Mitochondria regulate cardiac energy metabolism, calcium buffering, reactive oxygen species (ROS) signaling, and programmed cell death, playing a vital role in maintaining heart function and its response to physiological stress. Disruptions in mitochondrial quality control, dynamics, and metabolic signaling contribute fundamentally to the pathogenesis of heart failure, ischemic injury, cardiomyopathy and atrial fibrillation. Over the past five years, growing evidence has linked mitochondrial dysfunction to many cardiac pathologies, spurring research into five key areas: (1) molecular mechanisms, (2) mitochondrial biomarkers, (3) mitochondria-targeted therapies, (4) disease-specific insights, and (5) clinical trials. Below we summarize recent scientific and clinical progress in each area, highlighting emerging mechanisms and especially promising therapeutic directions.
- Open Access
- Review
Mitochondria in Cardiac Disease: Mechanisms, Therapeutic Targets, and Clinical Progress
- Edric X. Zhang †,
- Lucas J. Wang †,
- Shen Hu *
Author Information
Received: 03 Sep 2025 | Revised: 12 Nov 2025 | Accepted: 13 Dec 2025 | Published: 14 Apr 2026
Abstract
Graphical Abstract
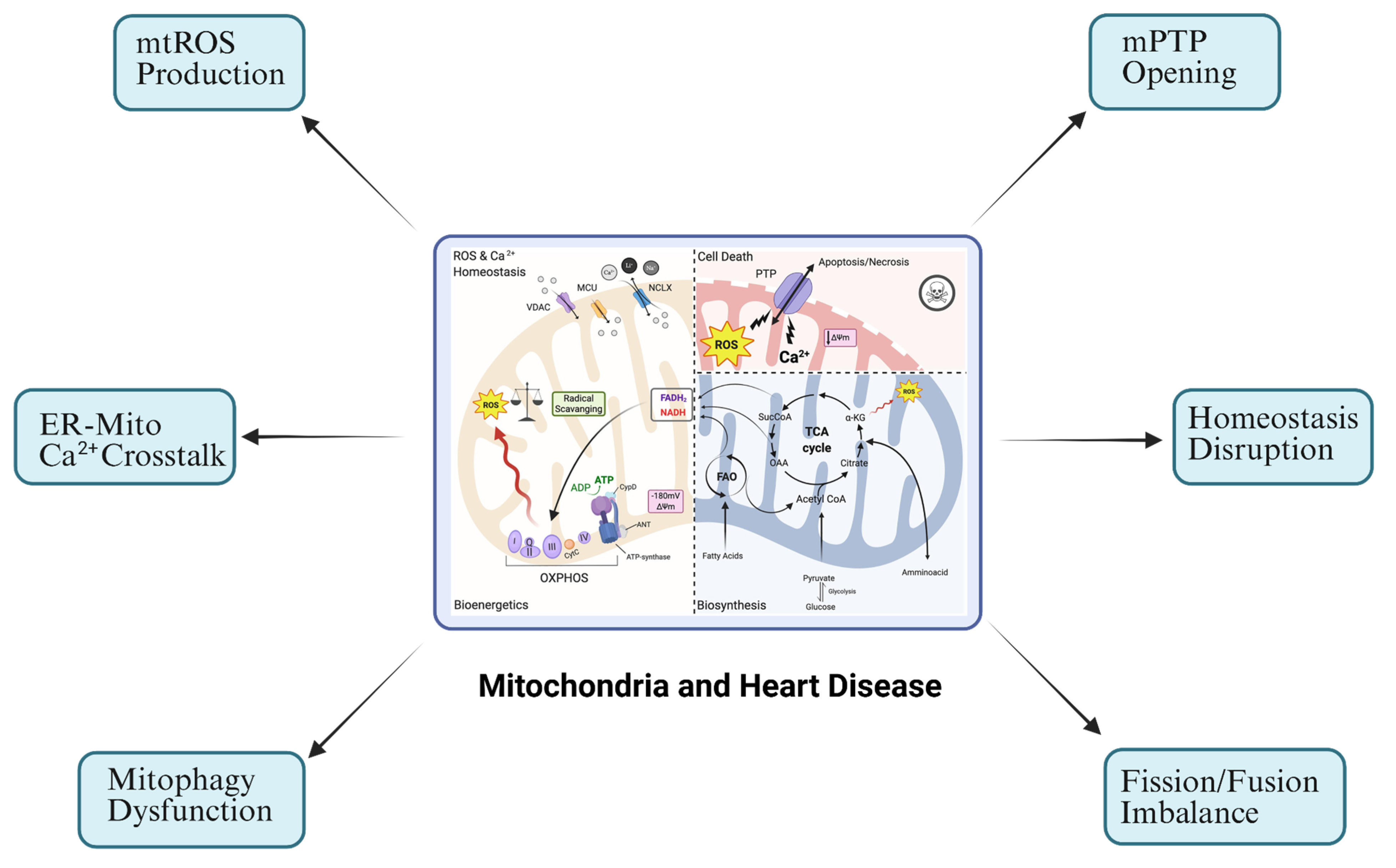
Keywords
mitochondria | reactive oxygen species (ROS) | heart failure | metabolism | therapeutic strategies
References
- 1.
Nunnari, J.; Suomalainen, A. Mitochondria: In Sickness and in Health. Cell 2012, 148, 1145–1159. https://doi.org/10.1016/j.cell.2012.02.035.
- 2.
Tokuyama, T.; Yanagi, S. Role of Mitochondrial Dynamics in Heart Diseases. Genes 2023, 14, 1876. https://doi.org/10.3390/genes14101876.
- 3.
Hinton, A.; Claypool, S.M.; Neikirk, K.; Senoo, N.; Wanjalla, C.N.; Kirabo, A.; Williams, C.R. Mitochondrial Structure and Function in Human Heart Failure. Circ. Res. 2024, 135, 372–396. https://doi.org/10.1161/CIRCRESAHA.124.323800.
- 4.
Ravindran, S.; Rau, C.D. The Multifaceted Role of Mitochondria in Cardiac Function: Insights and Approaches. Cell Commun. Signal. 2024, 22, 525. https://doi.org/10.1186/s12964-024-01899-x.
- 5.
Yang, H.-M. Mitochondrial Dysfunction in Cardiovascular Diseases. Int. J. Mol. Sci. 2025, 26, 1917. https://doi.org/10.3390/ijms26051917.
- 6.
