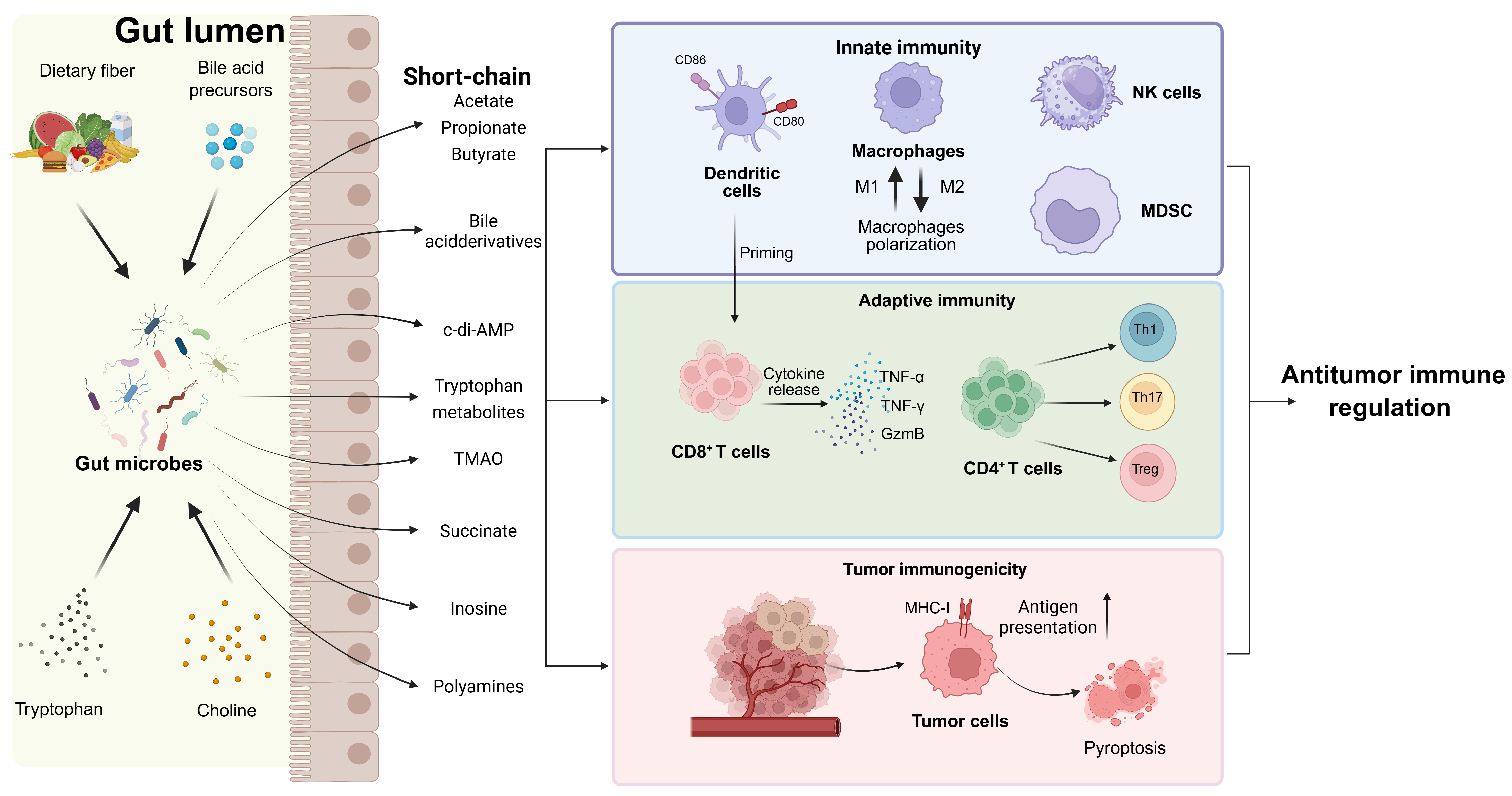
Gut microbiota-derived metabolites critically modulate the efficacy of tumor immunotherapy, particularly immune checkpoint inhibitors, by orchestrating key immunological mechanisms within the tumor immune microenvironment. Through multifaceted pathways, they dynamically regulate both innate and adaptive immunity or reshape tumor immunogenicity, thereby maintaining the delicate equilibrium between antitumor immune activation and suppression. Moreover, some microbial metabolites (e.g., butyrate, polyamines, succinic acid) have paradoxical or dual functions depending on the context (e.g., cancer type). A comprehensive understanding of the complex interplay among microbiota, metabolism, and immunity is important for clarifying individual variations in immunotherapeutic outcomes and may help inform strategies to overcome resistance. Current microbiota-based therapies, including probiotics, genetically engineered bacteria, and fecal microbiota transplantation, as well as interventions targeting metabolic pathways, are emerging as promising strategies to enhance immunotherapy by modulating host metabolic processes. However, several major challenges hinder clinical translation, including the bidirectional effects and concentration-dependent activity of metabolites, issues in delivery efficiency, and significant inter-individual heterogeneity. This review aims to systematically summarize the main mechanisms by which gut microbial metabolites regulate antitumor immunity and to explore the current landscape, strategies, and obstacles in their clinical application. Overall, it may provide a theoretical framework and practical perspectives for the future development of personalized tumor immunotherapies based on microbiome and metabolic interventions.
- Open Access
- Review
Interplay between Gut Microbiome, Metabolites, and Tumor Immunity: Mechanisms and Clinical Translation
- Zhen Zhang 1,2,3,4,
- Yongchao Gao 1,2,3,4,
- Fengyan Yuan 1,2,3,4,
- Quanlin Wang 1,2,3,4,
- Wei Zhang 1,2,3,4,*,
- Rong Liu 1,2,3,4,*
Author Information
Received: 06 Aug 2025 | Revised: 10 Oct 2025 | Accepted: 05 Nov 2025 | Published: 20 Apr 2026
Abstract
Graphical Abstract
Keywords
gut microbial metabolites | immunity | cancer immunotherapy | immune microenvironment | metabolic interventions
References
- 1.
Couzin-Frankel, J. Breakthrough of the year 2013. Cancer immunotherapy. Science 2013, 342, 1432–1433. https://doi.org/10.1126/science.342.6165.1432.
- 2.
Wang, D.R.; Wu, X.L.; Sun, Y.L. Therapeutic targets and biomarkers of tumor immunotherapy: Response versus non-response. Signal Transduct. Target. Ther. 2022, 7, 331. https://doi.org/10.1038/s41392-022-01136-2.
