 2017, 38, 5391–5420. https://doi.org/10.1002/hbm.23730.
- 22.
Basaia, S.; Agosta, F.; Wagner, L.; et al. Automated classification of Alzheimer’s disease and mild cognitive impairment using a single MRI and deep neural networks. NeuroImage Clin. 2019, 21, 101645. https://doi.org/10.1016/j.nicl.2018.101645.
- 23.
Chang, B.; Geng, Z.; Guo, T.; et al. Comprehensive clinical scale-based machine learning model for predicting subthalamic nucleus deep brain stimulation outcomes in Parkinson’s disease. Neurosurg. Rev. 2025, 48, 266. https://doi.org/10.1007/s10143-025-03424-1.
- 24.
Richards, B.A.; Lillicrap, T.P.; Beaudoin, P.; et al. A deep learning framework for neuroscience. Nat. Neurosci. 2019, 22, 1761–1770. https://doi.org/10.1038/s41593-019-0520-2.
- 25.
Saleem, T.J.; Zahra, S.R.; Wu, F.; et al. Deep Learning-Based Diagnosis of Alzheimer’s Disease. J. Pers. Med. 2022, 12, 815. https://doi.org/10.3390/jpm12050815.
- 26.
Pouyanfar, S.; Sadiq, S.; Yan, Y.; et al. A Survey on Deep Learning: Algorithms, Techniques, and Applications. ACM Comput. Surv. 2019, 51, 1–36. https://doi.org/10.1145/3234150.
- 27.
Ruffini, G.; Fox, M.D.; Ripolles, O.; et al. Optimization of multifocal transcranial current stimulation for weighted cortical pattern targeting from realistic modeling of electric fields. NeuroImage 2014, 89, 216–225. https://doi.org/10.1016/j.neuroimage.2013.12.002.
- 28.
Albizu, A.; Indahlastari, A.; Suen, P.; et al. Machine learning-optimized non-invasive brain stimulation and treatment response classification for major depression. Bioelectron. Med. 2024, 10, 25. https://doi.org/10.1186/s42234-024-00157-2.
- 29.
Albizu, A.; Indahlastari, A.; Huang, Z.; et al. Machine-learning defined precision tDCS for improving cognitive function. Brain Stimul. 2023, 16, 969–974. https://doi.org/10.1016/j.brs.2023.05.020.
- 30.
Xiao, W.; Moncy, J.C.; Ghazi-Noori, A.R.; et al. Enhanced network synchronization connectivity following transcranial direct current stimulation (tDCS) in bipolar depression: Effects on EEG oscillations and deep learning-based predictors of clinical remission. J. Affect. Disord. 2025, 369, 576–587. https://doi.org/10.1016/j.jad.2024.09.054.
- 31.
Andrade, S.M.; da Silva-Sauer, L.; de Carvalho, C.D.; et al. Identifying biomarkers for tDCS treatment response in Alzheimer’s disease patients: A machine learning approach using resting-state EEG classification. Front. Hum. Neurosci. 2023, 17, 1234168. https://doi.org/10.3389/fnhum.2023.1234168.
- 32.
Kim, S.; Yang, C.; Dong, S.-Y.; et al. Predictions of tDCS treatment response in PTSD patients using EEG based classification. Front. Psychiatry 2022, 13, 876036. https://doi.org/10.3389/fpsyt.2022.876036.
- 33.
Zhang, C.; Han, S.; Li, Z.; et al. Multidimensional Assessment of Electroencephalography in the Neuromodulation of Disorders of Consciousness. Front. Neurosci. 2022, 16, 903703. https://doi.org/10.3389/fnins.2022.903703.
- 34.
Dagnino, P.C.; Braboszcz, C.; Kroupi, E.; et al. Stratification of responses to tDCS intervention in a healthy pediatric population based on resting-state EEG profiles. Sci. Rep. 2023, 13, 8438. https://doi.org/10.1038/s41598-023-34724-5.
- 35.
Shinde, A.; Mohapatra, S.; Schlaug, G. Identifying the engagement of a brain network during a targeted tDCS-fMRI experiment using a machine learning approach. PLoS Comput. Biol. 2023, 19, e1011012. https://doi.org/10.1371/journal.pcbi.1011012.