Gallo, G.; Rubattu, S.; Volpe, M. Mitochondrial Dysfunction in Heart Failure: From Pathophysiological Mechanisms to Therapeutic Opportunities. Int. J. Mol. Sci. 2024, 25, 2667. https://doi.org/10.3390/ijms25052667.
- 7.
Werbner, B.; Tavakoli-Rouzbehani, O.M.; Fatahian, A.N.; Boudina, S. The Dynamic Interplay between Cardiac Mitochondrial Health and Myocardial Structural Remodeling in Metabolic Heart Disease, Aging, and Heart Failure. J. Cardiovasc. Aging 2023, 3, 9. https://doi.org/10.20517/jca.2022.42.
- 8.
Schwemmlein, J.; Maack, C.; Bertero, E. Mitochondria as Therapeutic Targets in Heart Failure. Curr. Heart Fail. Rep. 2022, 19, 27–37. https://doi.org/10.1007/s11897-022-00539-0.
- 9.
Xia, D.; Liu, Y.; Wu, P.; Wei, D. Current Advances of Mitochondrial Dysfunction and Cardiovascular Disease and Promising Therapeutic Strategies. Am. J. Pathol. 2023, 193, 1485–1500. https://doi.org/10.1016/j.ajpath.2023.06.013.
- 10.
Zong, Y.; Li, H.; Liao, P.; Chen, L.; Pan, Y.; Zheng, Y.; Zhang, C.; Liu, D.; Zheng, M.; Gao, J. Mitochondrial Dysfunction: Mechanisms and Advances in Therapy. Signal Transduct. Target. Ther. 2024, 9, 124. https://doi.org/10.1038/s41392-024-01839-8.
- 11.
Fonseka, O.; Gare, S.R.; Chen, X.; Zhang, J.; Alatawi, N.H.; Ross, C.; Liu, W. Molecular Mechanisms Underlying Heart Failure and Their Therapeutic Potential. Cells 2025, 14, 324. https://doi.org/10.3390/cells14050324.
- 12.
Shi, J.; Yu, Y.; Yuan, H.; Li, Y.; Xue, Y. Mitochondrial Dysfunction in AMI: Mechanisms and Therapeutic Perspectives. J. Transl. Med. 2025, 23, 418. https://doi.org/10.1186/s12967-025-06406-5.
- 13.
Huang, Y.; Zhou, B. Mitochondrial Dysfunction in Cardiac Diseases and Therapeutic Strategies. Biomedicines 2023, 11, 1500. https://doi.org/10.3390/biomedicines11051500.
- 14.
Sun, Q.; Karwi, Q.G.; Wong, N.; Lopaschuk, G.D. Advances in Myocardial Energy Metabolism: Metabolic Remodelling in Heart Failure and Beyond. Cardiovasc. Res. 2024, 120, 1996–2016. https://doi.org/10.1093/cvr/cvae231.
- 15.
Parker, A.M.; Lees, J.G.; Murray, A.J.; Velagic, A.; Lim, S.Y.; De Blasio, M.J.; Ritchie, R.H. Precision Medicine: Therapeutically Targeting Mitochondrial Alterations in Heart Failure. JACC Basic Transl. Sci. 2025, 10, 101345. https://doi.org/10.1016/j.jacbts.2025.101345.
- 16.
Zhang, M.; Zhang, T.; Zou, R.; He, K.; Huang, R.; Feng, J.; Hu, J.; Ge, T.; Fan, X.; Zhou, H.; et al. Mitochondrial Quality Control as a Therapeutic Target in Cardiovascular Disease: Mechanistic Insights and Future Directions. J. Transl. Intern. Med. 2025, 13, 211–240. https://doi.org/10.1515/jtim-2025-0030.
- 17.
Zhang, X.; Shao, S.; Li, Q.; Wang, Y.; Kong, M.; Zhang, C. Roles of Autophagy, Mitophagy, and Mitochondria in Left Ventricular Remodeling after Myocardial Infarction. Rev. Cardiovasc. Med. 2025, 26, 28195. https://doi.org/10.31083/RCM28195.
- 18.
Lazou, A.; Ramachandra, C.J. Protecting the Mitochondria in Cardiac Disease. Int. J. Mol. Sci. 2022, 23, 8115. https://doi.org/10.3390/ijms23158115.
- 19.
Ravindran, R.; Gustafsson, Å.B. Mitochondrial Quality Control in Cardiomyocytes: Safeguarding the Heart against Disease and Ageing. Nat. Rev. Cardiol. 2025, 22, 798–813. https://doi.org/10.1038/s41569-025-01142-1.
- 20.
Corral-Acero, J.; Margara, F.; Marciniak, M.; Rodero, C.; Loncaric, F.; Feng, Y.; Gilbert, A.; Fernandes, J.F.; Bukhari, H.A.; Wajdan, A.; et al. The ‘Digital Twin’ to Enable the Vision of Precision Cardiology. Eur. Heart J. 2020, 41, 4556–4564. https://doi.org/10.1093/eurheartj/ehaa159.
- 21.
Colman, M.A.; Alvarez-Lacalle, E.; Echebarria, B.; Sato, D.; Sutanto, H.; Heijman, J. Multi-Scale Computational Modeling of Spatial Calcium Handling from Nanodomain to Whole-Heart: Overview and Perspectives. Front. Physiol. 2022, 13, 836622. https://doi.org/10.3389/fphys.2022.836622.
- 22.
Qu, Z.; Yan, D.; Song, Z. Modeling Calcium Cycling in the Heart: Progress, Pitfalls, and Challenges. Biomolecules 2022, 12, 1686. https://doi.org/10.3390/biom12111686.
- 23.
Bertero, E.; Popoiu, T.-A.; Maack, C. Mitochondrial Calcium in Cardiac Ischemia/Reperfusion Injury and Cardioprotection. Basic Res. Cardiol. 2024, 119, 569–585. https://doi.org/10.1007/s00395-024-01060-2.
- 24.
Bernardi, P.; Gerle, C.; Halestrap, A.P.; Jonas, E.A.; Karch, J.; Mnatsakanyan, N.; Pavlov, E.; Sheu, S.-S.; Soukas, A.A. Identity, Structure, and Function of the Mitochondrial Permeability Transition Pore: Controversies, Consensus, Recent Advances, and Future Directions. Cell Death Differ. 2023, 30, 1869–1885. https://doi.org/10.1038/s41418-023-01187-0.
- 25.
