Atherosclerosis is a vascular disease characterized by dysfunction of vascular endothelial cells, infiltration of macrophages, formation of foam cells, and proliferation and migration of vascular smooth muscle cells. Current studies have shown that the NLRP3 inflammasome plays a key regulatory role in the cellular pathological process of atherosclerosis. This review systematically summarizes the role and underlying mechanism of the NLRP3 inflammasome in the cellular pathological processes of atherosclerosis: various risk factors activate this inflammasome to exacerbate cell damage, while many endogenous factors exert protective effects by inhibiting its activity. In addition, the article reviews intervention strategies ranging from specific chemical drugs to multi-target natural products, and discusses emerging new technologies such as nano-targeted delivery, providing a theoretical basis for anti-inflammatory therapeutic strategies. Significance statement: This study focuses on NLRP3 inflammasome, whose key role in the process of atherosclerosis is becoming increasingly clear. These findings suggest that targeting the NLRP3 inflammasome may represent a potential direction for developing new therapeutic strategies, although most agents remain at the preclinical stage and require further validation.
- Open Access
- Review
NLRP3 Inflammasome: A Potential Therapeutic Target for Atherosclerosis
- Mengqi Wang 1,†,
- Jun Cheng 2,†,
- Fen Feng 3,*,
- Linxi Chen 1,*
Author Information
Received: 06 Aug 2025 | Revised: 17 Sep 2025 | Accepted: 13 Nov 2025 | Published: 07 Apr 2026
Abstract
Graphical Abstract
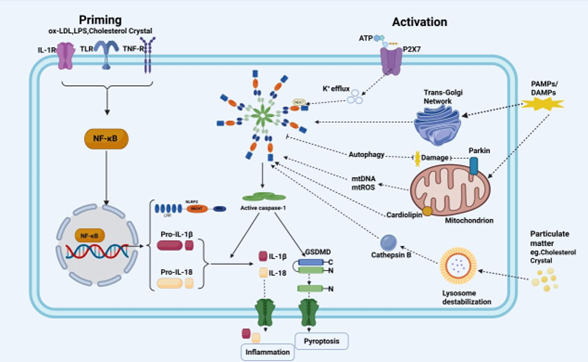
Keywords
NLRP3 inflammasome | atherosclerosis | endothelial dysfunction | macrophage infiltration | the proliferation and migration of vascular smooth muscle cells
References
- 1.
Emini Veseli, B.; Perrotta, P.; De Meyer, G.R.A.; Roth, L.; Van der Donckt, C.; Martinet, W.; De Meyer, G.R.Y. Animal models of atherosclerosis. Eur. J. Pharmacol. 2017, 816, 3–13. https://doi.org/10.1016/j.ejphar.2017.05.010.
- 2.
Miao, J.; Zang, X.; Cui, X.; Zhang, J. Autophagy, Hyperlipidemia, and Atherosclerosis. Adv. Exp. Med. Biol. 2020, 1207, 237–264. https://doi.org/10.1007/978-981-15-4272-5_18.
- 3.
Tall, A.R.; Yvan-Charvet, L. Cholesterol, inflammation and innate immunity. Nat. Rev. Immunol. 2015, 15, 104–116. https://doi.org/10.1038/nri3793.
- 4.
Ezzati, M.; Obermeyer, Z.; Tzoulaki, I.; Mayosi, B.M.; Elliott, P.; Leon, D.A. Contributions of risk factors and medical care to cardiovascular mortality trends. Nat. Rev. Cardiol. 2015, 12, 508–530. https://doi.org/10.1038/nrcardio.2015.82.
- 5.
Tsioufis, P.; Theofilis, P.; Tsioufis, K.; Tousoulis, D. The Impact of Cytokines in Coronary Atherosclerotic Plaque: Current Therapeutic Approaches. Int. J. Mol. Sci. 2022, 23, 15937. https://doi.org/10.3390/ijms232415937.
- 6.
Schroder, K.; Tschopp, J. The inflammasomes. Cell 2010, 140, 821–832. https://doi.org/10.1016/j.cell.2010.01.040.
- 7.
Cai, X.; Chen, J.; Xu, H.; Liu, S.; Jiang, Q.X.; Halfmann, R.; Chen, Z.J. Prion-like polymerization underlies signal transduction in antiviral immune defense and inflammasome activation. Cell 2014, 156, 1207–1222. https://doi.org/10.1016/j.cell.2014.01.063.
- 8.
Li, Y.; Fu, T.M.; Lu, A.; Witt, K.; Ruan, J.; Shen, C.; Wu, H. Cryo-EM structures of ASC and NLRC4 CARD filaments reveal a unified mechanism of nucleation and activation of caspase-1. Proc. Natl. Acad. Sci. USA 2018, 115, 10845–10852. https://doi.org/10.1073/pnas.1810524115.
- 9.
Shi, J.; Zhao, Y.; Wang, K.; Shi, X.; Wang, Y.; Huang, H.; Zhuang, Y.; Cai, T.; Wang, F.; Shao, F. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 2015, 526, 660–665. https://doi.org/10.1038/nature15514.
- 10.
Kesavardhana, S.; Kanneganti, T.D. Mechanisms governing inflammasome activation, assembly and pyroptosis induction. Int. Immunol. 2017, 29, 201–210. https://doi.org/10.1093/intimm/dxx018.
- 11.
Takahashi, M. NLRP3 inflammasome as a key driver of vascular disease. Cardiovasc. Res. 2022, 118, 372–385. https://doi.org/10.1093/cvr/cvab010.
- 12.
Christgen, S.; Place, D.E.; Kanneganti, T.D. Toward targeting inflammasomes: Insights into their regulation and activation. Cell Res 2020, 30, 315–327. https://doi.org/10.1038/s41422-020-0295-8.
- 13.
Christgen, S.; Kanneganti, T.D. Inflammasomes and the fine line between defense and disease. Curr. Opin. Immunol. 2020, 62, 39–44. https://doi.org/10.1016/j.coi.2019.11.007.
- 14.
Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. https://doi.org/10.3390/ijms20133328.
- 15.
Takahashi, M. Cell-Specific Roles of NLRP3 Inflammasome in Myocardial Infarction. J. Cardiovasc. Pharmacol. 2019, 74, 188–193. https://doi.org/10.1097/fjc.0000000000000709.
- 16.
McKee, C.M.; Coll, R.C. NLRP3 inflammasome priming: A riddle wrapped in a mystery inside an enigma. J. Leukoc. Biol. 2020, 108, 937–952. https://doi.org/10.1002/jlb.3mr0720-513r.
- 17.
Di Virgilio, F.; Dal Ben, D.; Sarti, A.C.; Giuliani, A.L.; Falzoni, S. The P2X7 Receptor in Infection and Inflammation. Immunity 2017, 47, 15–31. https://doi.org/10.1016/j.immuni.2017.06.020.
- 18.
He, Y.; Zeng, M.Y.; Yang, D.; Motro, B.; Núñez, G. NEK7 is an essential mediator of NLRP3 activation downstream of potassium efflux. Nature 2016, 530, 354–357. https://doi.org/10.1038/nature16959.
