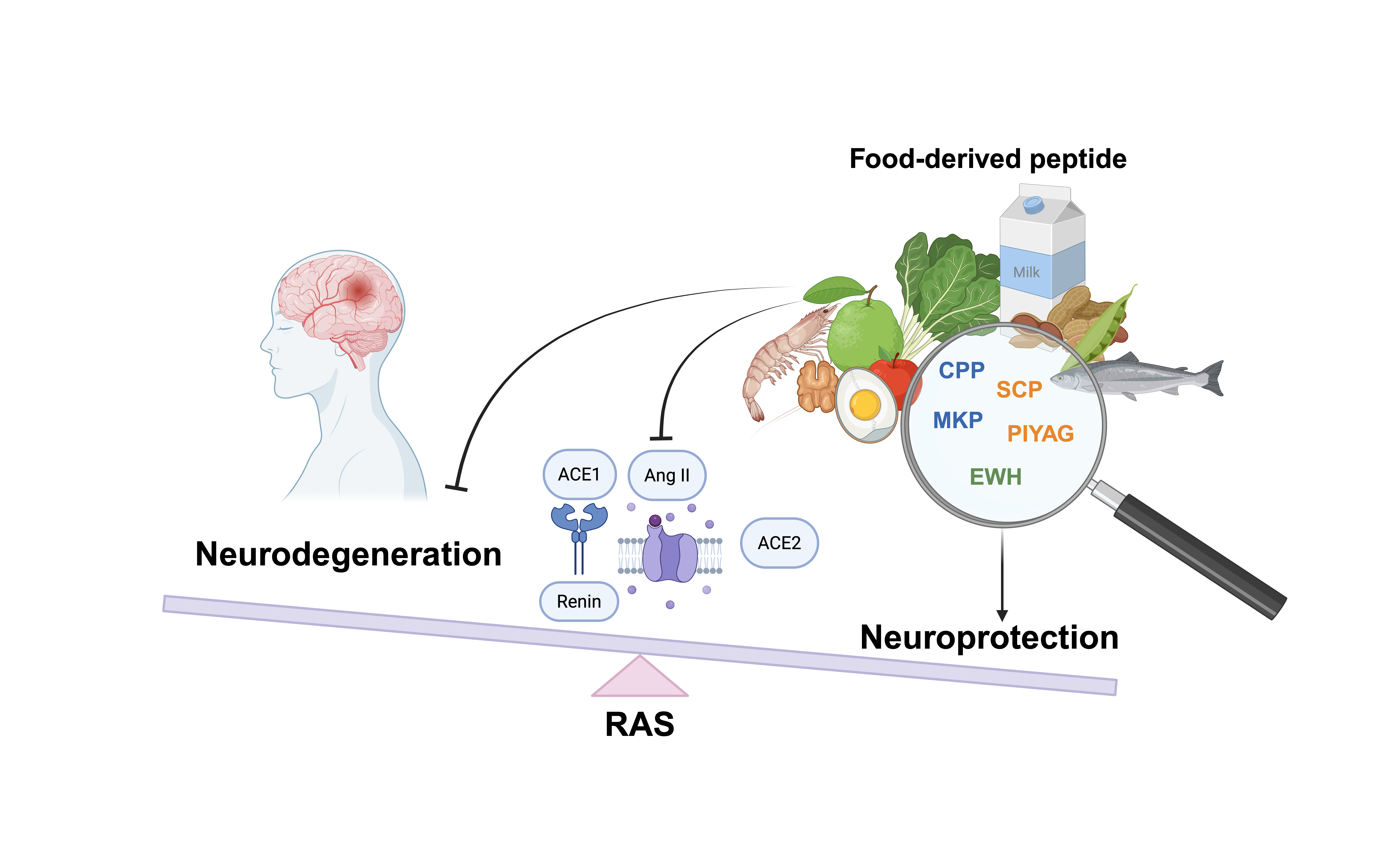
Neurodegenerative disease (ND) is a growing global health challenge with limited therapeutic options. Emerging research suggests food-derived bioactive peptides may exhibit neuroprotective activities by regulating the antioxidation, anti-neuroinflammation signaling pathways and the neurotransmitters release in the brain. The regulation is associated with the key components in the renin angiotensin system (RAS). In this paper, we review studies published up to 2025, focusing on the regulation of RAS components and RAS related neurotransmitters by food-derived bioactive peptides. This review describes the crucial role of RAS in the pathophysiology of ND, and discusses how food-derived peptides modulate RAS-mediated neurotransmission and its downstream signaling cascades. Specifically, the structure of peptides affects their penetrability to the blood-brain barrier (BBB), which in turn influences their neuroprotective efficacy. The evidence from animal models indicates that these peptides can affect RAS-related downstream pathways to potentially mitigate the loss of serotonergic and dopaminergic neurons, which are vital for cognition and memory. The findings suggest that food-derived bioactive peptides may hold potential for ameliorating ND symptoms via the regulation of RAS.
- Open Access
- Review
Food-Derived Bioactive Peptides Prevent Neurodegenerative Disease by Regulating Renin Angiotensin System (RAS) Related Neurotransmitters and Pathways
Author Information
Received: 15 Aug 2025 | Revised: 07 Feb 2026 | Accepted: 09 Feb 2026 | Published: 03 Mar 2026
Abstract
Graphical Abstract
Keywords
food-derived bioactive peptides | neurodegenerative disease | renin angiotensin system | neuroinflammation | blood-brain barrier
References
- 1.