He, J.; Liu, D.; Zhao, L.; Zhou, D.; Rong, J.; Zhang, L.; Xia, Z. Myocardial Ischemia/Reperfusion Injury: Mechanisms of Injury and Implications for Management (Review). Exp. Ther. Med. 2022, 23, 430. https://doi.org/10.3892/etm.2022.11357.
- 26.
Robichaux, D.J.; Harata, M.; Murphy, E.; Karch, J. Mitochondrial Permeability Transition Pore-Dependent Necrosis. J. Mol. Cell. Cardiol. 2023, 174, 47–55. https://doi.org/10.1016/j.yjmcc.2022.11.003.
- 27.
Lopaschuk, G.D.; Karwi, Q.G.; Tian, R.; Wende, A.R.; Abel, E.D. Cardiac Energy Metabolism in Heart Failure. Circ. Res. 2021, 128, 1487–1513. https://doi.org/10.1161/CIRCRESAHA.121.318241.
- 28.
Vue, Z.; Ajayi, P.T.; Neikirk, K.; Murphy, A.C.; Prasad, P.; Jenkins, B.C.; Vang, L.; Garza-Lopez, E.; Mungai, M.; Marshall, A.G.; et al. Human Heart Failure Alters Mitochondria and Fiber 3D Structure Triggering Metabolic Shifts. Biorxiv 2023. https://doi.org/10.1101/2023.11.28.569095.
- 29.
Wang, B.; Dai, L.; Liang, H.; He, J.; Zhou, J.; Guan, Y.; Wang, H. Mitochondrial Ultrastructural Pathology in Diabetic Cardiomyopathy: Integrated Analysis via Scanning Electron Microscopy and 3D Visualization Imaging. Cardiovasc. Diabetol. 2025, 24, 331. https://doi.org/10.1186/s12933-025-02884-5.
- 30.
Jin, J.; Wei, X.; Zhi, X.; Wang, X.; Meng, D. Drp1-Dependent Mitochondrial Fission in Cardiovascular Disease. Acta Pharmacol. Sin. 2021, 42, 655–664. https://doi.org/10.1038/s41401-020-00518-y.
- 31.
Franco, A.; Li, J.; Kelly, D.P.; Hershberger, R.E.; Marian, A.J.; Lewis, R.M.; Song, M.; Dang, X.; Schmidt, A.D.; Mathyer, M.E.; et al. A Human Mitofusin 2 Mutation Can Cause Mitophagic Cardiomyopathy. eLife 2023, 12, e84235. https://doi.org/10.7554/eLife.84235.
- 32.
Piao, L.; Fang, Y.; Fisher, M.; Hamanaka, R.B.; Ousta, A.; Wu, R.; Mutlu, G.M.; Garcia, A.J.; Archer, S.L.; Sharp, W.W. Dynamin-related Protein 1 Is a Critical Regulator of Mitochondrial Calcium Homeostasis during Myocardial Ischemia/Reperfusion Injury. FASEB J. 2024, 38, e23379. https://doi.org/10.1096/fj.202301040RR.
- 33.
Zhang, J.; Zhao, Y.; Yan, L.; Tan, M.; Jin, Y.; Yin, Y.; Han, L.; Ma, X.; Li, Y.; Yang, T.; et al. Corosolic Acid Attenuates Cardiac Ischemia/Reperfusion Injury through the PHB2/PINK1/Parkin/Mitophagy Pathway. iScience 2024, 27, 110448. https://doi.org/10.1016/j.isci.2024.110448.
- 34.
Turkieh, A.; El Masri, Y.; Pinet, F.; Dubois-Deruy, E. Mitophagy Regulation Following Myocardial Infarction. Cells 2022, 11, 199. https://doi.org/10.3390/cells11020199.
- 35.
Hu, M.-M.; Shu, H.-B. Mitochondrial DNA-Triggered Innate Immune Response: Mechanisms and Diseases. Cell. Mol. Immunol. 2023, 20, 1403–1412. https://doi.org/10.1038/s41423-023-01086-x.
- 36.
VanPortfliet, J.J.; Chute, C.; Lei, Y.; Shutt, T.E.; West, A.P. Mitochondrial DNA Release and Sensing in Innate Immune Responses. Hum. Mol. Genet. 2024, 33, R80–R91. https://doi.org/10.1093/hmg/ddae031.
- 37.
Hu, D.; Cui, Y.-X.; Wu, M.-Y.; Li, L.; Su, L.-N.; Lian, Z.; Chen, H. Cytosolic DNA Sensor cGAS Plays an Essential Pathogenetic Role in Pressure Overload-Induced Heart Failure. Am. J. Physiol.-Heart Circ. Physiol. 2020, 318, H1525–H1537. https://doi.org/10.1152/ajpheart.00097.2020.
- 38.
Rech, L.; Abdellatif, M.; Pöttler, M.; Stangl, V.; Mabotuwana, N.; Hardy, S.; Rainer, P.P. Small Molecule STING Inhibition Improves Myocardial Infarction Remodeling. Life Sci. 2022, 291, 120263. https://doi.org/10.1016/j.lfs.2021.120263.
- 39.
Sun, S.-J.; Jiao, X.-D.; Chen, Z.-G.; Cao, Q.; Zhu, J.-H.; Shen, Q.-R.; Liu, Y.; Zhang, Z.; Xu, F.-F.; Shi, Y.; et al. Gasdermin-E-Mediated Pyroptosis Drives Immune Checkpoint Inhibitor-Associated Myocarditis via cGAS-STING Activation. Nat. Commun. 2024, 15, 6640. https://doi.org/10.1038/s41467-024-50996-5.
- 40.
Oka, T.; Hikoso, S.; Yamaguchi, O.; Taneike, M.; Takeda, T.; Tamai, T.; Oyabu, J.; Murakawa, T.; Nakayama, H.; Nishida, K.; et al. Mitochondrial DNA That Escapes from Autophagy Causes Inflammation and Heart Failure. Nature 2012, 485, 251–255. https://doi.org/10.1038/nature10992.
- 41.
Chen, Y.; Ye, X.; Escames, G.; Lei, W.; Zhang, X.; Li, M.; Jing, T.; Yao, Y.; Qiu, Z.; Wang, Z.; et al. The NLRP3 Inflammasome: Contributions to Inflammation-Related Diseases. Cell. Mol. Biol. Lett. 2023, 28, 51. https://doi.org/10.1186/s11658-023-00462-9.
- 42.