- 19.
Sharif, H.; Wang, L.; Wang, W.L.; Magupalli, V.G.; Andreeva, L.; Qiao, Q.; Hauenstein, A.V.; Wu, Z.; Núñez, G.; Mao, Y.; et al. Structural mechanism for NEK7-licensed activation of NLRP3 inflammasome. Nature 2019, 570, 338–343. https://doi.org/10.1038/s41586-019-1295-z.
- 20.
Zhou, R.; Yazdi, A.S.; Menu, P.; Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 2011, 469, 221–225. https://doi.org/10.1038/nature09663.
- 21.
Shimada, K.; Crother, T.R.; Karlin, J.; Dagvadorj, J.; Chiba, N.; Chen, S.; Ramanujan, V.K.; Wolf, A.J.; Vergnes, L.; Ojcius, D.M.; et al. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity 2012, 36, 401–414. https://doi.org/10.1016/j.immuni.2012.01.009.
- 22.
Próchnicki, T.; Latz, E. Inflammasomes on the Crossroads of Innate Immune Recognition and Metabolic Control. Cell Metab. 2017, 26, 71–93. https://doi.org/10.1016/j.cmet.2017.06.018.
- 23.
Iyer, S.S.; He, Q.; Janczy, J.R.; Elliott, E.I.; Zhong, Z.; Olivier, A.K.; Sadler, J.J.; Knepper-Adrian, V.; Han, R.; Qiao, L.; et al. Mitochondrial cardiolipin is required for Nlrp3 inflammasome activation. Immunity 2013, 39, 311–323. https://doi.org/10.1016/j.immuni.2013.08.001.
- 24.
Zhong, Z.; Liang, S.; Sanchez-Lopez, E.; He, F.; Shalapour, S.; Lin, X.J.; Wong, J.; Ding, S.; Seki, E.; Schnabl, B.; et al. New mitochondrial DNA synthesis enables NLRP3 inflammasome activation. Nature 2018, 560, 198–203. https://doi.org/10.1038/s41586-018-0372-z.
- 25.
Zhong, Z.; Umemura, A.; Sanchez-Lopez, E.; Liang, S.; Shalapour, S.; Wong, J.; He, F.; Boassa, D.; Perkins, G.; Ali, S.R.; et al. NF-κB Restricts Inflammasome Activation via Elimination of Damaged Mitochondria. Cell 2016, 164, 896–910. https://doi.org/10.1016/j.cell.2015.12.057.
- 26.
Nakahira, K.; Haspel, J.A.; Rathinam, V.A.; Lee, S.J.; Dolinay, T.; Lam, H.C.; Englert, J.A.; Rabinovitch, M.; Cernadas, M.; Kim, H.P.; et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat. Immunol. 2011, 12, 222–230. https://doi.org/10.1038/ni.1980.
- 27.
Rajamäki, K.; Lappalainen, J.; Oörni, K.; Välimäki, E.; Matikainen, S.; Kovanen, P.T.; Eklund, K.K. Cholesterol crystals activate the NLRP3 inflammasome in human macrophages: A novel link between cholesterol metabolism and inflammation. PLoS ONE 2010, 5, e11765. https://doi.org/10.1371/journal.pone.0011765.
- 28.
Chen, J.; Chen, Z.J. PtdIns4P on dispersed trans-Golgi network mediates NLRP3 inflammasome activation. Nature 2018, 564, 71–76. https://doi.org/10.1038/s41586-018-0761-3.
- 29.
Jiang, C.; Xie, S.; Yang, G.; Wang, N. Spotlight on NLRP3 Inflammasome: Role in Pathogenesis and Therapies of Atherosclerosis. J. Inflamm. Res. 2021, 14, 7143–7172. https://doi.org/10.2147/jir.s344730.
- 30.
Theofilis, P.; Oikonomou, E.; Chasikidis, C.; Tsioufis, K.; Tousoulis, D. Inflammasomes in Atherosclerosis-From Pathophysiology to Treatment. Pharmaceuticals 2023, 16, 1211. https://doi.org/10.3390/ph16091211.
- 31.
He, L.; Zhang, C.L.; Chen, Q.; Wang, L.; Huang, Y. Endothelial shear stress signal transduction and atherogenesis: From mechanisms to therapeutics. Pharmacol. Ther. 2022, 235, 108152. https://doi.org/10.1016/j.pharmthera.2022.108152.
- 32.
Lv, Y.; Jiang, Z.; Zhou, W.; Yang, H.; Jin, G.; Wang, D.; Kong, C.; Qian, Z.; Gu, Y.; Chen, S.; et al. Low-Shear Stress Promotes Atherosclerosis via Inducing Endothelial Cell Pyroptosis Mediated by IKKepsilon/STAT1/NLRP3 Pathway. Inflammation 2024, 47, 1053–1066. https://doi.org/10.1007/s10753-023-01960-w.
- 33.
Zhuang, T.; Liu, J.; Chen, X.; Zhang, L.; Pi, J.; Sun, H.; Li, L.; Bauer, R.; Wang, H.; Yu, Z.; et al. Endothelial Foxp1 Suppresses Atherosclerosis via Modulation of Nlrp3 Inflammasome Activation. Circ. Res. 2019, 125, 590–605. https://doi.org/10.1161/circresaha.118.314402.
- 34.
Wu, X.; Zhang, H.; Qi, W.; Zhang, Y.; Li, J.; Li, Z.; Lin, Y.; Bai, X.; Liu, X.; Chen, X.; et al. Nicotine promotes atherosclerosis via ROS-NLRP3-mediated endothelial cell pyroptosis. Cell Death Dis. 2018, 9, 171. https://doi.org/10.1038/s41419-017-0257-3.
- 35.
Duan, F.; Zeng, C.; Liu, S.; Gong, J.; Hu, J.; Li, H.; Tan, H. α1-nAchR-Mediated Signaling Through Lipid Raft Is Required for Nicotine-Induced NLRP3 Inflammasome Activation and Nicotine-Accelerated Atherosclerosis. Front. Cell Dev. Biol. 2021, 9, 724699. https://doi.org/10.3389/fcell.2021.724699.
- 36.
Chen, C.Y.; Leu, H.B.; Wang, S.C.; Tsai, S.H.; Chou, R.H.; Lu, Y.W.; Tsai, Y.L.; Kuo, C.S.; Huang, P.H.; Chen, J.W.; et al. Inhibition of Trimethylamine N-Oxide Attenuates Neointimal Formation Through Reduction of Inflammasome and Oxidative Stress in a Mouse Model of Carotid Artery Ligation. Antioxid. Redox Signal. 2023, 38, 215–233. https://doi.org/10.1089/ars.2021.0115.
- 37.
Miyakawa, H.; Woo, S.K.; Dahl, S.C.; Handler, J.S.; Kwon, H.M. Tonicity-responsive enhancer binding protein, a rel-like protein that stimulates transcription in response to hypertonicity. Proc. Natl. Acad. Sci. USA 1999, 96, 2538–2542. https://doi.org/10.1073/pnas.96.5.2538.
- 38.