- 3.
Bagchi, S.; Yuan, R.; Engleman, E.G. Immune Checkpoint Inhibitors for the Treatment of Cancer: Clinical Impact and Mechanisms of Response and Resistance. Annu. Rev. Pathol. Mech. Dis. 2021, 16, 223–249. https://doi.org/10.1146/annurev-pathol-042020-042741.
- 4.
Morad, G.; Helmink, B.A.; Sharma, P.; Wargo, J.A. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell 2021, 184, 5309–5337. https://doi.org/10.1016/j.cell.2021.09.020.
- 5.
Sorysz, Z.; Kowalewski, P.; Walędziak, M.; Różańska-Walędziak, A. Do Gut Microbiomes Shift After Bariatric Surgery? A Literature Review. Medicina 2025, 61, 849. https://doi.org/10.3390/medicina61050849.
- 6.
Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. https://doi.org/10.1038/nature08821.
- 7.
Zimmermann, M.; Zimmermann-Kogadeeva, M.; Wegmann, R.; Goodman, A.L. Mapping human microbiome drug metabolism by gut bacteria and their genes. Nature 2019, 570, 462–467. https://doi.org/10.1038/s41586-019-1291-3.
- 8.
Clemente, J.C.; Ursell, L.K.; Parfrey, L.W.; Knight, R. The impact of the gut microbiota on human health: An integrative view. Cell 2012, 148, 1258–1270. https://doi.org/10.1016/j.cell.2012.01.035.
- 9.
de Vos, W.M.; Tilg, H.; Van Hul, M.; Cani, P.D. Gut microbiome and health: Mechanistic insights. Gut 2022, 71, 1020–1032. https://doi.org/10.1136/gutjnl-2021-326789.
- 10.
Gopalakrishnan, V.; Spencer, C.N.; Nezi, L.; Reuben, A.; Andrews, M.C.; Karpinets, T.V.; Prieto, P.A.; Vicente, D.; Hoffman, K.; Wei, S.C.; et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 2018, 359, 97–103. https://doi.org/10.1126/science.aan4236.
- 11.
Matson, V.; Fessler, J.; Bao, R.; Chongsuwat, T.; Zha, Y.; Alegre, M.L.; Luke, J.J.; Gajewski, T.F. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science 2018, 359, 104–108. https://doi.org/10.1126/science.aao3290.
- 12.
Routy, B.; Le Chatelier, E.; Derosa, L.; Duong, C.P.M.; Alou, M.T.; Daillère, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P.; et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 2018, 359, 91–97. https://doi.org/10.1126/science.aan3706.
- 13.
Vétizou, M.; Pitt, J.M.; Daillère, R.; Lepage, P.; Waldschmitt, N.; Flament, C.; Rusakiewicz, S.; Routy, B.; Roberti, M.P.; Duong, C.P.; et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 2015, 350, 1079–1084. https://doi.org/10.1126/science.aad1329.
- 14.
Nomura, M.; Nagatomo, R.; Doi, K.; Shimizu, J.; Baba, K.; Saito, T.; Matsumoto, S.; Inoue, K.; Muto, M. Association of Short-Chain Fatty Acids in the Gut Microbiome With Clinical Response to Treatment With Nivolumab or Pembrolizumab in Patients With Solid Cancer Tumors. JAMA Netw Open 2020, 3, e202895. https://doi.org/10.1001/jamanetworkopen.2020.2895.
- 15.
Bender, M.J.; McPherson, A.C.; Phelps, C.M.; Pandey, S.P.; Laughlin, C.R.; Shapira, J.H.; Medina Sanchez, L.; Rana, M.; Richie, T.G.; Mims, T.S.; et al. Dietary tryptophan metabolite released by intratumoral Lactobacillus reuteri facilitates immune checkpoint inhibitor treatment. Cell 2023, 186, 1846–1862.e1826. https://doi.org/10.1016/j.cell.2023.03.011.
- 16.
Davar, D.; Dzutsev, A.K.; McCulloch, J.A.; Rodrigues, R.R.; Chauvin, J.M.; Morrison, R.M.; Deblasio, R.N.; Menna, C.; Ding, Q.; Pagliano, O.; et al. Fecal microbiota transplant overcomes resistance to anti-PD-1 therapy in melanoma patients. Science 2021, 371, 595–602. https://doi.org/10.1126/science.abf3363.
- 17.
Li, D.; Lan, X.; Xu, L.; Zhou, S.; Luo, H.; Zhang, X.; Yu, W.; Yang, Y.; Fang, X. Influence of gut microbial metabolites on tumor immunotherapy: Mechanisms and potential natural products. Front Immunol 2025, 16, 1552010. https://doi.org/10.3389/fimmu.2025.1552010.
- 18.
Thulasinathan, B.; Suvilesh, K.N.; Maram, S.; Grossmann, E.; Ghouri, Y.; Teixeiro, E.P.; Chan, J.; Kaif, J.T.; Rachagani, S. The impact of gut microbial short-chain fatty acids on colorectal cancer development and prevention. Gut Microbes 2025, 17, 2483780. https://doi.org/10.1080/19490976.2025.2483780.
- 19.
Tremaroli, V.; Bäckhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 2012, 489, 242–249. https://doi.org/10.1038/nature11552.
- 20.
Du, A.; Wang, Z.; Huang, T.; Xue, S.; Jiang, C.; Qiu, G.; Yuan, K. Fatty acids in cancer: Metabolic functions and potential treatment. MedComm Oncol. 2023, 2, e25. https://doi.org/10.1002/mog2.25.
- 21.
Baba, Y.; Tsuge, D.; Aoki, R. Enhancement of carbohydrate metabolism by probiotic and prebiotic intake promotes short-chain fatty acid production in the gut microbiome: A randomized, double-blind, placebo-controlled crossover trial. Biosci. Biotechnol. Biochem. 2025, 89, 1191–1202. https://doi.org/10.1093/bbb/zbaf071.
- 22.
Arab, J.P.; Karpen, S.J.; Dawson, P.A.; Arrese, M.; Trauner, M. Bile acids and nonalcoholic fatty liver disease: Molecular insights and therapeutic perspectives. Hepatology 2017, 65, 350–362. https://doi.org/10.1002/hep.28709.
- 23.
Yang, W.; Cong, Y. Gut microbiota-derived metabolites in the regulation of host immune responses and immune-related inflammatory diseases. Cell. Mol. Immunol. 2021, 18, 866–877. https://doi.org/10.1038/s41423-021-00661-4.