Higashikuni, Y.; Liu, W.; Numata, G.; Tanaka, K.; Fukuda, D.; Tanaka, Y.; Hirata, Y.; Imamura, T.; Takimoto, E.; Komuro, I.; et al. NLRP3 Inflammasome Activation Through Heart-Brain Interaction Initiates Cardiac Inflammation and Hypertrophy During Pressure Overload. Circulation 2023, 147, 338–355. https://doi.org/10.1161/CIRCULATIONAHA.122.060860.
- 43.
Nicolás-Ávila, J.A.; Lechuga-Vieco, A.V.; Esteban-Martínez, L.; Sánchez-Díaz, M.; Díaz-García, E.; Santiago, D.J.; Rubio-Ponce, A.; Li, J.L.; Balachander, A.; Quintana, J.A.; et al. A Network of Macrophages Supports Mitochondrial Homeostasis in the Heart. Cell 2020, 183, 94–109.e23. https://doi.org/10.1016/j.cell.2020.08.031.
- 44.
Bordon, Y. Macrophages Clean up to Keep the Heart Pumping. Nat. Rev. Immunol. 2020, 20, 648–649. https://doi.org/10.1038/s41577-020-00459-x.
- 45.
Zhang, K.; Wang, Y.; Chen, S.; Mao, J.; Jin, Y.; Ye, H.; Zhang, Y.; Liu, X.; Gong, C.; Cheng, X.; et al. TREM2hi Resident Macrophages Protect the Septic Heart by Maintaining Cardiomyocyte Homeostasis. Nat. Metab. 2023, 5, 129–146. https://doi.org/10.1038/s42255-022-00715-5.
- 46.
Liang, W.; Sagar, S.; Ravindran, R.; Najor, R.H.; Quiles, J.M.; Chi, L.; Diao, R.Y.; Woodall, B.P.; Leon, L.J.; Zumaya, E.; et al. Mitochondria Are Secreted in Extracellular Vesicles When Lysosomal Function Is Impaired. Nat. Commun. 2023, 14, 5031. https://doi.org/10.1038/s41467-023-40680-5.
- 47.
Iorio, R.; Petricca, S.; Mattei, V.; Delle Monache, S. Horizontal Mitochondrial Transfer as a Novel Bioenergetic Tool for Mesenchymal Stromal/Stem Cells: Molecular Mechanisms and Therapeutic Potential in a Variety of Diseases. J. Transl. Med. 2024, 22, 491. https://doi.org/10.1186/s12967-024-05047-4.
- 48.
Chu, Q.; He, D.; Xie, W.; Li, S.; Dong, Z.; Fu, X. Gelated Microvesicle–Mediated Delivery of Mesenchymal Stem Cell Mitochondria for the Treatment of Myocardial Infarction. Proc. Natl. Acad. Sci. USA 2025, 122, e2424529122. https://doi.org/10.1073/pnas.2424529122.
- 49.
Vignais, M.-L.; Levoux, J.; Sicard, P.; Khattar, K.; Lozza, C.; Gervais, M.; Mezhoud, S.; Nakhle, J.; Relaix, F.; Agbulut, O.; et al. Transfer of Cardiac Mitochondria Improves the Therapeutic Efficacy of Mesenchymal Stem Cells in a Preclinical Model of Ischemic Heart Disease. Cells 2023, 12, 582. https://doi.org/10.3390/cells12040582.
- 50.
Shen, J.; Zhang, J.-H.; Xiao, H.; Wu, J.-M.; He, K.-M.; Lv, Z.-Z.; Li, Z.-J.; Xu, M.; Zhang, Y.-Y. Mitochondria Are Transported along Microtubules in Membrane Nanotubes to Rescue Distressed Cardiomyocytes from Apoptosis. Cell Death Dis. 2018, 9, 81. https://doi.org/10.1038/s41419-017-0145-x.
- 51.
Ahmad, T.; Mukherjee, S.; Pattnaik, B.; Kumar, M.; Singh, S.; Kumar, M.; Rehman, R.; Tiwari, B.K.; Jha, K.A.; Barhanpurkar, A.P.; et al. Miro1 Regulates Intercellular Mitochondrial Transport & Enhances Mesenchymal Stem Cell Rescue Efficacy. EMBO J. 2014, 33, 994–1010. https://doi.org/10.1002/embj.201386030.
- 52.
Lin, Y.-H.; Lin, K.-L.; Wang, X.-W.; Lee, J.-J.; Wang, F.-S.; Wang, P.-W.; Lan, M.-Y.; Liou, C.-W.; Lin, T.-K. Miro1 Improves the Exogenous Engraftment Efficiency and Therapeutic Potential of Mitochondria Transfer Using Wharton’s Jelly Mesenchymal Stem Cells. Mitochondrion 2024, 76, 101856. https://doi.org/10.1016/j.mito.2024.101856.
- 53.
Blitzer, D.; Guariento, A.; Doulamis, I.P.; Shin, B.; Moskowitzova, K.; Barbieri, G.R.; Orfany, A.; Del Nido, P.J.; McCully, J.D. Delayed Transplantation of Autologous Mitochondria for Cardioprotection in a Porcine Model. Ann. Thorac. Surg. 2020, 109, 711–719. https://doi.org/10.1016/j.athoracsur.2019.06.075.
- 54.
Xiang, M.; Yang, M.; Zhang, L.; Ouyang, X.; Sarapultsev, A.; Luo, S.; Hu, D. Mitochondrial DNA Dysfunction in Cardiovascular Diseases: A Novel Therapeutic Target. Antioxidants 2025, 14, 1138. https://doi.org/10.3390/antiox14091138.
- 55.
Mengozzi, A.; Armenia, S.; De Biase, N.; Punta, L.D.; Cappelli, F.; Duranti, E.; Nannipieri, V.; Remollino, R.; Tricò, D.; Virdis, A.; et al. Circulating Mitochondrial DNA Signature in Cardiometabolic Patients. Cardiovasc. Diabetol. 2025, 24, 106. https://doi.org/10.1186/s12933-025-02656-1.
- 56.
Torp, M.; Vaage, J.; Stensløkken, K. Mitochondria-derived Damage-associated Molecular Patterns and Inflammation in the Ischemic-reperfused Heart. Acta Physiol. 2023, 237, e13920. https://doi.org/10.1111/apha.13920.
- 57.