Boland, B.S.; Widjaja, C.E.; Banno, A.; Zhang, B.; Kim, S.H.; Stoven, S.; Peterson, M.R.; Jones, M.C.; Su, H.I.; Crowe, S.E.; et al. Immunodeficiency and autoimmune enterocolopathy linked to NFAT5 haploinsufficiency. J. Immunol. 2015, 194, 2551–2560. https://doi.org/10.4049/jimmunol.1401463.
- 39.
Ma, P.; Zha, S.; Shen, X.; Zhao, Y.; Li, L.; Yang, L.; Lei, M.; Liu, W. NFAT5 mediates hypertonic stress-induced atherosclerosis via activating NLRP3 inflammasome in endothelium. Cell Commun. Signal. 2019, 17, 102. https://doi.org/10.1186/s12964-019-0406-7.
- 40.
Hu, T.; Zhu, P.; Liu, Y.; Zhu, H.; Geng, J.; Wang, B.; Yuan, G.; Peng, Y.; Xu, B. PM2.5 induces endothelial dysfunction via activating NLRP3 inflammasome. Environ. Toxicol. 2021, 36, 1886–1893. https://doi.org/10.1002/tox.23309.
- 41.
Mendall, M.A.; Goggin, P.M.; Molineaux, N.; Levy, J.; Toosy, T.; Strachan, D.; Camm, A.J.; Northfield, T.C. Relation of Helicobacter pylori infection and coronary heart disease. Br. Heart J. 1994, 71, 437–439. https://doi.org/10.1136/hrt.71.5.437.
- 42.
Li, B.W.; Liu, Y.; Zhang, L.; Guo, X.Q.; Wen, C.; Zhang, F.; Luo, X.Y.; Xia, Y.P. Cytotoxin-associated gene A (CagA) promotes aortic endothelial inflammation and accelerates atherosclerosis through the NLRP3/caspase-1/IL-1β axis. FASEB J. 2021, 35, e21942. https://doi.org/10.1096/fj.202100695RR.
- 43.
Quarato, G.; Guy, C.S.; Grace, C.R.; Llambi, F.; Nourse, A.; Rodriguez, D.A.; Wakefield, R.; Frase, S.; Moldoveanu, T.; Green, D.R. Sequential Engagement of Distinct MLKL Phosphatidylinositol-Binding Sites Executes Necroptosis. Mol. Cell 2016, 61, 589–601. https://doi.org/10.1016/j.molcel.2016.01.011.
- 44.
Wu, Q.; He, X.; Wu, L.M.; Zhang, R.Y.; Li, L.M.; Wu, C.M.; Lu, Y.B.; Hu, B.; Shi, C.; Lu, Z.F.; et al. MLKL Aggravates Ox-LDL-Induced Cell Pyroptosis via Activation of NLRP3 Inflammasome in Human Umbilical Vein Endothelial Cells. Inflammation 2020, 43, 2222–2231. https://doi.org/10.1007/s10753-020-01289-8.
- 45.
Huang, Y.; Wang, Y.; Ouyang, Y. Elevated microRNA-135b-5p relieves neuronal injury and inflammation in post-stroke cognitive impairment by targeting NR3C2. Int. J. Neurosci. 2022, 132, 58–66. https://doi.org/10.1080/00207454.2020.1802265.
- 46.
Wang, C.; Hu, F. Long noncoding RNA SOX2OT silencing alleviates cerebral ischemia-reperfusion injury via miR-135a-5p-mediated NR3C2 inhibition. Brain Res. Bull. 2021, 173, 193–202. https://doi.org/10.1016/j.brainresbull.2021.05.018.
- 47.
Chen, X.; Li, W.; Chang, C. NR3C2 mediates oxidised low-density lipoprotein-induced human coronary endothelial cells dysfunction via modulation of NLRP3 inflammasome activation. Autoimmunity 2023, 56, 2189135. https://doi.org/10.1080/08916934.2023.2189135.
- 48.
Bian, F.; Yang, X.; Zhou, F.; Wu, P.H.; Xing, S.; Xu, G.; Li, W.; Chi, J.; Ouyang, C.; Zhang, Y.; et al. C-reactive protein promotes atherosclerosis by increasing LDL transcytosis across endothelial cells. Br. J. Pharmacol. 2014, 171, 2671–2684. https://doi.org/10.1111/bph.12616.
- 49.
Bian, F.; Yang, X.Y.; Xu, G.; Zheng, T.; Jin, S. CRP-Induced NLRP3 Inflammasome Activation Increases LDL Transcytosis Across Endothelial Cells. Front. Pharmacol. 2019, 10, 40. https://doi.org/10.3389/fphar.2019.00040.
- 50.
Tang, Y.; Yan, J.H.; Ge, Z.W.; Fei, A.H.; Zhang, Y.C. LncRNA Gaplinc promotes the pyroptosis of vascular endothelial cells through SP1 binding to enhance NLRP3 transcription in atherosclerosis. Cell. Signal. 2022, 99, 110420. https://doi.org/10.1016/j.cellsig.2022.110420.
- 51.
Hou, J.; Deng, Q.; Deng, X.; Zhong, W.; Liu, S.; Zhong, Z. MicroRNA-146a-5p alleviates lipopolysaccharide-induced NLRP3 inflammasome injury and pro-inflammatory cytokine production via the regulation of TRAF6 and IRAK1 in human umbilical vein endothelial cells (HUVECs). Ann. Transl. Med. 2021, 9, 1433. https://doi.org/10.21037/atm-21-3903.
- 52.
Bai, B.; Yang, Y.; Ji, S.; Wang, S.; Peng, X.; Tian, C.; Sun, R.C.; Yu, T.; Chu, X.M. MicroRNA-302c-3p inhibits endothelial cell pyroptosis via directly targeting NOD-, LRR- and pyrin domain-containing protein 3 in atherosclerosis. J. Cell. Mol. Med. 2021, 25, 4373–4386. https://doi.org/10.1111/jcmm.16500.
- 53.
Li, X.; Cao, Y.; Xu, X.; Wang, C.; Ni, Q.; Lv, X.; Yang, C.; Zhang, Z.; Qi, X.; Song, G. Sleep Deprivation Promotes Endothelial Inflammation and Atherogenesis by Reducing Exosomal miR-182-5p. Arterioscler. Thromb. Vasc. Biol. 2023, 43, 995–1014. https://doi.org/10.1161/atvbaha.123.319026.
- 54.
Kanter, J.E. FOXP1: A Gatekeeper of Endothelial Cell Inflammation. Circ. Res. 2019, 125, 606–608. https://doi.org/10.1161/circresaha.119.315687.
- 55.
Lin, Z.; Pan, X.; Wu, F.; Ye, D.; Zhang, Y.; Wang, Y.; Jin, L.; Lian, Q.; Huang, Y.; Ding, H.; et al. Fibroblast growth factor 21 prevents atherosclerosis by suppression of hepatic sterol regulatory element-binding protein-2 and induction of adiponectin in mice. Circulation 2015, 131, 1861–1871. https://doi.org/10.1161/circulationaha.115.015308.
- 56.