- 24.
Nie, Q.; Sun, Y.; Li, M.; Zuo, S.; Chen, C.; Lin, Q.; Nie, S. Targeted modification of gut microbiota and related metabolites via dietary fiber. Carbohydr. Polym. 2023, 316, 120986. https://doi.org/10.1016/j.carbpol.2023.120986.
- 25.
Zhou, Y.; Zhang, Y.; Jin, S.; Lv, J.; Li, M.; Feng, N. The gut microbiota derived metabolite trimethylamine N-oxide: Its important role in cancer and other diseases. Biomed. Pharmacother. 2024, 177, 117031. https://doi.org/10.1016/j.biopha.2024.117031.
- 26.
Goodwin, A.C.; Destefano Shields, C.E.; Wu, S.; Huso, D.L.; Wu, X.; Murray-Stewart, T.R.; Hacker-Prietz, A.; Rabizadeh, S.; Woster, P.M.; Sears, C.L.; et al. Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis-induced colon tumorigenesis. Proc. Natl. Acad. Sci. USA 2011, 108, 15354–15359. https://doi.org/10.1073/pnas.1010203108.
- 27.
Imana, Z.N.; Tseng, J.C.; Yang, J.X.; Liu, Y.L.; Lin, P.Y.; Huang, M.H.; Chen, L.; Luo, Y.; Wang, C.C.; Yu, G.Y.; et al. Cooperative tumor inhibition by CpG-oligodeoxynucleotide and cyclic dinucleotide in head and neck cancer involves T helper cytokine and macrophage phenotype reprogramming. Biomed. Pharmacother. 2024, 181, 117692. https://doi.org/10.1016/j.biopha.2024.117692.
- 28.
Jiang, S.S.; Xie, Y.L.; Xiao, X.Y.; Kang, Z.R.; Lin, X.L.; Zhang, L.; Li, C.S.; Qian, Y.; Xu, P.P.; Leng, X.X.; et al. Fusobacterium nucleatum-derived succinic acid induces tumor resistance to immunotherapy in colorectal cancer. Cell Host Microbe 2023, 31, 781–797.e789. https://doi.org/10.1016/j.chom.2023.04.010.
- 29.
Zhang, Q.; Zhao, Q.; Li, T.; Lu, L.; Wang, F.; Zhang, H.; Liu, Z.; Ma, H.; Zhu, Q.; Wang, J.; et al. Lactobacillus plantarum-derived indole-3-lactic acid ameliorates colorectal tumorigenesis via epigenetic regulation of CD8+ T cell immunity. Cell Metab. 2023, 35, 943–960.e949. https://doi.org/10.1016/j.cmet.2023.04.015.
- 30.
Inamoto, T.; Furuta, K.; Han, C.; Uneme, M.; Kano, T.; Ishikawa, K.; Kaito, C. Short-chain fatty acids stimulate dendrite elongation in dendritic cells by inhibiting histone deacetylase. FEBS J. 2023, 290, 5794–5810. https://doi.org/10.1111/febs.16945.
- 31.
Coutzac, C.; Jouniaux, J.M.; Paci, A.; Schmidt, J.; Mallardo, D.; Seck, A.; Asvatourian, V.; Cassard, L.; Saulnier, P.; Lacroix, L.; et al. Systemic short chain fatty acids limit antitumor effect of CTLA-4 blockade in hosts with cancer. Nat. Commun. 2020, 11, 2168. https://doi.org/10.1038/s41467-020-16079-x.
- 32.
Wu, Z.; He, J.; Zhang, Z.; Li, J.; Zou, H.; Tan, X.; Wang, Y.; Yao, Y.; Xiong, W. Propionic Acid Driven by the Lactobacillus johnsonii Culture Supernatant Alleviates Colitis by Inhibiting M1 Macrophage Polarization by Modulating the MAPK Pathway in Mice. J. Agric. Food Chem. 2023, 71, 14951–14966. https://doi.org/10.1021/acs.jafc.3c00278.
- 33.
Kes, M.M.G.; Van den Bossche, J.; Griffioen, A.W.; Huijbers, E.J.M. Oncometabolites lactate and succinate drive pro-angiogenic macrophage response in tumors. Biochim. Biophys. Acta Rev. Cancer 2020, 1874, 188427. https://doi.org/10.1016/j.bbcan.2020.188427.
- 34.
Shan, X.; Hu, P.; Ni, L.; Shen, L.; Zhang, Y.; Ji, Z.; Cui, Y.; Guo, M.; Wang, H.; Ran, L.; et al. Serine metabolism orchestrates macrophage polarization by regulating the IGF1-p38 axis. Cell. Mol. Immunol. 2022, 19, 1263–1278. https://doi.org/10.1038/s41423-022-00925-7.
- 35.
Jiang, Y.; Li, X.; Qian, F.; Sun, B.; Wang, X.; Zhang, Y.; Zhang, D.; Geng, M.; Xie, Z.; Yang, S. Fine-tuning Bacterial Cyclic di-AMP Production for Durable Antitumor Effects Through the Activation of the STING Pathway. Research 2023, 6, 0102. https://doi.org/10.34133/research.0102.
- 36.
Li, Z.; Zhang, Y.; Hong, W.; Wang, B.; Chen, Y.; Yang, P.; Zhou, J.; Fan, J.; Zeng, Z.; Du, S. Gut microbiota modulate radiotherapy-associated antitumor immune responses against hepatocellular carcinoma Via STING signaling. Gut Microbes 2022, 14, 2119055. https://doi.org/10.1080/19490976.2022.2119055.
- 37.
Liu, N.; Chen, L.; Yan, M.; Tao, Q.; Wu, J.; Chen, J.; Chen, X.; Zhang, W.; Peng, C. Eubacterium rectale Improves the Efficacy of Anti-PD1 Immunotherapy in Melanoma via l-Serine-Mediated NK Cell Activation. Research 2023, 6, 0127. https://doi.org/10.34133/research.0127.
- 38.
Wei, H.; Suo, C.; Gu, X.; Shen, S.; Lin, K.; Zhu, C.; Yan, K.; Bian, Z.; Chen, L.; Zhang, T.; et al. AKR1D1 suppresses liver cancer progression by promoting bile acid metabolism-mediated NK cell cytotoxicity. Cell Metab. 2025, 37, 1103–1118.e1107. https://doi.org/10.1016/j.cmet.2025.01.011.