Adampourezare, M.; Hasanzadeh, M.; Nikzad, B. Recent progress and challenges in the application of molecularly imprinted polymers for early-stage screening of neurodegenerative diseases-related protein biomarkers. Microchem. J. 2023, 192, 108931.
- 2.
Calderon-Garciduenas, L. Common Fatal Neurodegenerative Diseases Revisited: Beyond Age, Comorbidities, and Devastating Terminal Neuropathology There Is Hope with Prevention. Front. Neurol. 2022, 13, 901447.
- 3.
Gadhave, D.G.; Sugandhi, V.V.; Jha, S.K.; et al. Neurodegenerative disorders: Mechanisms of degeneration and therapeutic approaches with their clinical relevance. Ageing Res. Rev. 2024, 99, 102357.
- 4.
Imam, F.; Saloner, R.; Vogel, J.W.; et al. The Global Neurodegeneration Proteomics Consortium: Biomarker and drug target discovery for common neurodegenerative diseases and aging. Nat. Med. 2025, 31, 2556–2566.
- 5.
Alzheimer’s Association. 2023 Alzheimer’s Disease Facts and Figures; Alzheimer’s Association: Chicago, IL, USA, 2023; pp. 1598–1695.
- 6.
Alzheimer’s Association. 2024 Alzheimer’s Disease Facts and Figures; Alzheimer’s Association: Chicago, IL, USA, 2024; pp. 3708–3821.
- 7.
Jia, L.; Du, Y.; Chu, L.; et al. Prevalence, risk factors, and management of dementia and mild cognitive impairment in adults aged 60 years or older in China: A cross-sectional study. Lancet Public. Health 2020, 5, e661–e671.
- 8.
Alzheimer Europe. Dementia in Europe Yearbook 2019: Estimating the Prevalence of Dementia in Europe; Alzheimer Europe: Luxembourg, 2020; p. 16.
- 9.
Su, D.; Cui, Y.; He, C.; et al. Projections for prevalence of Parkinson’s disease and its driving factors in 195 countries and territories to 2050: Modelling study of Global Burden of Disease Study 2021. BMJ 2025, 388, e080952.
- 10.
Lei, J.; Gillespie, K. Projected Global Burden of Brain Disorders Through 2050 (P7-15.001). Neurology 2024, 102, 3234.
- 11.
Nandi, A.; Counts, N.; Chen, S.; et al. Global and regional projections of the economic burden of Alzheimer’s disease and related dementias from 2019 to 2050: A value of statistical life approach. EClinicalMedicine 2022, 51, 101580.
- 12.
Liu, T.; Li, J.; Sun, L.; et al. The role of ACE2 in RAS axis on microglia activation in Parkinson’s disease. Neuroscience 2024, 553, 128–144.
- 13.
Ramalingam, M.; Jang, S.; Jeong, H.S. Therapeutic Effects of Conditioned Medium of Neural Differentiated Human Bone Marrow-Derived Stem Cells on Rotenone-Induced Alpha-Synuclein Aggregation and Apoptosis. Stem Cells Int. 2021, 2021, 6658271.
- 14.
Dolgacheva, L.P.; Zinchenko, V.P.; Goncharov, N.V. Molecular and Cellular Interactions in Pathogenesis of Sporadic Parkinson Disease. Int. J. Mol. Sci. 2022, 23, 13043.
- 15.
Zhang, J.; Zhang, Y.; Wang, J.; et al. Recent advances in Alzheimer’s disease: Mechanisms, clinical trials and new drug development strategies. Signal Transduct. Target. Ther. 2024, 9, 211.
- 16.
Dzidic-Krivic, A.; Fajkic, A.; Farhat, E.K.; et al. Linking Metabolic Syndrome to Neurodegeneration Mechanisms and Potential Treatments. Mol. Neurobiol. 2025, 62, 14344–14366.
- 17.
Luther, J.M.; Brown, N.J. The renin-angiotensin-aldosterone system and glucose homeostasis. Trends Pharmacol. Sci. 2011, 32, 734–739.
- 18.
Sarkar, S.; Jayachandra, K.; Vishwanath, B.S. ACE 2/Ang (1–7)/Mas, Non-conventional RAS Axis: Endogenous Contributor of Cardio, and Reno-protective Responses. J. Cell. Signal. 2024, 5, 149–161.
- 19.
Loera-Valencia, R.; Eroli, F.; Garcia-Ptacek, S.; et al. Brain Renin-Angiotensin System as Novel and Potential Therapeutic Target for Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 22, 10139.
- 20.
Alenina, N.; Bader, M. ACE2 in Brain Physiology and Pathophysiology: Evidence from Transgenic Animal Models. Neurochem. Res. 2019, 44, 1323–1329.
- 21.
Rocha, N.P.; Simoes, E.S.A.C.; Teixeira, A.L. Editorial: The Role of the Renin-Angiotensin System in the Central Nervous System. Front. Neurosci. 2021, 15, 733084.
- 22.
Miners, J.S.; Ashby, E.; Van Helmond, Z.; et al. Angiotensin-converting enzyme (ACE) levels and activity in Alzheimer’s disease, and relationship of perivascular ACE-1 to cerebral amyloid angiopathy. Neuropathol. Appl. Neurobiol. 2008, 34, 181–193.