Zeng, Z.; Zheng, Q.; Chen, J.; Tan, X.; Li, Q.; Ding, L.; Zhang, R.; Lin, X. FGF21 mitigates atherosclerosis via inhibition of NLRP3 inflammasome-mediated vascular endothelial cells pyroptosis. Exp. Cell Res. 2020, 393, 112108. https://doi.org/10.1016/j.yexcr.2020.112108.
- 57.
Galkina, E.; Ley, K. Immune and inflammatory mechanisms of atherosclerosis (*). Annu. Rev. Immunol. 2009, 27, 165–197. https://doi.org/10.1146/annurev.immunol.021908.132620.
- 58.
Mehta, S.; Dhawan, V. Exposure of cigarette smoke condensate activates NLRP3 inflammasome in THP-1 cells in a stage-specific manner: An underlying role of innate immunity in atherosclerosis. Cell. Signal. 2020, 72, 109645. https://doi.org/10.1016/j.cellsig.2020.109645.
- 59.
Ni, H.; Xu, S.; Chen, H.; Dai, Q. Nicotine Modulates CTSS (Cathepsin S) Synthesis and Secretion Through Regulating the Autophagy-Lysosomal Machinery in Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 2054–2069. https://doi.org/10.1161/atvbaha.120.314053.
- 60.
Martinet, W.; Coornaert, I.; Puylaert, P.; De Meyer, G.R.Y. Macrophage Death as a Pharmacological Target in Atherosclerosis. Front. Pharmacol. 2019, 10, 306. https://doi.org/10.3389/fphar.2019.00306.
- 61.
Corrêa, R.; Silva, L.F.F.; Ribeiro, D.J.S.; Almeida, R.D.N.; Santos, I.O.; Corrêa, L.H.; de Sant'Ana, L.P.; Assunção, L.S.; Bozza, P.T.; Magalhães, K.G. Lysophosphatidylcholine Induces NLRP3 Inflammasome-Mediated Foam Cell Formation and Pyroptosis in Human Monocytes and Endothelial Cells. Front. Immunol. 2019, 10, 2927. https://doi.org/10.3389/fimmu.2019.02927.
- 62.
Xu, S.; Chen, H.; Ni, H.; Dai, Q. Targeting HDAC6 attenuates nicotine-induced macrophage pyroptosis via NF-κB/NLRP3 pathway. Atherosclerosis 2021, 317, 1–9. https://doi.org/10.1016/j.atherosclerosis.2020.11.021.
- 63.
Lin, Y.F.; Li, M.H.; Huang, R.H.; Zhang, S.Z.; Xu, X.F.; Zhou, H.M.; Liu, M.H.; Liao, X.X.; Liao, L.Z.; Guo, Y.; et al. GP73 enhances the ox-LDL-induced inflammatory response in THP-1 derived macrophages via affecting NLRP3 inflammasome signaling. Int. J. Cardiol. 2023, 387, 131109. https://doi.org/10.1016/j.ijcard.2023.05.059.
- 64.
Liu, C.; Wu, J.; Jia, H.; Lu, C.; Liu, J.; Li, Y.; Guo, M. Oncostatin M promotes the ox-LDL-induced activation of NLRP3 inflammasomes via the NF-κB pathway in THP-1 macrophages and promotes the progression of atherosclerosis. Ann. Transl. Med. 2022, 10, 456. https://doi.org/10.21037/atm-22-560.
- 65.
Wang, C.; Andreasson, K.I. Odorant receptors in macrophages: Potential targets for atherosclerosis. Trends Immunol. 2022, 43, 262–264. https://doi.org/10.1016/j.it.2022.02.006.
- 66.
Pazzaglia, S.; Pioli, C. Multifaceted Role of PARP-1 in DNA Repair and Inflammation: Pathological and Therapeutic Implications in Cancer and Non-Cancer Diseases. Cells 2019, 9, 41. https://doi.org/10.3390/cells9010041.
- 67.
Fehr, A.R.; Singh, S.A.; Kerr, C.M.; Mukai, S.; Higashi, H.; Aikawa, M. The impact of PARPs and ADP-ribosylation on inflammation and host-pathogen interactions. Genes Dev. 2020, 34, 341–359. https://doi.org/10.1101/gad.334425.119.
- 68.
Mustafa, K.; Han, Y.; He, D.; Wang, Y.; Niu, N.; Jose, P.A.; Jiang, Y.; Kopp, J.B.; Lee, H.; Qu, P. Poly-(ADP-ribose) polymerases inhibition by olaparib attenuates activities of the NLRP3 inflammasome and of NF-κB in THP-1 monocytes. PLoS ONE 2024, 19, e0295837. https://doi.org/10.1371/journal.pone.0295837.
- 69.
Liang, Y.; Xu, X.D.; Xu, X.; Cai, Y.B.; Zhu, Z.X.; Zhu, L.; Ren, K. Linc00657 promoted pyroptosis in THP-1-derived macrophages and exacerbated atherosclerosis via the miR-106b-5p/TXNIP/NLRP3 axis. Int. J. Biol. Macromol. 2023, 253, 126953. https://doi.org/10.1016/j.ijbiomac.2023.126953.
- 70.
Ding, Y.; Gong, W.; Zhang, S.; Shen, J.; Liu, X.; Wang, Y.; Chen, Y.; Meng, G. Protective role of sirtuin3 against oxidative stress and NLRP3 inflammasome in cholesterol accumulation and foam cell formation of macrophages with ox-LDL-stimulation. Biochem. Pharmacol. 2021, 192, 114665. https://doi.org/10.1016/j.bcp.2021.114665.
- 71.
Chen, J.; Sun, X.; Liu, Y.; Zhang, Y.; Zhao, M.; Shao, L. SENP3 attenuates foam cell formation by deSUMOylating NLRP3 in macrophages stimulated with ox-LDL. Cell. Signal. 2024, 117, 111092. https://doi.org/10.1016/j.cellsig.2024.111092.
- 72.
Lee, J.H.; Budanov, A.V.; Karin, M. Sestrins orchestrate cellular metabolism to attenuate aging. Cell Metab. 2013, 18, 792–801. https://doi.org/10.1016/j.cmet.2013.08.018.
- 73.
Keping, Y.; Yunfeng, S.; Pengzhuo, X.; Liang, L.; Chenhong, X.; Jinghua, M. Sestrin1 inhibits oxidized low-density lipoprotein-induced activation of NLRP3 inflammasome in macrophages in a murine atherosclerosis model. Eur. J. Immunol. 2020, 50, 1154–1166. https://doi.org/10.1002/eji.201948427.
- 74.
Yuan, R.; Zhang, W.; Nie, P.; Lan, K.; Yang, X.; Yin, A.; Xiao, Q.; Shen, Y.; Xu, K.; Wang, X.; et al. Nur77 Deficiency Exacerbates Macrophage NLRP3 Inflammasome-Mediated Inflammation and Accelerates Atherosclerosis. Oxidative Med. Cell. Longev. 2022, 2022, 2017815. https://doi.org/10.1155/2022/2017815.
- 75.