- 39.
Tian, P.; Yang, W.; Guo, X.; Wang, T.; Tan, S.; Sun, R.; Xiao, R.; Wang, Y.; Jiao, D.; Xu, Y.; et al. Early life gut microbiota sustains liver-resident natural killer cells maturation via the butyrate-IL-18 axis. Nat. Commun. 2023, 14, 1710. https://doi.org/10.1038/s41467-023-37419-7.
- 40.
He, Y.; Fu, L.; Li, Y.; Wang, W.; Gong, M.; Zhang, J.; Dong, X.; Huang, J.; Wang, Q.; Mackay, C.R.; et al. Gut microbial metabolites facilitate anticancer therapy efficacy by modulating cytotoxic CD8+ T cell immunity. Cell Metab. 2021, 33, 988–1000.e1007. https://doi.org/10.1016/j.cmet.2021.03.002.
- 41.
Wang, X.; Fang, Y.; Liang, W.; Wong, C.C.; Qin, H.; Gao, Y.; Liang, M.; Song, L.; Zhang, Y.; Fan, M.; et al. Fusobacterium nucleatum facilitates anti-PD-1 therapy in microsatellite stable colorectal cancer. Cancer Cell 2024, 42, 1729–1746.e1728. https://doi.org/10.1016/j.ccell.2024.08.019.
- 42.
Wang, H.; Rong, X.; Zhao, G.; Zhou, Y.; Xiao, Y.; Ma, D.; Jin, X.; Wu, Y.; Yan, Y.; Yang, H.; et al. The microbial metabolite trimethylamine N-oxide promotes antitumor immunity in triple-negative breast cancer. Cell Metab. 2022, 34, 581–594.e588. https://doi.org/10.1016/j.cmet.2022.02.010.
- 43.
Cong, J.; Liu, P.; Han, Z.; Ying, W.; Li, C.; Yang, Y.; Wang, S.; Yang, J.; Cao, F.; Shen, J.; et al. Bile acids modified by the intestinal microbiota promote colorectal cancer growth by suppressing CD8+ T cell effector functions. Immunity 2024, 57, 876–889.e811. https://doi.org/10.1016/j.immuni.2024.02.014.
- 44.
Jia, D.; Wang, Q.; Qi, Y.; Jiang, Y.; He, J.; Lin, Y.; Sun, Y.; Xu, J.; Chen, W.; Fan, L.; et al. Microbial metabolite enhances immunotherapy efficacy by modulating T cell stemness in pan-cancer. Cell 2024, 187, 1651–1665.e1621. https://doi.org/10.1016/j.cell.2024.02.022.
- 45.
Zhang, S.L.; Wang, X.; Cai, Q.Q.; Chen, C.; Zhang, Z.Y.; Xu, Y.Y.; Yang, M.X.; Jia, Q.A.; Wang, Y.; Wang, Z.M. Acarbose enhances the efficacy of immunotherapy against solid tumours by modulating the gut microbiota. Nat. Metab. 2024, 6, 1991–2009. https://doi.org/10.1038/s42255-024-01137-1.
- 46.
Campbell, C.; McKenney, P.T.; Konstantinovsky, D.; Isaeva, O.I.; Schizas, M.; Verter, J.; Mai, C.; Jin, W.B.; Guo, C.J.; Violante, S.; et al. Bacterial metabolism of bile acids promotes generation of peripheral regulatory T cells. Nature 2020, 581, 475–479. https://doi.org/10.1038/s41586-020-2193-0.
- 47.
Fiorucci, S.; Marchianò, S.; Distrutti, E.; Biagioli, M. Bile acids and their receptors in hepatic immunity. Liver Res. 2025, 9, 1–16. https://doi.org/10.1016/j.livres.2025.01.005.
- 48.
Solvay, M.; Holfelder, P.; Klaessens, S.; Pilotte, L.; Stroobant, V.; Lamy, J.; Naulaerts, S.; Spillier, Q.; Frédérick, R.; De Plaen, E.; et al. Tryptophan depletion sensitizes the AHR pathway by increasing AHR expression and GCN2/LAT1-mediated kynurenine uptake, and potentiates induction of regulatory T lymphocytes. J. Immunother. Cancer 2023, 11, e006728. https://doi.org/10.1136/jitc-2023-006728.
- 49.
Campesato, L.F.; Budhu, S.; Tchaicha, J.; Weng, C.H.; Gigoux, M.; Cohen, I.J.; Redmond, D.; Mangarin, L.; Pourpe, S.; Liu, C.; et al. Blockade of the AHR restricts a Treg-macrophage suppressive axis induced by L-Kynurenine. Nat. Commun. 2020, 11, 4011. https://doi.org/10.1038/s41467-020-17750-z.
- 50.
Zhang, H.; Wang, J.; Shen, J.; Chen, S.; Yuan, H.; Zhang, X.; Liu, X.; Yu, Y.; Li, X.; Gao, Z.; et al. Prophylactic supplementation with Bifidobacterium infantis or its metabolite inosine attenuates cardiac ischemia/reperfusion injury. Imeta 2024, 3, e220. https://doi.org/10.1002/imt2.220.
- 51.
Mager, L.F.; Burkhard, R.; Pett, N.; Cooke, N.C.A.; Brown, K.; Ramay, H.; Paik, S.; Stagg, J.; Groves, R.A.; Gallo, M.; et al. Microbiome-derived inosine modulates response to checkpoint inhibitor immunotherapy. Science 2020, 369, 1481–1489. https://doi.org/10.1126/science.abc3421.
- 52.
Zhou, J.; Zhao, L.; Liu, L.; He, L.; Chen, Y.; Wang, F.; Cui, D.; Wang, L.; Zhou, Q. The Emerging Mechanisms and Therapeutic Potentials of Dendritic Cells in NSCLC. J. Inflamm. Res. 2025, 18, 5061–5076. https://doi.org/10.2147/jir.S506644.
- 53.
Lee, S.Y.; Jhun, J.; Woo, J.S.; Lee, K.H.; Hwang, S.H.; Moon, J.; Park, G.; Choi, S.S.; Kim, S.J.; Jung, Y.J.; et al. Gut microbiome-derived butyrate inhibits the immunosuppressive factors PD-L1 and IL-10 in tumor-associated macrophages in gastric cancer. Gut Microbes 2024, 16, 2300846. https://doi.org/10.1080/19490976.2023.2300846.
- 54.