- 23.
Jeon, S.; Salvo, M.A.; Alia, A.O.; et al. Neuronal ACE1 knockout disrupts the hippocampal renin angiotensin system leading to memory impairment and vascular loss in normal aging. Neurobiol. Dis. 2024, 202, 106729.
- 24.
Tayler, H.M.; Skrobot, O.A.; Baron, D.H.; et al. Dysregulation of the renin-angiotensin system in vascular dementia. Brain Pathol. 2024, 34, e13251.
- 25.
Kobiec, T.; Otero-Losada, M.; Chevalier, G.; et al. The Renin-Angiotensin System Modulates Dopaminergic Neurotransmission: A New Player on the Scene. Front. Synaptic Neurosci. 2021, 13, 638519.
- 26.
Sica, D.A. Angiotensin-Converting Enzyme Inhibitors’ Side Effects—Physiologic and Non-Physiologic Considerations. J. Clin. Hypertens. 2005, 7, 17–23.
- 27.
Novotny, M.; Klimova, B.; Valis, M. Microbiome and Cognitive Impairment: Can Any Diets Influence Learning Processes in a Positive Way? Front. Aging Neurosci. 2019, 11, 170.
- 28.
Lu, Z.; Sun, N.; Dong, L.; et al. Production of Bioactive Peptides from Sea Cucumber and Its Potential Health Benefits: A Comprehensive Review. J. Agric. Food Chem. 2022, 70, 7607–7625.
- 29.
Parameswari, R.P.; Lakshmi, T. Microalgae as a potential therapeutic drug candidate for neurodegenerative diseases. J. Biotechnol. 2022, 358, 128–139.
- 30.
Ding, J.; Liang, R.; Yang, Y.; et al. Optimization of pea protein hydrolysate preparation and purification of antioxidant peptides based on an in silico analytical approach. Lwt 2020, 123, 109126.
- 31.
Malta, S.M.; Batista, L.L.; Silva, H.C.G.; et al. Identification of bioactive peptides from a Brazilian kefir sample, and their anti-Alzheimer potential in Drosophila melanogaster. Sci. Rep. 2022, 12, 11065.
- 32.
Hsieh, L.-S.; Hsu, Y.-C.; Chiang, W.-D. Neuroprotective peptides isolated from flavourzyme-pea protein hydrolysate protect human SH-SY5Y cells from Aβ1-42 induced apoptosis. J. Funct. Foods 2023, 108, 105755.
- 33.
Lu, Z.; Yang, J.; Xu, X.; et al. Regulation mechanisms of sea cucumber peptides against scopolamine-induced memory disorder and novel memory-improving peptides identification. Eur. J. Pharmacol. 2024, 968, 176430.
- 34.
Wang, S.M.; Chuu, J.J.; Lee, C.K.; et al. Exploring the therapeutic efficacy of Chlorella pyrenoidosa peptides in ameliorating Alzheimer’s disease. Heliyon 2023, 9, e15406.
- 35.
Ahmed, H.A.; Ishrat, T.; Pillai, B.; et al. Role of angiotensin system modulation on progression of cognitive impairment and brain MRI changes in aged hypertensive animals - A randomized double- blind pre-clinical study. Behav. Brain Res. 2018, 346, 29–40.
- 36.
Labandeira-Garcia, J.L.; Rodriguez-Perez, A.I.; Garrido-Gil, P.; et al. Brain Renin-Angiotensin System and Microglial Polarization: Implications for Aging and Neurodegeneration. Front. Aging Neurosci. 2017, 9, 129.
- 37.
Jackson, L.; Eldahshan, W.; Fagan, S.C.; et al. Within the Brain: The Renin Angiotensin System. Int. J. Mol. Sci. 2018, 19, 876.
- 38.
Chen, S.D.; Wu, C.L.; Lin, T.K.; et al. Renin inhibitor aliskiren exerts neuroprotection against amyloid beta-peptide toxicity in rat cortical neurons. Neurochem. Int. 2012, 61, 369–377.
- 39.
Dong, Y.F.; Kataoka, K.; Toyama, K.; et al. Attenuation of brain damage and cognitive impairment by direct renin inhibition in mice with chronic cerebral hypoperfusion. Hypertension 2011, 58, 635–642.
- 40.
Shinohara, K.; Liu, X.; Morgan, D.A.; et al. Selective Deletion of the Brain-Specific Isoform of Renin Causes Neurogenic Hypertension. Hypertension 2016, 68, 1385–1392.
- 41.
Labandeira-Garcia, J.L.; Labandeira, C.M.; Guerra, M.J.; et al. The role of the brain renin-angiotensin system in Parkinson’s disease. Transl. Neurodegener. 2024, 13, 22.
- 42.
Perez-Lloret, S.; Otero-Losada, M.; Toblli, J.E.; et al. Renin-angiotensin system as a potential target for new therapeutic approaches in Parkinson’s disease. Expert. Opin. Investig. Drugs 2017, 26, 1163–1173.
- 43.
Kunkle, B.W.; Grenier-Boley, B.; Sims, R.; et al. Genetic meta-analysis of diagnosed Alzheimer’s disease identifies new risk loci and implicates Aβ, tau, immunity and lipid processing. Nat. Genet. 2019, 51, 414–430.