Wu, Q.Q.; Liu, X.Y.; Xiong, L.X.; Shang, J.Y.; Mai, X.Y.; Pang, R.P.; Su, Y.X.; Yu, B.X.; Yuan, J.N.; Yang, C.; et al. Reduction of Intracellular Chloride Concentration Promotes Foam Cell Formation. Circ. J. 2016, 80, 1024–1033. https://doi.org/10.1253/circj.CJ-15-1209.
- 76.
Ding, W.; Li, J.; Wang, L.; Zhang, M.; Zheng, F. ClC-2 inhibition prevents macrophage foam cell formation by suppressing Nlrp3 inflammasome activation. Biosci. Biotechnol. Biochem. 2020, 84, 2096–2103. https://doi.org/10.1080/09168451.2020.1793294.
- 77.
Xu, W.; Qian, L.; Yuan, X.; Lu, Y. MicroRNA-223-3p inhibits oxidized low-density lipoprotein-mediated NLRP3 inflammasome activation via directly targeting NLRP3 and FOXO3. Clin. Hemorheol. Microcirc. 2022, 81, 241–253. https://doi.org/10.3233/ch-211232.
- 78.
Song, J.; Yang, S.; Yin, R.; Xiao, Q.; Ma, A.; Pan, X. MicroRNA-181a regulates the activation of the NLRP3 inflammatory pathway by targeting MEK1 in THP-1 macrophages stimulated by ox-LDL. J. Cell. Biochem. 2019, 120, 13640–13650. https://doi.org/10.1002/jcb.28637.
- 79.
Wang, G.; Jing, S.Y.; Liu, G.; Guo, X.J.; Zhao, W.; Jia, X.L.; Zheng, M.Q.; Tan, W.Y. miR-99a-5p: A Potential New Therapy for Atherosclerosis by Targeting mTOR and Then Inhibiting NLRP3 Inflammasome Activation and Promoting Macrophage Autophagy. Dis. Markers 2022, 2022, 7172583. https://doi.org/10.1155/2022/7172583.
- 80.
Yi, C.; Sun, W.; Ding, L.; Yan, M.; Sun, C.; Qiu, C.; Wang, D.; Wu, L. Short-Chain Fatty Acids Weaken Ox-LDL-Induced Cell Inflammatory Injury by Inhibiting the NLRP3/Caspase-1 Pathway and Affecting Cellular Metabolism in THP-1 Cells. Molecules 2022, 27, 8801. https://doi.org/10.3390/molecules27248801.
- 81.
Moon, J.S.; Nakahira, K.; Chung, K.P.; DeNicola, G.M.; Koo, M.J.; Pabón, M.A.; Rooney, K.T.; Yoon, J.H.; Ryter, S.W.; Stout-Delgado, H.; et al. NOX4-dependent fatty acid oxidation promotes NLRP3 inflammasome activation in macrophages. Nat. Med. 2016, 22, 1002–1012. https://doi.org/10.1038/nm.4153.
- 82.
Hohensinner, P.J.; Lenz, M.; Haider, P.; Mayer, J.; Richter, M.; Kaun, C.; Goederle, L.; Brekalo, M.; Salzmann, M.; Sharma, S.; et al. Pharmacological inhibition of fatty acid oxidation reduces atherosclerosis progression by suppression of macrophage NLRP3 inflammasome activation. Biochem. Pharmacol. 2021, 190, 114634. https://doi.org/10.1016/j.bcp.2021.114634.
- 83.
Lu, Q.B.; Wan, M.Y.; Wang, P.Y.; Zhang, C.X.; Xu, D.Y.; Liao, X.; Sun, H.J. Chicoric acid prevents PDGF-BB-induced VSMC dedifferentiation, proliferation and migration by suppressing ROS/NFκB/mTOR/P70S6K signaling cascade. Redox Biol. 2018, 14, 656–668. https://doi.org/10.1016/j.redox.2017.11.012.
- 84.
An, J.; Ouyang, L.; Yu, C.; Carr, S.M.; Ramprasath, T.; Liu, Z.; Song, P.; Zou, M.H.; Ding, Y. Nicotine exacerbates atherosclerosis and plaque instability via NLRP3 inflammasome activation in vascular smooth muscle cells. Theranostics 2023, 13, 2825–2842. https://doi.org/10.7150/thno.81388.
- 85.
Qiao, H.; Yang, B.; Lv, X.; Liu, Y. Exposure to TCBPA stimulates the growth of arterial smooth muscle cells through the activation of the ROS/NF-κB/NLRP3 signaling pathway. Toxicology 2024, 503, 153759. https://doi.org/10.1016/j.tox.2024.153759.
- 86.
Yang, X.; Wang, C.; Zhu, G.; Guo, Z.; Fan, L. METTL14/YTHDF1 axis-modified UCHL5 aggravates atherosclerosis by activating the NLRP3 inflammasome. Exp. Cell Res. 2023, 427, 113587. https://doi.org/10.1016/j.yexcr.2023.113587.
- 87.
Li, H.; Zhao, Q.; Liu, D.; Zhou, B.; Liao, F.; Chen, L. Cathepsin B aggravates atherosclerosis in ApoE-deficient mice by modulating vascular smooth muscle cell pyroptosis through NF-κB / NLRP3 signaling pathway. PLoS ONE 2024, 19, e0294514. https://doi.org/10.1371/journal.pone.0294514.
- 88.
You, L.; Zheng, Y.; Yang, J.; Hou, Q.; Wang, L.; Zhang, Y.; Zhao, C.; Xie, R. LncRNA MDRL Mitigates Atherosclerosis through miR-361/SQSTM1/NLRP3 Signaling. Mediat. Inflamm. 2022, 2022, 5463505. https://doi.org/10.1155/2022/5463505.
- 89.
Zhang, H.; Gong, X.; Ni, S.; Wang, Y.; Zhu, L.; Ji, N. C1q/TNF-related protein-9 attenuates atherosclerosis through AMPK-NLRP3 inflammasome singling pathway. Int. Immunopharmacol. 2019, 77, 105934. https://doi.org/10.1016/j.intimp.2019.105934.
- 90.
Borborema, M.E.A.; Crovella, S.; Oliveira, D.; de Azevêdo Silva, J. Inflammasome activation by NLRP1 and NLRC4 in patients with coronary stenosis. Immunobiology 2020, 225, 151940. https://doi.org/10.1016/j.imbio.2020.151940.
- 91.
Bleda, S.; de Haro, J.; Varela, C.; Ferruelo, A.; Acin, F. Elevated levels of triglycerides and vldl-cholesterol provoke activation of nlrp1 inflammasome in endothelial cells. Int. J. Cardiol. 2016, 220, 52–55. https://doi.org/10.1016/j.ijcard.2016.06.193.
- 92.
Bleda, S.; de Haro, J.; Varela, C.; Esparza, L.; Ferruelo, A.; Acin, F. NLRP1 inflammasome, and not NLRP3, is the key in the shift to proinflammatory state on endothelial cells in peripheral arterial disease. Int. J. Cardiol. 2014, 172, e282–e284. https://doi.org/10.1016/j.ijcard.2013.12.201.
- 93.