Lavoie, S.; Chun, E.; Bae, S.; Brennan, C.A.; Gallini Comeau, C.A.; Lang, J.K.; Michaud, M.; Hoveyda, H.R.; Fraser, G.L.; Fuller, M.H.; et al. Expression of Free Fatty Acid Receptor 2 by Dendritic Cells Prevents Their Expression of Interleukin 27 and Is Required for Maintenance of Mucosal Barrier and Immune Response Against Colorectal Tumors in Mice. Gastroenterology 2020, 158, 1359–1372.e1359. https://doi.org/10.1053/j.gastro.2019.12.027.
- 55.
Guan, F.; Wang, R.; Yi, Z.; Luo, P.; Liu, W.; Xie, Y.; Liu, Z.; Xia, Z.; Zhang, H.; Cheng, Q. Tissue macrophages: Origin, heterogenity, biological functions, diseases and therapeutic targets. Signal Transduct. Target. Ther. 2025, 10, 93. https://doi.org/10.1038/s41392-025-02124-y.
- 56.
Qiu, Y.; Chen, T.; Hu, R.; Zhu, R.; Li, C.; Ruan, Y.; Xie, X.; Li, Y. Next frontier in tumor immunotherapy: Macrophage-mediated immune evasion. Biomark. Res. 2021, 9, 72. https://doi.org/10.1186/s40364-021-00327-3.
- 57.
Zhang, W.; Liu, H.T. MAPK signal pathways in the regulation of cell proliferation in mammalian cells. Cell Res. 2002, 12, 9–18. https://doi.org/10.1038/sj.cr.7290105.
- 58.
Kwon, J.; Bakhoum, S.F. The Cytosolic DNA-Sensing cGAS-STING Pathway in Cancer. Cancer Discov. 2020, 10, 26–39. https://doi.org/10.1158/2159-8290.Cd-19-0761.
- 59.
Kuo, C.C.; Wu, J.Y.; Wu, K.K. Cancer-derived extracellular succinate: A driver of cancer metastasis. J. Biomed. Sci. 2022, 29, 93. https://doi.org/10.1186/s12929-022-00878-z.
- 60.
Mirji, G.; Worth, A.; Bhat, S.A.; El Sayed, M.; Kannan, T.; Goldman, A.R.; Tang, H.Y.; Liu, Q.; Auslander, N.; Dang, C.V.; et al. The microbiome-derived metabolite TMAO drives immune activation and boosts responses to immune checkpoint blockade in pancreatic cancer. Sci. Immunol. 2022, 7, eabn0704. https://doi.org/10.1126/sciimmunol.abn0704.
- 61.
Yurdagul, A., Jr.; Kong, N.; Gerlach, B.D.; Wang, X.; Ampomah, P.; Kuriakose, G.; Tao, W.; Shi, J.; Tabas, I. ODC (Ornithine Decarboxylase)-Dependent Putrescine Synthesis Maintains MerTK (MER Tyrosine-Protein Kinase) Expression to Drive Resolution. Arterioscler. Thromb. Vasc. Biol. 2021, 41, e144–e159. https://doi.org/10.1161/atvbaha.120.315622.
- 62.
Myers, J.A.; Miller, J.S. Exploring the NK cell platform for cancer immunotherapy. Nat Rev Clin Oncol 2021, 18, 85–100. https://doi.org/10.1038/s41571-020-0426-7.
- 63.
Fares, J.; Davis, Z.B.; Rechberger, J.S.; Toll, S.A.; Schwartz, J.D.; Daniels, D.J.; Miller, J.S.; Khatua, S. Advances in NK cell therapy for brain tumors. NPJ Precis. Oncol. 2023, 7, 17. https://doi.org/10.1038/s41698-023-00356-1.
- 64.
Gungabeesoon, J.; Gort-Freitas, N.A.; Kiss, M.; Bolli, E.; Messemaker, M.; Siwicki, M.; Hicham, M.; Bill, R.; Koch, P.; Cianciaruso, C.; et al. A neutrophil response linked to tumor control in immunotherapy. Cell 2023, 186, 1448–1464.e1420. https://doi.org/10.1016/j.cell.2023.02.032.
- 65.
Yao, J.; Ji, L.; Wang, G.; Ding, J. Effect of neutrophils on tumor immunity and immunotherapy resistance with underlying mechanisms. Cancer Commun. 2025, 45, 15–42. https://doi.org/10.1002/cac2.12613.
- 66.
Li, G.; Lin, J.; Zhang, C.; Gao, H.; Lu, H.; Gao, X.; Zhu, R.; Li, Z.; Li, M.; Liu, Z. Microbiota metabolite butyrate constrains neutrophil functions and ameliorates mucosal inflammation in inflammatory bowel disease. Gut Microbes 2021, 13, 1968257. https://doi.org/10.1080/19490976.2021.1968257.
- 67.
Park, S.Y.; Pylaeva, E.; Bhuria, V.; Gambardella, A.R.; Schiavoni, G.; Mougiakakos, D.; Kim, S.H.; Jablonska, J. Harnessing myeloid cells in cancer. Mol. Cancer 2025, 24, 69. https://doi.org/10.1186/s12943-025-02249-2.
- 68.
Salman, S.; Meyers, D.J.; Wicks, E.E.; Lee, S.N.; Datan, E.; Thomas, A.M.; Anders, N.M.; Hwang, Y.; Lyu, Y.; Yang, Y.; et al. HIF inhibitor 32-134D eradicates murine hepatocellular carcinoma in combination with anti-PD1 therapy. J. Clin. Invest. 2022, 132. https://doi.org/10.1172/jci156774.
- 69.
Martens, A.; Wistuba-Hamprecht, K.; Geukes Foppen, M.; Yuan, J.; Postow, M.A.; Wong, P.; Romano, E.; Khammari, A.; Dreno, B.; Capone, M.; et al. Baseline Peripheral Blood Biomarkers Associated with Clinical Outcome of Advanced Melanoma Patients Treated with Ipilimumab. Clin. Cancer Res. 2016, 22, 2908–2918. https://doi.org/10.1158/1078-0432.Ccr-15-2412.
- 70.
Wang, R.; Li, B.; Huang, B.; Li, Y.; Liu, Q.; Lyu, Z.; Chen, R.; Qian, Q.; Liang, X.; Pu, X.; et al. Gut Microbiota-Derived Butyrate Induces Epigenetic and Metabolic Reprogramming in Myeloid-Derived Suppressor Cells to Alleviate Primary Biliary Cholangitis. Gastroenterology 2024, 167, 733–749.e733. https://doi.org/10.1053/j.gastro.2024.05.014.