- 44.
Jochemsen, H.M.; Teunissen, C.E.; Ashby, E.L.; et al. The association of angiotensin-converting enzyme with biomarkers for Alzheimer’s disease. Alzheimer’s Res. Ther. 2014, 6, 27.
- 45.
Xie, J.; Chen, S.; Huan, P.; et al. A novel angiotensin I-converting enzyme inhibitory peptide from walnut (Juglans sigillata) protein hydrolysates and its evaluation in Ang II-induced HUVECs and hypertensive rats. Int. J. Biol. Macromol. 2024, 266, 131152.
- 46.
Song, W.; Huang, H.; Shen, Y.; et al. Hazelnut-Derived Peptide YYLLVR Improves Endothelial Dysfunction in Hypertension by Activating ACE2. J. Agric. Food Chem. 2025, 73, 17024–17039.
- 47.
Van Thiel, B.S.; Goes Martini, A.; Te Riet, L.; et al. Brain Renin-Angiotensin System: Does It Exist? Hypertension 2017, 69, 1136–1144.
- 48.
Biancardi, V.C.; Son, S.J.; Ahmadi, S.; et al. Circulating angiotensin II gains access to the hypothalamus and brain stem during hypertension via breakdown of the blood-brain barrier. Hypertension 2014, 63, 572–579.
- 49.
Ren, L.; Lu, X.; Danser, A.H.J. Revisiting the Brain Renin-Angiotensin System-Focus on Novel Therapies. Curr. Hypertens. Rep. 2019, 21, 28.
- 50.
Gouveia, F.; Camins, A.; Ettcheto, M.; et al. Targeting brain Renin-Angiotensin System for the prevention and treatment of Alzheimer’s disease: Past, present and future. Ageing Res. Rev. 2022, 77, 101612.
- 51.
Subudhi, B.B.; Sahu, P.K. Chapter 4—Targeting Renin–Angiotensin System: A Strategy for Drug Development Against Neurological Disorders. In Angiotensin; Pilowsky, P.M., Ed.; Academic Press: Cambridge, MA, USA, 2023; pp. 107–150.
- 52.
Inaba, S.; Iwai, M.; Furuno, M.; et al. Continuous activation of renin-angiotensin system impairs cognitive function in renin/angiotensinogen transgenic mice. Hypertension 2009, 53, 356–362.
- 53.
Iwai, M.; Liu, H.W.; Chen, R.; et al. Possible inhibition of focal cerebral ischemia by angiotensin II type 2 receptor stimulation. Circulation 2004, 110, 843–848.
- 54.
Chelmis, F.; Sorotou, I.; Pakataridis, P.; et al. The Ameliorative Effects of AT2 Receptor Activation with the Hexapeptide Novokinin on Streptozotocin-induced Model of Alzheimer’s Disease in SHR. Proc. Bulg. Acad. Sci. 2024, 77, 1852–1860.
- 55.
Cosarderelioglu, C.; Nidadavolu, L.S.; George, C.J.; et al. Brain Renin-Angiotensin System at the Intersect of Physical and Cognitive Frailty. Front. Neurosci. 2020, 14, 586314.
- 56.
Labandeira-Garcia, J.L.; Rodriguez-Pallares, J.; Villar-Cheda, B.; et al. Aging, Angiotensin System and Dopaminergic Degeneration in the Substantia Nigra. Aging Dis. 2011, 2, 257–274.
- 57.
Mei, M.; Zhou, Y.; Liu, M.; et al. Antioxidant and anti-inflammatory effects of dexrazoxane on dopaminergic neuron degeneration in rodent models of Parkinson’s disease. Neuropharmacology 2019, 160, 107758.
- 58.
Delva, A.; Van Weehaeghe, D.; Koole, M.; et al. Loss of Presynaptic Terminal Integrity in the Substantia Nigra in Early Parkinson’s Disease. Mov. Disord. 2020, 35, 1977–1986.
- 59.
Gildea, J.J. Dopamine and angiotensin as renal counterregulatory systems controlling sodium balance. Curr. Opin. Nephrol. Hypertens. 2009, 18, 28–32.
- 60.
Buck, S.A.; Erickson-Oberg, M.Q.; Bhatte, S.H.; et al. Roles of VGLUT2 and Dopamine/Glutamate Co-Transmission in Selective Vulnerability to Dopamine Neurodegeneration. ACS Chem. Neurosci. 2022, 13, 187–193.
- 61.
Daniels, R.W.; Miller, B.R.; DiAntonio, A. Increased vesicular glutamate transporter expression causes excitotoxic neurodegeneration. Neurobiol. Dis. 2011, 41, 415–420.
- 62.
Gabor, A.; Leenen, F.H. Central mineralocorticoid receptors and the role of angiotensin II and glutamate in the paraventricular nucleus of rats with angiotensin II-induced hypertension. Hypertension 2013, 61, 1083–1090.
- 63.
Kim, O.-H.; Lee, G.Y.; Kim, K.Y.; et al. 3-Hydroxymorphinan protects against hypoxia-induced cell death in primary astrocyte by regulating Ca2+ influx and the glutamate homeostasis. Mol. Cell. Toxicol. 2022, 19, 145–153.
- 64.