Peng, N.; Guo, L.; Wei, Z.; Wang, X.; Zhao, L.; Kang, L.; Wang, K.; Zhou, W.; Cheng, S.; Yin, S.; et al. Platelet Mitochondrial DNA Methylation: A Novel Biomarker for Myocardial Infarction—A Preliminary Study. Int. J. Cardiol. 2024, 398, 131606. https://doi.org/10.1016/j.ijcard.2023.131606.
- 58.
Hong, Y.S.; Longchamps, R.J.; Zhao, D.; Castellani, C.A.; Loehr, L.R.; Chang, P.P.; Matsushita, K.; Grove, M.L.; Boerwinkle, E.; Arking, D.E.; et al. Mitochondrial DNA Copy Number and Incident Heart Failure: The Atherosclerosis Risk in Communities (ARIC) Study. Circulation 2020, 141, 1823–1825. https://doi.org/10.1161/CIRCULATIONAHA.120.046001.
- 59.
Qin, P.; Qin, T.; Liang, L.; Li, X.; Jiang, B.; Wang, X.; Ma, J.; Hu, F.; Zhang, M.; Hu, D. The Role of Mitochondrial DNA Copy Number in Cardiometabolic Disease: A Bidirectional Two-Sample Mendelian Randomization Study. Cardiovasc. Diabetol. 2024, 23, 45. https://doi.org/10.1186/s12933-023-02074-1.
- 60.
Iboleon-Jimenez, A.; Sánchez-Quintero, M.J.; Carmona-Segovia, A.D.M.; Sojo, B.; Fernández-Ramos, A.M.; García-Rodríguez, L.; Molina-Ramos, A.I.; García-Pinilla, J.M.; Jimenez-Navarro, M.; Ortega-Gomez, A. Circulating Mitochondrial Biomarkers in Acute Coronary Syndrome. Front. Med. 2025, 12, 1568305. https://doi.org/10.3389/fmed.2025.1568305.
- 61.
Bourgonje, A.R.; Connelly, M.A.; Van Goor, H.; Van Dijk, P.R.; Dullaart, R.P.F. Plasma Citrate Levels Are Associated with an Increased Risk of Cardiovascular Mortality in Patients with Type 2 Diabetes (Zodiac-64). J. Clin. Med. 2023, 12, 6670. https://doi.org/10.3390/jcm12206670.
- 62.
Ramalingam, A.; Budin, S.B.; Mohd Fauzi, N.; Ritchie, R.H.; Zainalabidin, S. Targeting Mitochondrial Reactive Oxygen Species-Mediated Oxidative Stress Attenuates Nicotine-Induced Cardiac Remodeling and Dysfunction. Sci. Rep. 2021, 11, 13845. https://doi.org/10.1038/s41598-021-93234-4.
- 63.
Ji, Y.; Leng, Y.; Lei, S.; Qiu, Z.; Ming, H.; Zhang, Y.; Zhang, A.; Wu, Y.; Xia, Z. The Mitochondria-Targeted Antioxidant MitoQ Ameliorates Myocardial Ischemia–Reperfusion Injury by Enhancing PINK1/Parkin-Mediated Mitophagy in Type 2 Diabetic Rats. Cell Stress Chaperones 2022, 27, 353–367. https://doi.org/10.1007/s12192-022-01273-1.
- 64.
Wong, H.-S.; Monternier, P.-A.; Brand, M.D. S1QELs Suppress Mitochondrial Superoxide/Hydrogen Peroxide Production from Site IQ without Inhibiting Reverse Electron Flow through Complex I. Free Radic. Biol. Med. 2019, 143, 545–559. https://doi.org/10.1016/j.freeradbiomed.2019.09.006.
- 65.
Dai, W.; Amoedo, N.D.; Perry, J.; Le Grand, B.; Boucard, A.; Carreno, J.; Zhao, L.; Brown, D.A.; Rossignol, R.; Kloner, R.A. Effects of OP2113 on Myocardial Infarct Size and No Reflow in a Rat Myocardial Ischemia/Reperfusion Model. Cardiovasc. Drugs Ther. 2022, 36, 217–227. https://doi.org/10.1007/s10557-020-07113-7.
- 66.
Piao, L.; Fang, Y.-H.; Hamanaka, R.B.; Mutlu, G.M.; Dezfulian, C.; Archer, S.L.; Sharp, W.W. Suppression of Superoxide-Hydrogen Peroxide Production at Site IQ of Mitochondrial Complex I Attenuates Myocardial Stunning and Improves Postcardiac Arrest Outcomes. Crit. Care Med. 2020, 48, e133–e140. https://doi.org/10.1097/CCM.0000000000004095.
- 67.
Chen, Q.; Thompson, J.; Hu, Y.; Wang, H.; Slotabec, L.; Nguyen, J.D.; Rouhi, N.; Li, J.; Lesnefsky, E.J. High-Dose Metformin Treatment to Inhibit Complex I during Early Reperfusion Protects the Aged Mouse Heart via Decreased Mitochondrial Permeability Transition Pore Opening. J. Pharmacol. Exp. Ther. 2025, 392, 100529. https://doi.org/10.1016/j.jpet.2024.100529.
- 68.
Wang, Q.; Zuurbier, C.J.; Huhn, R.; Torregroza, C.; Hollmann, M.W.; Preckel, B.; Van Den Brom, C.E.; Weber, N.C. Pharmacological Cardioprotection against Ischemia Reperfusion Injury—The Search for a Clinical Effective Therapy. Cells 2023, 12, 1432. https://doi.org/10.3390/cells12101432.
- 69.
Koay, Y.C.; Liu, R.P.; McIntosh, B.; Vigder, N.; Lauren, S.; Bai, A.Y.; Tomita, S.; Li, D.; Harney, D.; Hunter, B.; et al. The Efficacy of Risk Factor Modification Compared to NAD+ Repletion in Diastolic Heart Failure. JACC Basic Transl. Sci. 2024, 9, 733–750. https://doi.org/10.1016/j.jacbts.2024.01.011.
- 70.
Wang, D.D.; Airhart, S.E.; Zhou, B.; Shireman, L.M.; Jiang, S.; Melendez Rodriguez, C.; Kirkpatrick, J.N.; Shen, D.D.; Tian, R.; O’Brien, K.D. Safety and Tolerability of Nicotinamide Riboside in Heart Failure With Reduced Ejection Fraction. JACC Basic Transl. Sci. 2022, 7, 1183–1196. https://doi.org/10.1016/j.jacbts.2022.06.012.
- 71.