- 71.
Chen, Y.; Yu, D.; Qian, H.; Shi, Y.; Tao, Z. CD8+ T cell-based cancer immunotherapy. J. Transl. Med. 2024, 22, 394. https://doi.org/10.1186/s12967-024-05134-6.
- 72.
Ma, K.; Xu, Y.; Cheng, H.; Tang, K.; Ma, J.; Huang, B. T cell-based cancer immunotherapy: Opportunities and challenges. Sci. Bull. 2025, 70, 1872–1890. https://doi.org/10.1016/j.scib.2025.03.054.
- 73.
Kang, X.; Liu, C.; Ding, Y.; Ni, Y.; Ji, F.; Lau, H.C.H.; Jiang, L.; Sung, J.J.; Wong, S.H.; Yu, J. Roseburia intestinalis generated butyrate boosts anti-PD-1 efficacy in colorectal cancer by activating cytotoxic CD8+ T cells. Gut 2023, 72, 2112–2122. https://doi.org/10.1136/gutjnl-2023-330291.
- 74.
Andrusaite, A.; Lewis, J.; Frede, A.; Farthing, A.; Kästele, V.; Montgomery, J.; Mowat, A.; Mann, E.; Milling, S. Microbiota-derived butyrate inhibits cDC development via HDAC inhibition, diminishing their ability to prime T cells. Mucosal. Immunol. 2024, 17, 1199–1211. https://doi.org/10.1016/j.mucimm.2024.08.003.
- 75.
Wang, W.; Dernst, A.; Martin, B.; Lorenzi, L.; Cadefau-Fabregat, M.; Phulphagar, K.; Wagener, A.; Budden, C.; Stair, N.; Wagner, T.; et al. Butyrate and propionate are microbial danger signals that activate the NLRP3 inflammasome in human macrophages upon TLR stimulation. Cell Rep. 2024, 43, 114736. https://doi.org/10.1016/j.celrep.2024.114736.
- 76.
Wang, J.; Yang, Y.; Shao, F.; Meng, Y.; Guo, D.; He, J.; Lu, Z. Acetate reprogrammes tumour metabolism and promotes PD-L1 expression and immune evasion by upregulating c-Myc. Nat. Metab. 2024, 6, 914–932. https://doi.org/10.1038/s42255-024-01037-4.
- 77.
Zhou, C.; Basnet, R.; Zhen, C.; Ma, S.; Guo, X.; Wang, Z.; Yuan, Y. Trimethylamine N-oxide promotes the proliferation and migration of hepatocellular carcinoma cell through the MAPK pathway. Discov. Oncol. 2024, 15, 346. https://doi.org/10.1007/s12672-024-01178-8.
- 78.
Schlichtner, S.; Yasinska, I.M.; Klenova, E.; Abooali, M.; Lall, G.S.; Berger, S.M.; Ruggiero, S.; Cholewa, D.; Milošević, M.; Gibbs, B.F.; et al. L-Kynurenine participates in cancer immune evasion by downregulating hypoxic signaling in T lymphocytes. Oncoimmunology 2023, 12, 2244330. https://doi.org/10.1080/2162402x.2023.2244330.
- 79.
Wu, Z.Y.; Wu, Q.W.; Han, Y.; Xiang, S.J.; Wang, Y.N.; Wu, W.W.; Chen, Y.X.; Feng, Z.Q.; Wang, Y.Y.; Xu, Z.G.; et al. Alistipes finegoldii augments the efficacy of immunotherapy against solid tumors. Cancer Cell 2025, 43, 1714–1730.e1712. https://doi.org/10.1016/j.ccell.2025.07.002.
- 80.
Paik, D.; Yao, L.; Zhang, Y.; Bae, S.; D’Agostino, G.D.; Zhang, M.; Kim, E.; Franzosa, E.A.; Avila-Pacheco, J.; Bisanz, J.E.; et al. Human gut bacteria produce Τ(Η)17-modulating bile acid metabolites. Nature 2022, 603, 907–912. https://doi.org/10.1038/s41586-022-04480-z.
- 81.
Urbani, G.; Rondini, E.; Distrutti, E.; Marchianò, S.; Biagioli, M.; Fiorucci, S. Phenotyping the Chemical Communications of the Intestinal Microbiota and the Host: Secondary Bile Acids as Postbiotics. Cells 2025, 14, 595. https://doi.org/10.3390/cells14080595.
- 82.
Zhang, X.; Liu, X.; Zhou, W.; Du, Q.; Yang, M.; Ding, Y.; Hu, R. Blockade of IDO-Kynurenine-AhR Axis Ameliorated Colitis-Associated Colon Cancer via Inhibiting Immune Tolerance. Cell. Mol. Gastroenterol. Hepatol. 2021, 12, 1179–1199. https://doi.org/10.1016/j.jcmgh.2021.05.018.
- 83.
Yang, M.; Cao, M.; Zhang, X.; Fu, B.; Chen, Y.; Tan, Y.; Xuan, C.; Su, Y.; Tan, D.; Hu, R. IDO1 inhibitors are synergistic with CXCL10 agonists in inhibiting colon cancer growth. Biomed. Pharmacother. 2024, 179, 117412. https://doi.org/10.1016/j.biopha.2024.117412.
- 84.
Wei, L.; Pan, Y.; Guo, Y.; Zhu, Y.; Jin, H.; Gu, Y.; Li, C.; Wang, Y.; Lin, J.; Chen, Y.; et al. Symbiotic combination of Akkermansia muciniphila and inosine alleviates alcohol-induced liver injury by modulating gut dysbiosis and immune responses. Front. Microbiol. 2024, 15, 1355225. https://doi.org/10.3389/fmicb.2024.1355225.
- 85.
Wang, T.; Gnanaprakasam, J.N.R.; Chen, X.; Kang, S.; Xu, X.; Sun, H.; Liu, L.; Rodgers, H.; Miller, E.; Cassel, T.A.; et al. Inosine is an alternative carbon source for CD8+-T-cell function under glucose restriction. Nat. Metab. 2020, 2, 635–647. https://doi.org/10.1038/s42255-020-0219-4.
- 86.
Zhao, H.; Zhang, W.; Lu, Y.; Dong, Y.; He, Z.; Zhen, H.; Li, Q. Inosine enhances the efficacy of immune-checkpoint inhibitors in advanced solid tumors: A randomized, controlled, Phase 2 study. Cancer Med. 2024, 13, e70143. https://doi.org/10.1002/cam4.70143.