Xu, J.; Sriramula, S.; Lazartigues, E. Excessive Glutamate Stimulation Impairs ACE2 Activity Through ADAM17-Mediated Shedding in Cultured Cortical Neurons. Cell Mol. Neurobiol. 2018, 38, 1235–1243.
- 65.
Ceyzeriat, K.; Gloria, Y.; Tsartsalis, S.; et al. Alterations in dopamine system and in its connectivity with serotonin in a rat model of Alzheimer’s disease. Brain Commun. 2021, 3, fcab029.
- 66.
Klempin, F.; Mosienko, V.; Matthes, S.; et al. Depletion of angiotensin-converting enzyme 2 reduces brain serotonin and impairs the running-induced neurogenic response. Cell Mol. Life Sci. 2018, 75, 3625–3634.
- 67.
Tian, J.; Stucky, C.S.; Wang, T.; et al. Mitochondrial Dysfunction Links to Impaired Hippocampal Serotonin Release in a Mouse Model of Alzheimer’s Disease. J. Alzheimers Dis. 2023, 93, 605–619.
- 68.
Szapacs, M.E.; Mathews, T.A.; Tessarollo, L.; et al. Exploring the relationship between serotonin and brain-derived neurotrophic factor: Analysis of BDNF protein and extraneuronal 5-HT in mice with reduced serotonin transporter or BDNF expression. J. Neurosci. Methods 2004, 140, 81–92.
- 69.
Duffuler, P.; Bhullar, K.S.; de Campos Zani, S.C.; et al. Bioactive Peptides: From Basic Research to Clinical Trials and Commercialization. J. Agric. Food Chem. 2022, 70, 3585–3595.
- 70.
Basha, S.; Ks, P.; Chattopadhyay, A.; et al. Emerging insights into dairy products and Alzheimer’s disease: Exploring the potential neuroprotective effects. Crit. Rev. Food Sci. Nutr. 2025, 1–28. https://doi.org/10.1080/10408398.2025.2578711.
- 71.
Akazawa, N.; Hamasaki, A.; Tanahashi, K.; et al. Lactotripeptide ingestion increases cerebral blood flow velocity in middle-aged and older adults. Nutr. Res. 2018, 53, 61–66.
- 72.
Butikofer, U.; Meyer, J.; Sieber, R.; et al. Occurrence of the angiotensin-converting enzyme inhibiting tripeptides Val-Pro-Pro and Ile-Pro-Pro in different cheese varieties of Swiss origin. J. Dairy. Sci. 2008, 91, 29–38.
- 73.
Aihara, K.; Kajimoto, O.; Hirata, H.; et al. Effect of Powdered Fermented Milk with Lactobacillus helveticus on Subjects with High-Normal Blood Pressure or Mild Hypertension. J. Am. Coll. Nutr. 2005, 24, 257–265.
- 74.
Kajimoto, O.; Aihara, K.; Hirata, H.; et al. Hypotensive Effects of The Tablets Containing “Lactotripeptides (VPP, IPP)”. J. Nutr. Food 2001, 4, 51–61.
- 75.
Gregory, K.S.; Cozier, G.E.; Schwager, S.L.U.; et al. Structural insights into the inhibitory mechanism of angiotensin-I-converting enzyme by the lactotripeptides IPP and VPP. FEBS Lett. 2024, 598, 242–251.
- 76.
Yamada, A.; Sakurai, T.; Ochi, D.; et al. Novel angiotensin I-converting enzyme inhibitory peptide derived from bovine casein. Food Chem. 2013, 141, 3781–3789.
- 77.
Tada, A.M.; Hamezah, H.S.; Yanagisawa, D.; et al. Neuroprotective Effects of Casein-Derived Peptide Met-Lys-Pro (MKP) in a Hypertensive Model. Front. Neurosci. 2020, 14, 845.
- 78.
Sanjukta, S.; Rai, A.K. Production of bioactive peptides during soybean fermentation and their potential health benefits. Trends Food Sci. Technol. 2016, 50, 1–10.
- 79.
Patel, K.; Mani, A. Food-derived Peptides as Promising Neuroprotective Agents: Mechanism and Therapeutic Potential. Curr. Top. Med. Chem. 2024, 24, 1212–1229.
- 80.
Ju, D.T.; Kuo, W.W.; Ho, T.-J.; et al. Bioactive Peptide VHVV Upregulates the Long-Term Memory-Related Biomarkers in Adult Spontaneously Hypertensive Rats. Int. J. Mol. Sci. 2019, 20, 3069.
- 81.
Song, W.; Fu, J.; Zeng, Q.; et al. Improving ACE inhibitory activity of hazelnut peptide modified by plastein: Physicochemical properties and action mechanism. Food Chem. 2023, 402, 134498.
- 82.
Zhao, F.; Guo, L.; Huang, T.; et al. Interaction between the Neuroprotective and Hyperglycemia Mitigation Effects of Walnut-Derived Peptide LVRL via the Wnt3a/β-Catenin/GSK-3β Pathway in a Type 2 Diabetes Mellitus Model. J. Agric. Food Chem. 2024, 72, 16204–16220.
- 83.
Zheng, X.; Li, D.S.; Ding, K. Purification and identification of angiotensin I-converting enzyme inhibitory peptides from fermented walnut residues. Int. J. Food Prop. 2018, 20, S3326–S3333.
- 84.