Patel, N.; Johnson, M.A.; Vapniarsky, N.; Van Brocklin, M.W.; Williams, T.K.; Youngquist, S.T.; Ford, R.; Ewer, N.; Neff, L.P.; Hoareau, G.L. Elamipretide Mitigates Ischemia-Reperfusion Injury in a Swine Model of Hemorrhagic Shock. Sci. Rep. 2023, 13, 4496. https://doi.org/10.1038/s41598-023-31374-5.
- 72.
Thompson, W.R.; Manuel, R.; Abbruscato, A.; Carr, J.; Campbell, J.; Hornby, B.; Vaz, F.M.; Vernon, H.J. Long-Term Efficacy and Safety of Elamipretide in Patients with Barth Syndrome: 168-Week Open-Label Extension Results of TAZPOWER. Genet. Med. 2024, 26, 101138. https://doi.org/10.1016/j.gim.2024.101138.
- 73.
Stealth BioTherapeutics Announces FDA Accelerated Approval of FORZINITY™ (Elamipretide) Injection, the First Therapy for Progressive and Life-Limiting Ultra-Rare Genetic Disease Barth Syndrome. Available online: https://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-first-treatment-barth-syndrome (accessed on 14 February 2026).
- 74.
Shu, H.; Hang, W.; Peng, Y.; Nie, J.; Wu, L.; Zhang, W.; Wang, D.W.; Zhou, N. Trimetazidine Attenuates Heart Failure by Improving Myocardial Metabolism via AMPK. Front. Pharmacol. 2021, 12, 707399. https://doi.org/10.3389/fphar.2021.707399.
- 75.
Xiao, Z.; Guan, L.; Shi, H.; Yu, Y.; Yu, Z.; Qin, S.; Su, Y.; Chen, R.; Li, M.; Ge, J. Trimetazidine Affects Mitochondrial Calcium Uniporter Expression to Restore Ischemic Heart Function via Reactive Oxygen Species/NFκB Pathway Inhibition. J. Cardiovasc. Pharmacol. 2023, 82, 104–116. https://doi.org/10.1097/FJC.0000000000001434.
- 76.
Nassiri, S.; Van De Bovenkamp, A.A.; Remmelzwaal, S.; Sorea, O.; De Man, F.; Handoko, M.L. Effects of Trimetazidine on Heart Failure with Reduced Ejection Fraction and Associated Clinical Outcomes: A Systematic Review and Meta-Analysis. Open Heart 2024, 11, e002579. https://doi.org/10.1136/openhrt-2023-002579.
- 77.
Anker, S.D.; Butler, J.; Filippatos, G.; Ferreira, J.P.; Bocchi, E.; Böhm, M.; Brunner–La Rocca, H.-P.; Choi, D.-J.; Chopra, V.; Chuquiure-Valenzuela, E.; et al. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N. Engl. J. Med. 2021, 385, 1451–1461. https://doi.org/10.1056/NEJMoa2107038.
- 78.
Kolijn, D.; Pabel, S.; Tian, Y.; Lódi, M.; Herwig, M.; Carrizzo, A.; Zhazykbayeva, S.; Kovács, Á.; Fülöp, G.Á.; Falcão-Pires, I.; Reusch, P.H.; et al. Empagliflozin Improves Endothelial and Cardiomyocyte Function in Human Heart Failure with Preserved Ejection Fraction via Reduced Pro-Inflammatory-Oxidative Pathways and Protein Kinase Gα Oxidation. Cardiovasc. Res. 2021, 117, 495–507. https://doi.org/10.1093/cvr/cvaa123.
- 79.
Jhund, P.S.; Kondo, T.; Butt, J.H.; Docherty, K.F.; Claggett, B.L.; Desai, A.S.; Vaduganathan, M.; Gasparyan, S.B.; Bengtsson, O.; Lindholm, D.; et al. Dapagliflozin across the Range of Ejection Fraction in Patients with Heart Failure: A Patient-Level, Pooled Meta-Analysis of DAPA-HF and DELIVER. Nat. Med. 2022, 28, 1956–1964. https://doi.org/10.1038/s41591-022-01971-4.
- 80.
Packer, M. SGLT2 Inhibitors: Role in Protective Reprogramming of Cardiac Nutrient Transport and Metabolism. Nat. Rev. Cardiol. 2023, 20, 443–462. https://doi.org/10.1038/s41569-022-00824-4.
- 81.
Nuzzi, V.; Manca, P.; Parisi, F.; Madaudo, C.; Sciacca, S.; Cannizzo, N.; Mulè, M.; Cipriani, M.G. SGLT2 Inhibitor Therapy in Patients with Advanced Heart Failure and Reduced Ejection Fraction. Curr. Probl. Cardiol. 2024, 49, 102823. https://doi.org/10.1016/j.cpcardiol.2024.102823.
- 82.
Savage, P.; Watson, C.; Coburn, J.; Cox, B.; Shahmohammadi, M.; Grieve, D.; Dixon, L. Impact of SGLT2 Inhibition on Markers of Reverse Cardiac Remodelling in Heart Failure: Systematic Review and Meta-analysis. ESC Heart Fail. 2024, 11, 3636–3648. https://doi.org/10.1002/ehf2.14993.
- 83.
Girardi, A.C.C.; Polidoro, J.Z.; Castro, P.C.; Pio-Abreu, A.; Noronha, I.L.; Drager, L.F. Mechanisms of Heart Failure and Chronic Kidney Disease Protection by SGLT2 Inhibitors in Nondiabetic Conditions. Am. J. Physiol.-Cell Physiol. 2024, 327, C525–C544. https://doi.org/10.1152/ajpcell.00143.2024.
- 84.
Nandave, M. A Comprehensive Review of SGLT2 Inhibitor’s Effects on Cardiovascular Events, Mortality, and Significant Safety Outcomes in Individuals with Type 2 Diabetes. In Sodium-glucose Cotransporter-2 (SGLT2) Inhibitors in Heart Failure; Nandave, M., Ed.; Springer Nature Singapore: Singapore, 2024; pp. 501–528. https://doi.org/10.1007/978-981-97-7568-2_9.
- 85.
Yan, Q.; Chen, X.; Yu, C.; Yin, Y. Long-Term Surrogate Cardiovascular Outcomes of SGLT2 Inhibitor Empagliflozin in Chronic Heart Failure: A Systematic Review and Meta-Analysis. BMC Cardiovasc. Disord. 2024, 24, 663. https://doi.org/10.1186/s12872-024-04316-w.