- 87.
Haskó, G.; Kuhel, D.G.; Németh, Z.H.; Mabley, J.G.; Stachlewitz, R.F.; Virág, L.; Lohinai, Z.; Southan, G.J.; Salzman, A.L.; Szabó, C. Inosine inhibits inflammatory cytokine production by a posttranscriptional mechanism and protects against endotoxin-induced shock. J. Immunol. 2000, 164, 1013–1019. https://doi.org/10.4049/jimmunol.164.2.1013.
- 88.
Cekic, C.; Linden, J. Purinergic regulation of the immune system. Nat. Rev. Immunol. 2016, 16, 177–192. https://doi.org/10.1038/nri.2016.4.
- 89.
Fucikova, J.; Kepp, O.; Kasikova, L.; Petroni, G.; Yamazaki, T.; Liu, P.; Zhao, L.; Spisek, R.; Kroemer, G.; Galluzzi, L. Detection of immunogenic cell death and its relevance for cancer therapy. Cell Death Dis 2020, 11, 1013. https://doi.org/10.1038/s41419-020-03221-2.
- 90.
Zhang, L.; Jiang, L.; Yu, L.; Li, Q.; Tian, X.; He, J.; Zeng, L.; Yang, Y.; Wang, C.; Wei, Y.; et al. Inhibition of UBA6 by inosine augments tumour immunogenicity and responses. Nat. Commun. 2022, 13, 5413. https://doi.org/10.1038/s41467-022-33116-z.
- 91.
Kaźmierczak-Siedlecka, K.; Daca, A.; Fic, M.; van de Wetering, T.; Folwarski, M.; Makarewicz, W. Therapeutic methods of gut microbiota modification in colorectal cancer management—Fecal microbiota transplantation, prebiotics, probiotics, and synbiotics. Gut Microbes 2020, 11, 1518–1530. https://doi.org/10.1080/19490976.2020.1764309.
- 92.
Chattopadhyay, I.; Nandi, D.; Nag, A. The pint- sized powerhouse: Illuminating the mighty role of the gut microbiome in improving the outcome of anti- cancer therapy. Semin. Cancer Biol. 2021, 70, 98–111. https://doi.org/10.1016/j.semcancer.2020.07.012.
- 93.
Masheghati, F.; Asgharzadeh, M.R.; Jafari, A.; Masoudi, N.; Maleki-Kakelar, H. The role of gut microbiota and probiotics in preventing, treating, and boosting the immune system in colorectal cancer. Life Sci. 2024, 344, 122529. https://doi.org/10.1016/j.lfs.2024.122529.
- 94.
Mazhar, M.; Zhu, Y.; Qin, L. The Interplay of Dietary Fibers and Intestinal Microbiota Affects Type 2 Diabetes by Generating Short-Chain Fatty Acids. Foods 2023, 12, 1023. https://doi.org/10.3390/foods12051023.
- 95.
Owens, J.A.; Saeedi, B.J.; Naudin, C.R.; Hunter-Chang, S.; Barbian, M.E.; Eboka, R.U.; Askew, L.; Darby, T.M.; Robinson, B.S.; Jones, R.M. Lactobacillus rhamnosus GG Orchestrates an Antitumor Immune Response. Cell. Mol. Gastroenterol. Hepatol. 2021, 12, 1311–1327. https://doi.org/10.1016/j.jcmgh.2021.06.001.
- 96.
Wang, J.; Huang, M.; Du, Y.; Chen, H.; Li, Z.; Zhai, T.; Ou, Z.; Huang, Y.; Bu, F.; Zhen, H.; et al. Lactobacillus rhamnosus GG Regulates Host IFN-I Through the RIG-I Signalling Pathway to Inhibit Herpes Simplex Virus Type 2 Infection. Probiotics Antimicrob Proteins 2024, 16, 1966–1978. https://doi.org/10.1007/s12602-023-10137-8.
- 97.
Xie, Y.; Liu, F. The role of the gut microbiota in tumor, immunity, and immunotherapy. Front. Immunol. 2024, 15, 1410928. https://doi.org/10.3389/fimmu.2024.1410928.
- 98.
Si, W.; Zhao, X.; Li, R.; Li, Y.; Ma, C.; Zhao, X.; Bugno, J.; Qin, Y.; Zhang, J.; Liu, H.; et al. Lactobacillus rhamnosus GG induces STING-dependent IL-10 in intestinal monocytes and alleviates inflammatory colitis in mice. J. Clin. Invest. 2025, 135. https://doi.org/10.1172/jci174910.
- 99.
Luo, Z.W.; Xia, K.; Liu, Y.W.; Liu, J.H.; Rao, S.S.; Hu, X.K.; Chen, C.Y.; Xu, R.; Wang, Z.X.; Xie, H. Extracellular Vesicles from Akkermansia muciniphila Elicit Antitumor Immunity Against Prostate Cancer via Modulation of CD8+ T Cells and Macrophages. Int. J. Nanomed. 2021, 16, 2949–2963. https://doi.org/10.2147/ijn.S304515.
- 100.
Xu, Y.; Tan, X.; Yang, Q.; Fang, Z.; Chen, W. Akkermansia muciniphila outer membrane protein regulates recruitment of CD8+ T cells in lung adenocarcinoma and through JAK-STAT signalling pathway. Microb. Biotechnol. 2024, 17, e14522. https://doi.org/10.1111/1751-7915.14522.
- 101.
Derosa, L.; Routy, B.; Thomas, A.M.; Iebba, V.; Zalcman, G.; Friard, S.; Mazieres, J.; Audigier-Valette, C.; Moro-Sibilot, D.; Goldwasser, F.; et al. Intestinal Akkermansia muciniphila predicts clinical response to PD-1 blockade in patients with advanced non-small-cell lung cancer. Nat. Med. 2022, 28, 315–324. https://doi.org/10.1038/s41591-021-01655-5.
- 102.
Liu, R.; Zou, Y.; Wang, W.Q.; Chen, J.H.; Zhang, L.; Feng, J.; Yin, J.Y.; Mao, X.Y.; Li, Q.; Luo, Z.Y.; et al. Gut microbial structural variation associates with immune checkpoint inhibitor response. Nat. Commun. 2023, 14, 7421. https://doi.org/10.1038/s41467-023-42997-7.
- 103.