Zhao, F.; Liu, C.; Fang, L.; et al. Walnut-Derived Peptide Activates PINK1 via the NRF2/KEAP1/HO-1 Pathway, Promotes Mitophagy, and Alleviates Learning and Memory Impairments in a Mice Model. J. Agric. Food Chem. 2021, 69, 2758–2772.
- 85.
Li, X.; Guo, M.; Chi, J.; et al. Bioactive Peptides from Walnut Residue Protein. Molecules 2020, 25, 1285.
- 86.
Li, Z.; Dang, Q.; Wang, P.; et al. Food-Derived Peptides: Beneficial CNS Effects and Cross-BBB Transmission Strategies. J. Agric. Food Chem. 2023, 71, 20453–20478.
- 87.
Zheng, J.; Gao, Y.; Ding, J.; et al. Antarctic krill peptides improve scopolamine-induced memory impairment in mice. Food Biosci. 2022, 49, 101987.
- 88.
Ren, Q.; Jiang, X.; Zhang, S.; et al. Neuroprotective effect of YIAEDAER peptide against Parkinson’s disease like pathology in zebrafish. Biomed. Pharmacother. 2022, 147, 112629.
- 89.
Yang, J.; Ding, J.; Lu, Z.; et al. Digestive and Absorptive Properties of the Antarctic Krill Tripeptide Phe-Pro-Phe (FPF) and Its Auxiliary Memory-Enhancing Effect. J. Agric. Food Chem. 2024, 72, 8491–8505.
- 90.
Zhang, Q.; Zhao, L.; Li, C.; et al. Investigation of a Neuroprotective Peptide from Round Scad (Decapterus maruadsi): Bioactivity Validation, Gastrointestinal Digestion Stability, and Structure-Activity Relationship Analysis. J. Agric. Food Chem. 2025, 73, 32302–32314.
- 91.
Lu, Z.; Lv, R.; Dong, L.; et al. Sea cucumber peptides protect against memory impairment by regulating dopamine/serotonin metabolization and synapse plasticity of mice hippocampus. J. Funct. Foods 2023, 108, 105732.
- 92.
Lemus-Conejo, A.; Millan-Linares, M.D.C.; Toscano, R.; et al. GPETAFLR, a peptide from Lupinus angustifolius L. prevents inflammation in microglial cells and confers neuroprotection in brain. Nutr. Neurosci. 2022, 25, 472–484.
- 93.
Ni, L.; Zhuge, F.; Yang, S.; et al. Hydrolyzed Chicken Meat Extract Attenuates Neuroinflammation and Cognitive Impairment in Middle-Aged Mouse by Regulating M1/M2 Microglial Polarization. J. Agric. Food Chem. 2021, 69, 9800–9812.
- 94.
Li, B.; Shi, X.; Chen, E.; et al. Improvement effects of cyclic peptides from Annona squamosa on cognitive decline in neuroinflammatory mice. Food Sci. Biotechnol. 2024, 33, 1437–1448.
- 95.
Min, L.J.; Kobayashi, Y.; Mogi, M.; et al. Administration of bovine casein-derived peptide prevents cognitive decline in Alzheimer disease model mice. PLoS ONE 2017, 12, e0171515.
- 96.
Rizzetti, D.A.; Fernandez, F.; Moreno, S.; et al. Egg white hydrolysate promotes neuroprotection for neuropathic disorders induced by chronic exposure to low concentrations of mercury. Brain Res. 2016, 1646, 482–489.
- 97.
Yu, T.; Guo, J.; Zhu, S.; et al. Protective effects of selenium-enriched peptides from Cardamine violifolia on d-galactose-induced brain aging by alleviating oxidative stress, neuroinflammation, and neuron apoptosis. J. Funct. Foods 2020, 75, 104277.
- 98.
Reed, B.N.; Sueta, C.A. A practical guide for the treatment of symptomatic heart failure with reduced ejection fraction (HFrEF). Curr. Cardiol. Rev. 2015, 11, 23–32.
- 99.
Esteras, R.; Perez-Gomez, M.V.; Rodriguez-Osorio, L.; et al. Combination use of medicines from two classes of renin–angiotensin system blocking agents: Risk of hyperkalemia, hypotension, and impaired renal function. Ther. Adv. Drug Saf. 2015, 6, 166–176.
- 100.
Schaafsma, G. Safety of protein hydrolysates, fractions thereof and bioactive peptides in human nutrition. Eur. J. Clin. Nutr. 2009, 63, 1161–1168.
- 101.
Chakrabarti, S.; Jahandideh, F.; Wu, J. Food-derived bioactive peptides on inflammation and oxidative stress. Biomed. Res. Int. 2014, 2014, 608979.
- 102.
Meleti, E.; Koureas, M.; Manouras, A.; et al. Bioactive Peptides from Dairy Products: A Systematic Review of Advances, Mechanisms, Benefits, and Functional Potential. Dairy 2025, 6, 65.
- 103.
Chakrabarti, S.; Guha, S.; Majumder, K. Food-Derived Bioactive Peptides in Human Health: Challenges and Opportunities. Nutrients 2018, 10, 1738.
- 104.
Jacob, M.C.M.; da Silva-Maia, J.K.; Albuquerque, U.P.; et al. Culture matters: A systematic review of antioxidant potential of tree legumes in the semiarid region of Brazil and local processing techniques as a driver of bioaccessibility. PLoS ONE 2022, 17, e0264950.
- 105.