- 86.
Raja, A.; Dogar, M.-A.; Raja, S.; Shuja, M.H.; Amin, S.B.; Khelani, M.; Fatima, U.; Soomro, A.; Habiba, A.; Mustafa, I.; et al. Dapagliflozin in Acute Heart Failure Management: A Systematic Review and Meta-Analysis of Safety and Effectiveness. BMC Cardiovasc. Disord. 2024, 24, 749. https://doi.org/10.1186/s12872-024-04412-x.
- 87.
Prosperi, S.; D’Amato, A.; Labbro Francia, A.; Monosilio, S.; Cestiè, C.; Marek Iannucci, S.; Netti, L.; Angotti, D.; Filomena, D.; Mariani, M.V.; et al. Sodium-Glucose Cotransporter 2 Inhibitor Therapy in Different Scenarios of Heart Failure: An Overview of the Current Literature. Int. J. Mol. Sci. 2024, 25, 11458. https://doi.org/10.3390/ijms252111458.
- 88.
Piperis, C.; Marathonitis, A.; Anastasiou, A.; Theofilis, P.; Mourouzis, K.; Giannakodimos, A.; Tryfou, E.; Oikonomou, E.; Siasos, G.; Tousoulis, D. Multifaceted Impact of SGLT2 Inhibitors in Heart Failure Patients: Exploring Diverse Mechanisms of Action. Biomedicines 2024, 12, 2314. https://doi.org/10.3390/biomedicines12102314.
- 89.
Gao, F.M.; Ali, A.S.; Bellomo, R.; Gaca, M.; Lecamwasam, A.; Churilov, L.; Ekinci, E.I. A Systematic Review and Meta-Analysis on the Safety and Efficacy of Sodium–Glucose Cotransporter 2 Inhibitor Use in Hospitalized Patients. Diabetes Care 2024, 47, 2275–2290. https://doi.org/10.2337/dc24-0946.
- 90.
Su, A.Y.; Csere, M.M.; Shan, R.; Pasupuleti, V.; Valenzuela, G.V.; Hernandez, A.V. Comparative Efficacy and Safety of SGLT2 Inhibitor Class Members in Patients with Heart Failure and Type 2 Diabetes: A Systematic Review and Network Meta-Analysis of Randomized Controlled Trials. Diabetes Res. Clin. Pract. 2025, 224, 112219. https://doi.org/10.1016/j.diabres.2025.112219.
- 91.
Soni, R.; Pal, D.; Gupta, A.K.; Mishra, A.; Vaishnav, Y.; Jain, S.K. SGLT2 Inhibitors in Translational Medicine: A Paradigm Shift for Diabetes and Heart Health. Eur. J. Med. Chem. 2025, 298, 117977. https://doi.org/10.1016/j.ejmech.2025.117977.
- 92.
Liu, M.; Luo, X.; Hu, H.; Chen, L. Autophagy: A Major Regulatory Factor in Glucose Metabolism. Health Metab. 2025. https://doi.org/10.53941/hm.2025.100024.
- 93.
Das, B.B.; Niu, J. A Systematic Review and Meta-Analysis of the Safety and Efficacy of SGLT2 Inhibitors in Chronic Heart Failure in ACHD Patients. Am. J. Cardiovasc. Drugs 2025, 25, 231–240. https://doi.org/10.1007/s40256-024-00697-7.
- 94.
Khalid, N.; Afzal, M.A.; Abdullah, M.; Haiy, A.-U.; Shamoon, Y.; Elkattawy, S.; Laghari, M.A.; Vasudev, R.; Fayez, S.E.; Rajeswaran, Y.; et al. Efficacy and Safety of Empagliflozin in Acute Heart Failure: A Systematic Review and Meta-Analysis. Future Cardiol. 2025, 21, 495–501. https://doi.org/10.1080/14796678.2025.2499374.
- 95.
Packer, M. Critical Reanalysis of the Mechanisms Underlying the Cardiorenal Benefits of SGLT2 Inhibitors and Reaffirmation of the Nutrient Deprivation Signaling/Autophagy Hypothesis. Circulation 2022, 146, 1383–1405. https://doi.org/10.1161/CIRCULATIONAHA.122.061732.
- 96.
Packer, M. Mechanisms Leading to Differential Hypoxia-Inducible Factor Signaling in the Diabetic Kidney: Modulation by SGLT2 Inhibitors and Hypoxia Mimetics. Am. J. Kidney Dis. 2021, 77, 280–286. https://doi.org/10.1053/j.ajkd.2020.04.016.
- 97.
Huang, K.; Luo, X.; Liao, B.; Li, G.; Feng, J. Insights into SGLT2 Inhibitor Treatment of Diabetic Cardiomyopathy: Focus on the Mechanisms. Cardiovasc. Diabetol. 2023, 22, 86. https://doi.org/10.1186/s12933-023-01816-5.
- 98.
Marso, S.P.; Daniels, G.H.; Brown-Frandsen, K.; Kristensen, P.; Mann, J.F.E.; Nauck, M.A.; Nissen, S.E.; Pocock, S.; Poulter, N.R.; Ravn, L.S.; et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 311–322. https://doi.org/10.1056/NEJMoa1603827.
- 99.
Katogiannis, K.; Thymis, J.; Kousathana, F.; Pavlidis, G.; Korakas, E.; Kountouri, A.; Balampanis, K.; Prentza, V.; Kostelli, G.; Michalopoulou, H.; et al. Effects of Liraglutide, Empagliflozin and Their Combination on Left Atrial Strain and Arterial Function. Medicina 2024, 60, 395. https://doi.org/10.3390/medicina60030395.
- 100.
Liu, Y.; Li, Z.; Xu, X.; Zou, Y.; Zhang, M.; Chen, Y.; Zhu, W.; Han, B. Semaglutide Attenuates Myocardial Ischemia-Reperfusion Injury by Inhibiting Ferroptosis of Cardiomyocytes via Activation of PKC-S100A9 Axis. Front. Pharmacol. 2025, 16, 1529652. https://doi.org/10.3389/fphar.2025.1529652.
- 101.
Nissanka, N.; Moraes, C.T. Mitochondrial DNA Heteroplasmy in Disease and Targeted Nuclease-based Therapeutic Approaches. EMBO Rep. 2020, 21, e49612. https://doi.org/10.15252/embr.201949612.
- 102.