Paz Del Socorro, T.; Oka, K.; Boulard, O.; Takahashi, M.; Poulin, L.F.; Hayashi, A.; Chamaillard, M. The biotherapeutic Clostridium butyricum MIYAIRI 588 strain potentiates enterotropism of Rorγt+Treg and PD-1 blockade efficacy. Gut Microbes 2024, 16, 2315631. https://doi.org/10.1080/19490976.2024.2315631.
- 104.
Dizman, N.; Meza, L.; Bergerot, P.; Alcantara, M.; Dorff, T.; Lyou, Y.; Frankel, P.; Cui, Y.; Mira, V.; Llamas, M.; et al. Nivolumab plus ipilimumab with or without live bacterial supplementation in metastatic renal cell carcinoma: A randomized phase 1 trial. Nat. Med. 2022, 28, 704–712. https://doi.org/10.1038/s41591-022-01694-6.
- 105.
Ebrahimi, H.; Dizman, N.; Meza, L.; Malhotra, J.; Li, X.; Dorff, T.; Frankel, P.; Llamas-Quitiquit, M.; Hsu, J.; Zengin, Z.B.; et al. Cabozantinib and nivolumab with or without live bacterial supplementation in metastatic renal cell carcinoma: A randomized phase 1 trial. Nat. Med. 2024, 30, 2576–2585. https://doi.org/10.1038/s41591-024-03086-4.
- 106.
Xu, T.; Wu, X.; Liu, J.; Sun, J.; Wang, X.; Fan, G.; Meng, X.; Zhang, J.; Zhang, Y. The regulatory roles of dietary fibers on host health via gut microbiota-derived short chain fatty acids. Curr. Opin. Pharmacol. 2022, 62, 36–42. https://doi.org/10.1016/j.coph.2021.11.001.
- 107.
Yang, J.; Yang, H.; Li, Y. The triple interactions between gut microbiota, mycobiota and host immunity. Crit. Rev. Food Sci. Nutr. 2023, 63, 11604–11624. https://doi.org/10.1080/10408398.2022.2094888.
- 108.
Gurbatri, C.R.; Lia, I.; Vincent, R.; Coker, C.; Castro, S.; Treuting, P.M.; Hinchliffe, T.E.; Arpaia, N.; Danino, T. Engineered probiotics for local tumor delivery of checkpoint blockade nanobodies. Sci. Transl. Med. 2020, 12, eaax0876. https://doi.org/10.1126/scitranslmed.aax0876.
- 109.
Qin, S.; Liu, Y.; He, G.; Yang, J.; Zeng, F.; Lu, Q.; Wang, M.; He, B.; Song, Y. Spatiotemporal Delivery of Dual Nanobodies by Engineered Probiotics to Reverse Tumor Immunosuppression via Targeting Tumor-Derived Exosomes. ACS Nano 2024, 18, 26858–26871. https://doi.org/10.1021/acsnano.4c08117.
- 110.
Canale, F.P.; Basso, C.; Antonini, G.; Perotti, M.; Li, N.; Sokolovska, A.; Neumann, J.; James, M.J.; Geiger, S.; Jin, W.; et al. Metabolic modulation of tumours with engineered bacteria for immunotherapy. Nature 2021, 598, 662–666. https://doi.org/10.1038/s41586-021-04003-2.
- 111.
Wang, H.; Xu, F.; Yao, C.; Dai, H.; Xu, J.; Wu, B.; Tian, B.; Shi, X.; Wang, C. Engineering bacteria for cancer immunotherapy by inhibiting IDO activity and reprogramming CD8+ T cell response. Proc. Natl. Acad Sci. USA 2024, 121, e2412070121. https://doi.org/10.1073/pnas.2412070121.
- 112.
Routy, B.; Lenehan, J.G.; Miller, W.H., Jr.; Jamal, R.; Messaoudene, M.; Daisley, B.A.; Hes, C.; Al, K.F.; Martinez-Gili, L.; Punčochář, M.; et al. Fecal microbiota transplantation plus anti-PD-1 immunotherapy in advanced melanoma: A phase I trial. Nat. Med. 2023, 29, 2121–2132. https://doi.org/10.1038/s41591-023-02453-x.
- 113.
Qiao, X.; Biliński, J.; Wang, L.; Yang, T.; Luo, R.; Fu, Y.; Yang, G. Safety and efficacy of fecal microbiota transplantation in the treatment of graft-versus-host disease. Bone Marrow Transplant. 2023, 58, 10–19. https://doi.org/10.1038/s41409-022-01824-1.
- 114.
Lin, A.; Jiang, A.; Huang, L.; Li, Y.; Zhang, C.; Zhu, L.; Mou, W.; Liu, Z.; Zhang, J.; Cheng, Q.; et al. From chaos to order: Optimizing fecal microbiota transplantation for enhanced immune checkpoint inhibitors efficacy. Gut Microbes 2025, 17, 2452277. https://doi.org/10.1080/19490976.2025.2452277.
- 115.
DeLeon, O.; Mocanu, M.; Tan, A.; Sidebottom, A.M.; Koval, J.; Ceccato, H.D.; Kralicek, S.; Colgan, J.J.; St George, M.M.; Lake, J.M.; et al. Microbiome mismatches from microbiota transplants lead to persistent off-target metabolic and immunomodulatory effects. Cell 2025, 188, 3927–3941. https://doi.org/10.1016/j.cell.2025.05.014.
- 116.
Zhu, X.; Hu, M.; Huang, X.; Li, L.; Lin, X.; Shao, X.; Li, J.; Du, X.; Zhang, X.; Sun, R.; et al. Interplay between gut microbial communities and metabolites modulates pan-cancer immunotherapy responses. Cell Metab. 2025, 37, 806–823. https://doi.org/10.1016/j.cmet.2024.12.013.
- 117.
Saravanan, C.; Gopinath, N.K.; Ganesan, R.; Thirumurugan, D. Challenges and limitations in using bacterial metabolites as immunomodulators. Front. Cell. Infect. Microbiol. 2025, 15, 1535394. https://doi.org/10.3389/fcimb.2025.1535394.
- 118.
Kong, C.; Liang, L.; Liu, G.; Du, L.; Yang, Y.; Liu, J.; Shi, D.; Li, X.; Ma, Y. Integrated metagenomic and metabolomic analysis reveals distinct gut-microbiome-derived phenotypes in early-onset colorectal cancer. Gut 2023, 72, 1129–1142. https://doi.org/10.1136/gutjnl-2022-327156.

This work is licensed under a Creative Commons Attribution 4.0 International License.