Zhang, Q.; Zheng, L.; Luo, D.; et al. In Vitro Simulated Gastrointestinal Digestion Stability of a Neuroprotective Octapeptide WCPFSRSF and Prediction of Potential Bioactive Peptides in Its Digestive Fragments by Multiple Bioinformatics Tools. J. Agric. Food Chem. 2023, 71, 6987–6998.
- 106.
Pavlicevic, M.; Maestri, E.; Marmiroli, M. Marine Bioactive Peptides-An Overview of Generation, Structure and Application with a Focus on Food Sources. Mar. Drugs 2020, 18, 424.
- 107.
Bhattacharjee, M.J.; Bala, A.; Khan, M.R.; et al. Functional impact of bioactive peptides derived from fermented foods on diverse human populations. Food Chem. 2025, 492, 145416.
- 108.
Cicero, A.F.G.; Fogacci, F.; Colletti, A. Potential role of bioactive peptides in prevention and treatment of chronic diseases: A narrative review. Br. J. Pharmacol. 2017, 174, 1378–1394.
- 109.
Majura, J.J.; Cao, W.; Chen, Z.; et al. The current research status and strategies employed to modify food-derived bioactive peptides. Front. Nutr. 2022, 9, 950823.
- 110.
Vlieghe, P.; Khrestchatisky, M. Peptide-based vectors for blood-brain barrier targeting and delivery of drugs to the central nervous system. Ther. Deliv. 2010, 1, 489–494.
- 111.
Zhang, X.; Tu, D.; Li, S.; et al. A novel synthetic peptide SVHRSP attenuates dopaminergic neurodegeneration by inhibiting NADPH oxidase-mediated neuroinflammation in experimental models of Parkinson’s disease. Free Radic. Biol. Med. 2022, 188, 363–374.
- 112.
Ano, Y.; Kutsukake, T.; Sasaki, T.; et al. Identification of a Novel Peptide from β-Casein That Enhances Spatial and Object Recognition Memory in Mice. J. Agric. Food Chem. 2019, 67, 8160–8167.
- 113.
Shah, S.A.; Lee, H.Y.; Bressan, R.A.; et al. Novel osmotin attenuates glutamate-induced synaptic dysfunction and neurodegeneration via the JNK/PI3K/Akt pathway in postnatal rat brain. Cell Death Dis. 2014, 5, e1026.
- 114.
Zhang, T.L.; Fu, J.L.; Geng, Z.; et al. The neuroprotective effect of losartan through inhibiting AT1/ASK1/MKK4/JNK3 pathway following cerebral I/R in rat hippocampal CA1 region. CNS Neurosci. Ther. 2012, 18, 981–987.
- 115.
Gupta, N.; Quazi, S.; Jha, S.K.; et al. Chickpea Peptide: A Nutraceutical Molecule Corroborating Neurodegenerative and ACE-I Inhibition. Nutrients 2022, 14, 4824.
- 116.
Roy, F.; Boye, J.I.; Simpson, B.K. Bioactive proteins and peptides in pulse crops: Pea, chickpea and lentil. Food Res. Int. 2010, 43, 432–442.
- 117.
Mukhamedov, N.; Asrorov, A.; Yashinov, A.; et al. Synthesis and Characterisation of Chickpea Peptides-Zinc Chelates Having ACE2 Inhibitory Activity. Protein J. 2023, 42, 547–562.
- 118.
Suwanangul, S.; Aluko, R.E.; Sangsawad, P.; et al. Antioxidant and enzyme inhibitory properties of sacha inchi (Plukenetia volubilis) protein hydrolysate and its peptide fractions. J. Food Biochem. 2022, 46, e14464.
- 119.
Yang, P.; Abel-Hamid, M.; Romieh, E.; et al. Effect of peptides synthesized from lactoferrin of buffalo milk on oxidative stress in kunming mice. J. Anim. Plant Sci. 2020, 30, 65–71.
- 120.
Martinez Leo, E.E.; Segura Campos, M.R. Neuroprotective effect from Salvia hispanica peptide fractions on pro-inflammatory modulation of HMC3 microglial cells. J. Food Biochem. 2020, 44, e13207.
- 121.
Abdul-Muneer, P.M.; Bhowmick, S.; Briski, N. Angiotensin II Causes Neuronal Damage in Stretch-Injured Neurons: Protective Effects of Losartan, an Angiotensin T1 Receptor Blocker. Mol. Neurobiol. 2018, 55, 5901–5912.
- 122.
Zhao, Y.T.; Yin, H.; Hu, C.; et al. Tilapia Skin Peptides Ameliorate Cyclophosphamide-Induced Anxiety- and Depression-Like Behavior via Improving Oxidative Stress, Neuroinflammation, Neuron Apoptosis, and Neurogenesis in Mice. Front. Nutr. 2022, 9, 882175.
- 123.
Zhu, W.-Y.; Wang, Y.-M.; Dong, X.-M.; et al. Antioxidant peptides from Antarctic krill (Euphausia superba) hydrolysate: Stability, ACE inhibitory activity, and endothelial cells protection by regulating Keap1/Nrf2 pathway. J. Agric. Food Res. 2025, 20, 101745.
- 124.
Dong, Y.; Hu, Q.; Zhao, L.; et al. A novel neuroprotective peptide YVYAETY identified and screened from Flammulina velutipes protein hydrolysates attenuates scopolamine-induced cognitive impairment in mice. Food Funct. 2024, 15, 6082–6094.