Ge, J.Y.; Yan, X.J.; Yang, J.; Jin, H.; Sun, Z.K.; Guo, J.L.; Zhu, Y.; Wang, F.F. LINC00346 regulates NLRP1-mediated pyroptosis and autophagy via binding to microRNA-637 in vascular endothelium injury. Cell. Signal. 2023, 109, 110740. https://doi.org/10.1016/j.cellsig.2023.110740.
- 94.
Zeng, W.; Wu, D.; Sun, Y.; Suo, Y.; Yu, Q.; Zeng, M.; Gao, Q.; Yu, B.; Jiang, X.; Wang, Y. The selective NLRP3 inhibitor MCC950 hinders atherosclerosis development by attenuating inflammation and pyroptosis in macrophages. Sci. Rep. 2021, 11, 19305. https://doi.org/10.1038/s41598-021-98437-3.
- 95.
Tapia-Abellán, A.; Angosto-Bazarra, D.; Martínez-Banaclocha, H.; de Torre-Minguela, C.; Cerón-Carrasco, J.P.; Pérez-Sánchez, H.; Arostegui, J.I.; Pelegrin, P. MCC950 closes the active conformation of NLRP3 to an inactive state. Nat. Chem. Biol. 2019, 15, 560–564. https://doi.org/10.1038/s41589-019-0278-6.
- 96.
Chen, S.; Wang, Y.; Pan, Y.; Liu, Y.; Zheng, S.; Ding, K.; Mu, K.; Yuan, Y.; Li, Z.; Song, H.; et al. Novel Role for Tranilast in Regulating NLRP3 Ubiquitination, Vascular Inflammation, and Atherosclerosis. J. Am. Heart Assoc. 2020, 9, e015513. https://doi.org/10.1161/jaha.119.015513.
- 97.
Traughber, C.A.; Iacano, A.J.; Neupane, K.; Khan, M.R.; Opoku, E.; Nunn, T.; Prince, A.; Sangwan, N.; Hazen, S.L.; Smith, J.D.; et al. Impavido attenuates inflammation, reduces atherosclerosis, and alters gut microbiota in hyperlipidemic mice. iScience 2023, 26, 106453. https://doi.org/10.1016/j.isci.2023.106453.
- 98.
Luo, R.; Ma, M.; Wang, D.; Luo, L.; Xu, X.; Huang, L.; Wang, F.; Kuang, G.; Liu, H.; Ni, R.; et al. GSK461364 Inhibits NLRP3 Inflammasome by Targeting NEK7 Phosphorylation. Adv. Sci 2025, 12, e04816. https://doi.org/10.1002/advs.202504816.
- 99.
Wang, L.; Zhao, X.; Ding, J.; Liu, Y.; Liu, H.; Zheng, L.; Zhao, H.; Sun, Z.; Li, K.; Cai, J.; et al. Oridonin attenuates the progression of atherosclerosis by inhibiting NLRP3 and activating Nrf2 in apolipoprotein E-deficient mice. Inflammopharmacology 2023, 31, 1993–2005. https://doi.org/10.1007/s10787-023-01161-9.
- 100.
Liu, P.; Wang, Y.; Tian, K.; Bai, X.; Wang, Y.; Wang, Y. Artesunate inhibits macrophage-like phenotype switching of vascular smooth muscle cells and attenuates vascular inflammatory injury in atherosclerosis via NLRP3. Biomed. Pharmacother. 2024, 172, 116255. https://doi.org/10.1016/j.biopha.2024.116255.
- 101.
Zhao, J.; Wang, Z.; Yuan, Z.; Lv, S.; Su, Q. Baicalin ameliorates atherosclerosis by inhibiting NLRP3 inflammasome in apolipoprotein E-deficient mice. Diabetes Vasc. Dis. Res. 2020, 17, 1479164120977441. https://doi.org/10.1177/1479164120977441.
- 102.
Wen, J.; Chang, Y.; Huo, S.; Li, W.; Huang, H.; Gao, Y.; Lin, H.; Zhang, J.; Zhang, Y.; Zuo, Y.; et al. Tanshinone IIA attenuates atherosclerosis via inhibiting NLRP3 inflammasome activation. Aging 2020, 13, 910–932. https://doi.org/10.18632/aging.202202.
- 103.
Kim, N.H.; Kim, H.K.; Lee, J.H.; Jo, S.I.; Won, H.M.; Lee, G.S.; Lee, H.S.; Nam, K.W.; Kim, W.J.; Han, M.D. Juglone Suppresses LPS-induced Inflammatory Responses and NLRP3 Activation in Macrophages. Molecules 2020, 25, 3104. https://doi.org/10.3390/molecules25133104.
- 104.
Li, Y.; Zhang, Y.; Lu, J.; Yin, Y.; Xie, J.; Xu, B. Anti-inflammatory mechanisms and research progress of colchicine in atherosclerotic therapy. J. Cell. Mol. Med. 2021, 25, 8087–8094. https://doi.org/10.1111/jcmm.16798.
- 105.
Li, H.; Xiao, L.; He, H.; Zeng, H.; Liu, J.; Jiang, C.; Mei, G.; Yu, J.; Chen, H.; Yao, P.; et al. Quercetin Attenuates Atherosclerotic Inflammation by Inhibiting Galectin-3-NLRP3 Signaling Pathway. Mol. Nutr. Food Res. 2021, 65, e2000746. https://doi.org/10.1002/mnfr.202000746.
- 106.
Ruan, H.; Zeng, X.; Shen, S. Mechanism of Curcumin Inhibiting NLRP3 Inflammatory Body and Improving Atherosclerotic Endothelial Cell Injury. Discov. Med. 2024, 36, 121–128. https://doi.org/10.24976/Discov.Med.202436180.11.
- 107.
Liu, Y.; Wu, H.; Wang, T.; Shi, X.; He, H.; Huang, H.; Yang, Y.; Dai, M. Paeonol reduces microbial metabolite α-hydroxyisobutyric acid to alleviate the ROS/TXNIP/NLRP3 pathway-mediated endothelial inflammation in atherosclerosis mice. Chin. J. Nat. Med. 2023, 21, 759–774. https://doi.org/10.1016/s1875-5364(23)60506-0.
- 108.
Zhang, K.; Liu, J.; Jiang, Y.; Sun, S.; Wang, R.; Sun, J.; Ma, C.; Chen, Y.; Wang, W.; Hou, X.; et al. Sorbremnoids A and B: NLRP3 Inflammasome Inhibitors Discovered from Spatially Restricted Crosstalk of Biosynthetic Pathways. J. Am. Chem. Soc. 2024, 146, 18172–18183. https://doi.org/10.1021/jacs.4c06538.
- 109.
Lv, Q.; Zhang, Y.; Wang, J.; Lin, W.; Xie, Y.; Yang, H.; Yin, X.; Zhu, Z.; Cui, Y.; Hu, Y.; et al. Dehydrocostus Lactone Effectively Alleviates Inflammatory Diseases by Covalently and Irreversibly Targeting NLRP3. MedComm 2025, 6, e70367. https://doi.org/10.1002/mco2.70367.
- 110.