- 36.
Wards, Y.; Ehrhardt, S.E.; Garner, K.G.; et al. Stimulating prefrontal cortex facilitates training transfer by increasing representational overlap. Cereb. Cortex 2024, 34, bhae209. https://doi.org/10.1093/cercor/bhae209.
- 37.
Albizu, A.; Fang, R.; Indahlastari, A.; et al. Machine learning and individual variability in electric field characteristics predict tDCS treatment response. Brain Stimul. 2020, 13, 1753–1764. https://doi.org/10.1016/j.brs.2020.10.001.
- 38.
Paul, A.K.; Bose, A.; Kalmady, S.V.; et al. Superior temporal gyrus functional connectivity predicts transcranial direct current stimulation response in Schizophrenia: A machine learning study. Front. Psychiatry 2022, 13, 923938. https://doi.org/10.3389/fpsyt.2022.923938.
- 39.
Cardon, E.; Jacquemin, L.; Schecklmann, M.; et al. Random Forest Classification to Predict Response to High-Definition Transcranial Direct Current Stimulation for Tinnitus Relief: A Preliminary Feasibility Study. Ear Hear. 2022, 43, 1816–1823. https://doi.org/10.1097/AUD.0000000000001246.
- 40.
Jia, X.; Sayed, S.B.; Hasan, N.I.; et al. DeeptDCS: Deep Learning-Based Estimation of Currents Induced During Transcranial Direct Current Stimulation. IEEE Trans. Biomed. Eng. 2023, 70, 1231–1241. https://doi.org/10.1109/TBME.2022.3213266.
- 41.
Lee, J.; Lee, M.; Lee, J.; et al. Fine-grained brain tissue segmentation for brain modeling of stroke patient. Comput. Biol. Med. 2023, 153, 106472. https://doi.org/10.1016/j.compbiomed.2022.106472.
- 42.
Stolte, S.E.; Indahlastari, A.; Chen, J.; et al. Precise and rapid whole-head segmentation from magnetic resonance images of older adults using deep learning. Imaging Neurosci. 2024, 2, 1–21. https://doi.org/10.1162/imag_a_00090.
- 43.
Huang, Y.; Datta, A.; Bikson, M.; et al. Realistic volumetric-approach to simulate transcranial electric stimulation—ROAST—A fully automated open-source pipeline. J. Neural Eng. 2019, 16, 056006. https://doi.org/10.1088/1741-2552/ab208d.
- 44.
Indahlastari, A.; Albizu, A.; O’Shea, A.; et al. Modeling transcranial electrical stimulation in the aging brain. Brain Stimul. 2020, 13, 664–674. https://doi.org/10.1016/j.brs.2020.02.007.
- 45.
Henschel, L.; Conjeti, S.; Estrada, S.; et al. FastSurfer—A fast and accurate deep learning based neuroimaging pipeline. NeuroImage 2020, 219, 117012. https://doi.org/10.1016/j.neuroimage.2020.117012.
- 46.
Rashed, E.A.; Gomez-Tames, J.; Hirata, A. Deep Learning-Based Development of Personalized Human Head Model with Non-Uniform Conductivity for Brain Stimulation. IEEE Trans. Med. Imaging 2020, 39, 2351–2362. https://doi.org/10.1109/TMI.2020.2969682.
- 47.
Guha Roy, A.; Conjeti, S.; Navab, N.; et al. QuickNAT: A fully convolutional network for quick and accurate segmentation of neuroanatomy. NeuroImage 2019, 186, 713–727. https://doi.org/10.1016/j.neuroimage.2018.11.042.
- 48.
Mackay, C.T.; Nowell, D. Informed machine learning methods for application in engineering: A review. Proc. Inst. Mech. Eng. Part C J. Mech. Eng. Sci. 2023, 237, 4525–4544. https://doi.org/10.1177/09544062231164575.
- 49.
Van Norman, G.A. Drugs, Devices, and the FDA: Part 2: An Overview of Approval Processes: FDA Approval of Medical Devices. JACC Basic Transl. Sci. 2016, 1, 277–287. https://doi.org/10.1016/j.jacbts.2016.03.009.