- 125.
Wang, S.; Wu, S.; Yang, G.; et al. A review on the progress, challenges and prospects in commercializing microalgal fucoxanthin. Biotechnol. Adv. 2021, 53, 107865.
- 126.
Bamdad, F.; Shin, S.H.; Suh, J.W.; et al. Anti-Inflammatory and Antioxidant Properties of Casein Hydrolysate Produced Using High Hydrostatic Pressure Combined with Proteolytic Enzymes. Molecules 2017, 22, 609.
- 127.
Haqqani, A.S.; Belanger, K.; Stanimirovic, D.B. Receptor-mediated transcytosis for brain delivery of therapeutics: Receptor classes and criteria. Front. Drug Deliv. 2024, 4, 1360302.
- 128.
Tang, J.; Krushelnycky, L.; Shaqo, A.; et al. A Comprehensive Review of Nutritional Influences on the Serotonergic System. Adv. Nutr. 2025, 16, 100524.
- 129.
Liu, Z.; Udenigwe, C.C. Role of food-derived opioid peptides in the central nervous and gastrointestinal systems. J. Food Biochem. 2019, 43, e12629.
- 130.
Sakaguchi, M.; Koseki, M.; Wakamatsu, M.; et al. Effects of systemic administration of β-casomorphin-5 on learning and memory in mice. Eur. J. Pharmacol. 2006, 530, 81–87.
- 131.
Dang, Q.; Wu, D.; Li, Y.; et al. Walnut-derived peptides ameliorate d-galactose-induced memory impairments in a mouse model via inhibition of MMP-9-mediated blood-brain barrier disruption. Food Res. Int. 2022, 162, 112029.
- 132.
Chataigner, M.; Mortessagne, P.; Lucas, C.; et al. Dietary fish hydrolysate supplementation containing n-3 LC-PUFAs and peptides prevents short-term memory and stress response deficits in aged mice. Brain Behav. Immun. 2021, 91, 716–730.
- 133.
Diez-Vives, C.; Gay, M.; Garcia-Matas, S.; et al. Proteomic study of neuron and astrocyte cultures from senescence-accelerated mouse SAMP8 reveals degenerative changes. J. Neurochem. 2009, 111, 945–955.
- 134.
Furukawa, A.; Oikawa, S.; Hasegawa-Ishii, S.; et al. Proteomic analysis of aging brain in SAMP10 mouse: A model of age-related cerebral degeneration. Mech. Ageing Dev. 2010, 131, 379–388.
- 135.
Seino, Y.; Kawarabayashi, T.; Wakasaya, Y.; et al. Amyloid β accelerates phosphorylation of tau and neurofibrillary tangle formation in an amyloid precursor protein and tau double-transgenic mouse model. J. Neurosci. Res. 2010, 88, 3547–3554.
- 136.
Li, Y.; Qiang, X.; Luo, L.; et al. Aurone Mannich base derivatives as promising multifunctional agents with acetylcholinesterase inhibition, anti-β-amyloid aggragation and neuroprotective properties for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2017, 126, 762–775.
- 137.
Lawlor, P.A.; Young, D. Aβ Infusion and Related Models of Alzheimer Dementia. In Animal Models of Dementia; Humana Press: Totowa, NJ, USA, 2011; pp. 347–370.
- 138.
Liu, L.; Li, S.; Zheng, J.; et al. Safety considerations on food protein-derived bioactive peptides. Trends Food Sci. Technol. 2020, 96, 199–207.
- 139.
Katayama, S.; Imai, R.; Sugiyama, H.; et al. Oral administration of soy peptides suppresses cognitive decline by induction of neurotrophic factors in SAMP8 mice. J. Agric. Food Chem. 2014, 62, 3563–3569.
- 140.
Feng, L.; Wang, X.; Peng, F.; et al. Walnut Protein Hydrolysates Play a Protective Role on Neurotoxicity Induced by d-Galactose and Aluminum Chloride in Mice. Molecules 2018, 23, 2308.
- 141.
Wu, D.; Li, M.; Ding, J.; et al. Structure-activity relationship and pathway of antioxidant shrimp peptides in a PC12 cell model. J. Funct. Foods 2020, 70, 103978.
- 142.
Zhao, Y.; Lu, Z.; Xu, X.; et al. Sea Cucumber-Derived Peptide Attenuates Scopolamine-Induced Cognitive Impairment by Preventing Hippocampal Cholinergic Dysfunction and Neuronal Cell Death. J. Agric. Food Chem. 2022, 70, 567–576.
- 143.
Ren, D.; Zhao, F.; Liu, C.; et al. Antioxidant hydrolyzed peptides from Manchurian walnut (Juglans mandshurica Maxim.) attenuate scopolamine-induced memory impairment in mice. J. Sci. Food Agric. 2018, 98, 5142–5152.
- 144.
Zou, J.; Cai, P.S.; Xiong, C.M.; et al. Neuroprotective effect of peptides extracted from walnut (Juglans sigilata Dode) proteins on Aβ25-35-induced memory impairment in mice. J. Huazhong Univ. Sci. Technol. Med. Sci. 2016, 36, 21–30.

This work is licensed under a Creative Commons Attribution 4.0 International License.