Xiaoyang, C.; Yijun, C.; Chenguang, Z.; Wanying, D.; Zijun, C.; Jun, W.; Xuegong, X.; Wei, W.; Chun, L. Resibufogenin protects against atherosclerosis in ApoE(-/-) mice through blocking NLRP3 inflammasome assembly. J. Adv. Res. 2025. https://doi.org/10.1016/j.jare.2025.04.029.
- 111.
Fu, Y.; Yuan, J.; Sang, F.; Shao, M.; Yan, S.; Li, L.; Zhu, R.; Wang, Z. Biejiajian Pill Ameliorates Diabetes-Associated Atherosclerosis through Inhibition of the NLRP3 Inflammasome. Evid. Based Complement. Altern. Med. 2022, 2022, 9131178. https://doi.org/10.1155/2022/9131178.
- 112.
Hou, C.; Jiang, X.; Sheng, W.; Zhang, Y.; Lin, Q.; Hong, S.; Zhao, J.; Wang, T.; Ye, X. Xinmaikang (XMK) tablets alleviate atherosclerosis by regulating the SREBP2-mediated NLRP3/ASC/Caspase-1 signaling pathway. J. Ethnopharmacol. 2024, 319, 117240. https://doi.org/10.1016/j.jep.2023.117240.
- 113.
Tian, Y.; Ling, X.Y.; Chen, D.L.; Zhang, X.Q.; Qiu, C.M. Interleukin-36 receptor antagonist attenuates atherosclerosis development by inhibiting NLRP3 inflammasome. J. Cell. Physiol. 2020, 235, 9992–9996. https://doi.org/10.1002/jcp.29813.
- 114.
Ma, M.H.; Li, F.F.; Li, W.F.; Zhao, H.; Jiang, M.; Yu, Y.Y.; Dong, Y.C.; Zhang, Y.X.; Li, P.; Bu, W.J.; et al. Repurposing nitazoxanide as a novel anti-atherosclerotic drug based on mitochondrial uncoupling mechanisms. Br. J. Pharmacol. 2023, 180, 62–79. https://doi.org/10.1111/bph.15949.
- 115.
Zhang, X.; Wang, Z.; Li, X.; Chen, J.; Yu, Z.; Li, X.; Sun, C.; Hu, L.; Wu, M.; Liu, L. Polydatin protects against atherosclerosis by activating autophagy and inhibiting pyroptosis mediated by the NLRP3 inflammasome. J. Ethnopharmacol. 2023, 309, 116304. https://doi.org/10.1016/j.jep.2023.116304.
- 116.
Chen, C.C.; Lii, C.K.; Liu, K.L.; Lin, Y.L.; Lo, C.W.; Li, C.C.; Yang, Y.C.; Chen, H.W. Andrographolide Attenuates Oxidized LDL-Induced Activation of the NLRP3 Inflammasome in Bone Marrow-Derived Macrophages and Mitigates HFCCD-Induced Atherosclerosis in Mice. Am. J. Chin. Med. 2023, 51, 2175–2193. https://doi.org/10.1142/s0192415x23500933.
- 117.
Weng, X.; Luo, X.; Dai, X.; Lv, Y.; Zhang, S.; Bai, X.; Bao, X.; Wang, Y.; Zhao, C.; Zeng, M.; et al. Apigenin inhibits macrophage pyroptosis through regulation of oxidative stress and the NF-κB pathway and ameliorates atherosclerosis. Phytother. Res. 2023, 37, 5300–5314. https://doi.org/10.1002/ptr.7962.
- 118.
Cheng, Y.; Pan, X.; Wang, J.; Li, X.; Yang, S.; Yin, R.; Ma, A.; Zhu, X. Fucoidan Inhibits NLRP3 Inflammasome Activation by Enhancing p62/SQSTM1-Dependent Selective Autophagy to Alleviate Atherosclerosis. Oxidative Med. Cell. Longev. 2020, 2020, 3186306. https://doi.org/10.1155/2020/3186306.
- 119.
Varghese, J.F.; Patel, R.; Singh, M.; Yadav, U.C.S. Fisetin Prevents Oxidized Low-density Lipoprotein-Induced Macrophage Foam Cell Formation. J. Cardiovasc. Pharmacol. 2021, 78, e729–e737. https://doi.org/10.1097/fjc.0000000000001096.
- 120.
He, J.; Deng, Y.; Ren, L.; Jin, Z.; Yang, J.; Yao, F.; Liu, Y.; Zheng, Z.; Chen, D.; Wang, B.; et al. Isoliquiritigenin from licorice flavonoids attenuates NLRP3-mediated pyroptosis by SIRT6 in vascular endothelial cells. J. Ethnopharmacol. 2023, 303, 115952. https://doi.org/10.1016/j.jep.2022.115952.
- 121.
Peng, Z.; Zhan, H.; Shao, Y.; Xiong, Y.; Zeng, L.; Zhang, C.; Liu, Z.; Huang, Z.; Su, H.; Yang, Z. 13-Methylberberine improves endothelial dysfunction by inhibiting NLRP3 inflammasome activation via autophagy induction in human umbilical vein endothelial cells. Chin. Med. 2020, 15, 8. https://doi.org/10.1186/s13020-020-0286-1.
- 122.
Jiang, X.; Ma, C.; Gao, Y.; Cui, H.; Zheng, Y.; Li, J.; Zong, W.; Zhang, Q. Tongxinluo attenuates atherosclerosis by inhibiting ROS/NLRP3/caspase-1-mediated endothelial cell pyroptosis. J. Ethnopharmacol. 2023, 304, 116011. https://doi.org/10.1016/j.jep.2022.116011.
- 123.
Delfos, L.; Depuydt, M.A.C.; Chemaly, M.; Coyle, S.; Schaftenaar, F.H.; van Santbrink, P.J.; Lindenbergh, P.P.; Bernabé Kleijn, M.N.A.; Costello, C.; Power, C.A.; et al. NLRP3 Inflammasome Inhibition by the Novel Bispecific Antibody InflamAb Attenuates Atherosclerosis in Apolipoprotein E-Deficient Mice. JACC Basic Transl. Sci. 2025, 10, 826–840. https://doi.org/10.1016/j.jacbts.2024.12.012.
- 124.
Yang, H.; Song, R.; Xie, Y.; Qian, Q.; Wu, Z.; Han, S.; Li, X. Apple Polyphenol Extract Ameliorates Atherosclerosis and Associated Cognitive Impairment through Alleviating Neuroinflammation by Weakening TLR4 Signaling and NLRP3 Inflammasome in High-Fat/Cholesterol Diet-Fed LDLR(-/-) Male Mice. J. Agric. Food Chem. 2023, 71, 15506–15521. https://doi.org/10.1021/acs.jafc.3c01966.
- 125.
Ma, Q.; Yang, Q.; Chen, J.; Yu, C.; Zhang, L.; Zhou, W.; Chen, M. Salvianolic Acid A Ameliorates Early-Stage Atherosclerosis Development by Inhibiting NLRP3 Inflammasome Activation in Zucker Diabetic Fatty Rats. Molecules 2020, 25, 1089. https://doi.org/10.3390/molecules25051089.