- 50.
21 CFR 860.7—Determination of Safety and Effectiveness. Available online: https://www.ecfr.gov/current/title-21/part-860/section-860.7 (accessed on 15 November 2025).
- 51.
Kaplan, A.V.; Baim, D.S.; Smith, J.J.; et al. Medical Device Development: From prototype to regulatory approval. Circulation 2004, 109, 3068–3072. https://doi.org/10.1161/01.CIR.0000134695.65733.64.
- 52.
Herrmann, R.; Dreher, M.; Farb, A.; et al. US FDA best practices for initiating early feasibility studies for neurological devices in the United States. J. Neurosurg. 2021, 136, 282–286. https://doi.org/10.3171/2020.11.JNS203653.
- 53.
National Academies of Sciences, Engineering, and Medicine. Preventing and Treating Dementia: Research Priorities to Accelerate Progress; The National Academies Press: Washington, DC, USA, 2025; p. 28588.
- 54.
Peralta, M.; Jannin, P.; Baxter, J.S.H. Machine learning in deep brain stimulation: A systematic review. Artif. Intell. Med. 2021, 122, 102198. https://doi.org/10.1016/j.artmed.2021.102198.
- 55.
Gebodh, N.; Miskovic, V.; Laszlo, S.; et al. A Scalable Framework for Closed-Loop Neuromodulation with Deep Learning. bioRxiv 2023, 2023.01.18.524615. https://doi.org/10.1101/2023.01.18.524615.
- 56.
Mellot, A.; Collas, A.; Chevallier, S.; et al. Physics-Informed and Unsupervised Riemannian Domain Adaptation for Machine Learning on Heterogeneous EEG Datasets. In Proceedings of the 2024 32nd European Signal Processing Conference (EUSIPCO), Lyon, France, 26–30 August 2024; pp. 1367–1371.
- 57.
Baillet, S.; Mosher, J.C.; Leahy, R.M. Electromagnetic brain mapping. IEEE Signal Process. Mag. 2001, 18, 14–30. https://doi.org/10.1109/79.962275.
- 58.
Morik, M.; Hashemi, A.; Müller, K.-R.; et al. Enhancing Brain Source Reconstruction through Physics-Informed 3D Neural Networks. arXiv 2024, arXiv:2411.00143. https://doi.org/10.48550/arXiv.2411.00143.
- 59.
Krishnapriya, S.; Karuna, Y. Pre-trained deep learning models for brain MRI image classification. Front. Hum. Neurosci. 2023, 17, 1150120. https://doi.org/10.3389/fnhum.2023.1150120.
- 60.
Wang, C.; Jiang, Y.; Peng, Z.; et al. Towards a general-purpose foundation model for fMRI analysis. arXiv 2025, arXiv:2506.11167. https://doi.org/10.48550/arXiv.2506.11167.
- 61.
Caro, J.O.; Fonseca, A.H.D.O.; Averill, C.; et al. BrainLM: A Foundation Model for Brain Activity Recordings. Presented at the Twelfth International Conference on Learning Representations (ICLR 2024). Available online: https://openreview.net/forum?id=RwI7ZEfR27 (accessed on 20 August 2025).
- 62.
Wei, X.; Zhao, K.; Jiao, Y.; et al. A Brain Graph Foundation Model: Pre-Training and Prompt-Tuning for Any Atlas and Disorder. arXiv 2025, arXiv:2506.02044. https://doi.org/10.48550/arXiv.2506.02044.
- 63.
Lai, J.; Wei, J.; Yao, L.; et al. A Simple Review of EEG Foundation Models: Datasets, Advancements and Future Perspectives. arXiv 2025, arXiv:2504.20069. https://doi.org/10.48550/arXiv.2504.20069.
- 64.
Radford, A.; Kim, J.W.; Hallacy, C.; et al. Learning Transferable Visual Models from Natural Language Supervision. arXiv 2021, arXiv:2103.00020. https://doi.org/10.48550/arXiv.2103.00020.

This work is licensed under a Creative Commons Attribution 4.0 International License.