- 126.
Zhang, Y.; Feng, X.; Du, M.; Ding, J.; Liu, P. Salvianolic acid B attenuates the inflammatory response in atherosclerosis by regulating MAPKs/ NF-κB signaling pathways in LDLR-/- mice and RAW264.7 cells. Int. J. Immunopathol. Pharmacol. 2022, 36, 3946320221079468. https://doi.org/10.1177/03946320221079468.
- 127.
Lu, Z.; Liu, L.; Zhao, S.; Zhao, J.; Li, S.; Li, M. Apigenin attenuates atherosclerosis and non-alcoholic fatty liver disease through inhibition of NLRP3 inflammasome in mice. Sci. Rep. 2023, 13, 7996. https://doi.org/10.1038/s41598-023-34654-2.
- 128.
Luo, J.; Wang, X.; Jiang, X.; Liu, C.; Li, Y.; Han, X.; Zuo, X.; Li, Y.; Li, N.; Xu, Y.; et al. Rutaecarpine derivative R3 attenuates atherosclerosis via inhibiting NLRP3 inflammasome-related inflammation and modulating cholesterol transport. FASEB J. 2020, 34, 1398–1411. https://doi.org/10.1096/fj.201900903RRR.
- 129.
Jiang, X.; Li, Y.; Wang, W.; Han, X.; Han, J.; Chen, M.; Zhang, J.; Wang, C.; Li, S.; Luo, J.; et al. Nuclear Factor Erythroid 2 Related Factor 2 Activator JC-5411 Inhibits Atherosclerosis Through Suppression of Inflammation and Regulation of Lipid Metabolism. Front. Pharmacol. 2020, 11, 532568. https://doi.org/10.3389/fphar.2020.532568.
- 130.
Liu, M.; Luo, G.; Liu, T.; Yang, T.; Wang, R.; Ren, W.; Liu, P.; Lai, X.; Zhou, H.; Yang, S. Zhilong Huoxue Tongyu Capsule Alleviated the Pyroptosis of Vascular Endothelial Cells Induced by ox-LDL through miR-30b-5p/NLRP3. Evid. Based Complement. Altern. Med. 2022, 2022, 3981350. https://doi.org/10.1155/2022/3981350.
- 131.
Liu, M.; Wang, R.; Chen, M.; Hu, Z.; Han, M.; Mazhar, M.; Xue, J.; Zou, Y.; Wu, Q.; Yang, S. Zhilong Huoxue Tongyu capsule inhibits rabbit model of hyperlipidemia and atherosclerosis through NF-κB/NLRP3 signaling pathway. Heliyon 2023, 9, e20026. https://doi.org/10.1016/j.heliyon.2023.e20026.
- 132.
Luo, Q.; Dai, L.; Li, J.; Chen, H.; Hao, Y.; Li, Q.; Pan, L.; Song, C.; Qian, Z.; Chen, M. Intracellular and extracellular synergistic therapy for restoring macrophage functions via anti-CD47 antibody-conjugated bifunctional nanoparticles in atherosclerosis. Bioact. Mater. 2024, 34, 326–337. https://doi.org/10.1016/j.bioactmat.2023.12.024.
- 133.
Jia, X.; Bai, X.; Yang, X.; Wang, L.; Lu, Y.; Zhu, L.; Zhao, Y.; Cheng, W.; Shu, M.; Mei, Q.; et al. VCAM-1-binding peptide targeted cationic liposomes containing NLRP3 siRNA to modulate LDL transcytosis as a novel therapy for experimental atherosclerosis. Metabolism 2022, 135, 155274. https://doi.org/10.1016/j.metabol.2022.155274.
- 134.
Ni, H.; Zhou, H.; Liang, X.; Ge, Y.; Chen, H.; Liu, J.; Wang, B.; Chen, H.; Zhang, Y.; Luo, S.; et al. Reactive Oxygen Species-Responsive Nanoparticle Delivery of Small Interfering Ribonucleic Acid Targeting Olfactory Receptor 2 for Atherosclerosis Theranostics. ACS Nano 2024, 18, 23599–23614. https://doi.org/10.1021/acsnano.4c07988.
- 135.
Li, D.; Chen, J.; Lu, Y.; Yan, X.; Yang, X.; Zhang, F.; Tang, Y.; Cao, M.; Wang, J.; Pan, M.; et al. Codelivery of Dual Gases with Metal-Organic Supramolecular Cage-Based Microenvironment-Responsive Nanomedicine for Atherosclerosis Therapy. Small 2024, 20, e2402673. https://doi.org/10.1002/smll.202402673.
- 136.
Anghelache, M.; Voicu, G.; Anton, R.; Safciuc, F.; Boteanu, D.; Deleanu, M.; Turtoi, M.; Simionescu, M.; Manduteanu, I.; Calin, M. Inflammation resolution-based treatment of atherosclerosis using biomimetic nanocarriers loaded with specialized pro-resolving lipid mediators. Mater. Today Bio 2025, 32, 101733. https://doi.org/10.1016/j.mtbio.2025.101733.
- 137.
Yu, W.; Jiang, W.; Wu, W.; Wang, G.; Zhao, D.; Yan, C.; Lin, P. Combining idebenone and rosuvastatin prevents atherosclerosis by suppressing oxidative stress and NLRP3 inflammasome activation. Eur. J. Pharmacol. 2023, 955, 175911. https://doi.org/10.1016/j.ejphar.2023.175911.
- 138.
Hu, S.; Wan, X.; Li, X.; Wang, X. Aerobic exercise alleviates pyroptosis-related diseases by regulating NLRP3 inflammasome. Front. Physiol. 2022, 13, 965366. https://doi.org/10.3389/fphys.2022.965366.
- 139.
Tardif, J.C.; Kouz, S.; Waters, D.D.; Bertrand, O.F.; Diaz, R.; Maggioni, A.P.; Pinto, F.J.; Ibrahim, R.; Gamra, H.; Kiwan, G.S.; et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N. Engl. J. Med. 2019, 381, 2497–2505. https://doi.org/10.1056/NEJMoa1912388.
- 140.
Nidorf, S.M.; Fiolet, A.T.L.; Mosterd, A.; Eikelboom, J.W.; Schut, A.; Opstal, T.S.J.; The, S.H.K.; Xu, X.F.; Ireland, M.A.; Lenderink, T.; et al. Colchicine in Patients with Chronic Coronary Disease. N. Engl. J. Med. 2020, 383, 1838–1847. https://doi.org/10.1056/NEJMoa2021372.
- 141.
Yang, Y.; Li, X.; Chen, G.; Xian, Y.; Zhang, H.; Wu, Y.; Yang, Y.; Wu, J.; Wang, C.; He, S.; et al. Traditional Chinese Medicine Compound (Tongxinluo) and Clinical Outcomes of Patients with Acute Myocardial Infarction: The CTS-AMI Randomized Clinical Trial. JAMA 2023, 330, 1534–1545. https://doi.org/10.1001/jama.2023.19524.

This work is licensed under a Creative Commons Attribution 4.0 International License.