Mok, B.Y.; De Moraes, M.H.; Zeng, J.; Bosch, D.E.; Kotrys, A.V.; Raguram, A.; Hsu, F.; Radey, M.C.; Peterson, S.B.; Mootha, V.K.; et al. A Bacterial Cytidine Deaminase Toxin Enables CRISPR-Free Mitochondrial Base Editing. Nature 2020, 583, 631–637. https://doi.org/10.1038/s41586-020-2477-4.
- 103.
Mok, B.Y.; Kotrys, A.V.; Raguram, A.; Huang, T.P.; Mootha, V.K.; Liu, D.R. CRISPR-Free Base Editors with Enhanced Activity and Expanded Targeting Scope in Mitochondrial and Nuclear DNA. Nat. Biotechnol. 2022, 40, 1378–1387. https://doi.org/10.1038/s41587-022-01256-8.
- 104.
Cho, S.-I.; Lim, K.; Hong, S.; Lee, J.; Kim, A.; Lim, C.J.; Ryou, S.; Lee, J.M.; Mok, Y.G.; Chung, E.; et al. Engineering TALE-Linked Deaminases to Facilitate Precision Adenine Base Editing in Mitochondrial DNA. Cell 2024, 187, 95–109.e26. https://doi.org/10.1016/j.cell.2023.11.035.
- 105.
McFarland, R.; Hyslop, L.A.; Feeney, C.; Pillai, R.N.; Blakely, E.L.; Moody, E.; Prior, M.; Devlin, A.; Taylor, R.W.; Herbert, M.; et al. Mitochondrial Donation in a Reproductive Care Pathway for mtDNA Disease. N. Engl. J. Med. 2025, 393, 461–468. https://doi.org/10.1056/NEJMoa2503658.
- 106.
Zhang, X.; Huang, L.; Li, C.; Yang, J.; Duan, F.; Su, Q.; Zhang, Y.; Kou, M.; Zhou, X.; Guo, L.; et al. Gene Therapy Prevents Onset of Mitochondrial Cardiomyopathy in Neonatal Mice with Ndufs6 Deficiency. Cell Death Discov. 2025, 11, 249. https://doi.org/10.1038/s41420-025-02524-7.
- 107.
Liu, D.; Gao, Y.; Liu, J.; Huang, Y.; Yin, J.; Feng, Y.; Shi, L.; Meloni, B.P.; Zhang, C.; Zheng, M.; et al. Intercellular Mitochondrial Transfer as a Means of Tissue Revitalization. Signal Transduct. Target. Ther. 2021, 6, 65. https://doi.org/10.1038/s41392-020-00440-z.
- 108.
Hayashida, K.; Takegawa, R.; Shoaib, M.; Aoki, T.; Choudhary, R.C.; Kuschner, C.E.; Nishikimi, M.; Miyara, S.J.; Rolston, D.M.; Guevara, S.; et al. Mitochondrial Transplantation Therapy for Ischemia Reperfusion Injury: A Systematic Review of Animal and Human Studies. J. Transl. Med. 2021, 19, 214. https://doi.org/10.1186/s12967-021-02878-3.
- 109.
Ali Pour, P.; Hosseinian, S.; Kheradvar, A. Mitochondrial Transplantation in Cardiomyocytes: Foundation, Methods, and Outcomes. Am. J. Physiol.-Cell Physiol. 2021, 321, C489–C503. https://doi.org/10.1152/ajpcell.00152.2021.
- 110.
Chen, J.; Zhong, J.; Wang, L.; Chen, Y. Mitochondrial Transfer in Cardiovascular Disease: From Mechanisms to Therapeutic Implications. Front. Cardiovasc. Med. 2021, 8, 771298. https://doi.org/10.3389/fcvm.2021.771298.
- 111.
Borcherding, N.; Brestoff, J.R. The Power and Potential of Mitochondria Transfer. Nature 2023, 623, 283–291. https://doi.org/10.1038/s41586-023-06537-z.
- 112.
Zuo, B.; Li, X.; Xu, D.; Zhao, L.; Yang, Y.; Luan, Y.; Zhang, B. Targeting Mitochondrial Transfer: A New Horizon in Cardiovascular Disease Treatment. J. Transl. Med. 2024, 22, 1160. https://doi.org/10.1186/s12967-024-05979-x.
- 113.
Doulamis, I.P.; Tzani, A.; Alemany, V.S.; Nomoto, R.S.; Celik, A.; Recco, D.P.; Saeed, M.Y.; Guariento, A.; Plutzky, J.; Emani, S.M.; et al. Mitochondrial Transplantation Normalizes Transcriptomic and Proteomic Shift Associated with Ischemia Reperfusion Injury in Neonatal Hearts Donated after Circulatory Death. Sci. Rep. 2024, 14, 31236. https://doi.org/10.1038/s41598-024-82578-2.
- 114.
Baharvand, F.; Habibi Roudkenar, M.; Pourmohammadi-Bejarpasi, Z.; Najafi-Ghalehlou, N.; Feizkhah, A.; Bashiri Aliabadi, S.; Salari, A.; Mohammadi Roushandeh, A. Safety and Efficacy of Platelet-Derived Mitochondrial Transplantation in Ischaemic Heart Disease. Int. J. Cardiol. 2024, 410, 132227. https://doi.org/10.1016/j.ijcard.2024.132227.
- 115.
Wang, X.; Liu, Z.; Zhang, L.; Hu, G.; Tao, L.; Zhang, F. Mitochondrial Transplantation for the Treatment of Cardiac and Noncardiac Diseases: Mechanisms, Prospective, and Challenges. Life Med. 2024, 3, lnae017. https://doi.org/10.1093/lifemedi/lnae017.
- 116.
Liu, Q.; Zhang, X.; Zhu, T.; Xu, Z.; Dong, Y.; Chen, B. Mitochondrial Transfer from Mesenchymal Stem Cells: Mechanisms and Functions. Mitochondrion 2024, 79, 101950. https://doi.org/10.1016/j.mito.2024.101950.
- 117.
Kim, Y.S.; Yoo, S.; Jung, Y.J.; Yoon, J.W.; Kwon, Y.S.; Lee, N.; Cheon, C.K.; Kim, J.H. Allogenic Mitochondria Transfer Improves Cardiac Function in iPS-Cell-Differentiated Cardiomyocytes of a Patient with Barth Syndrome. Exp. Mol. Med. 2025, 57, 1260–1271. https://doi.org/10.1038/s12276-025-01472-7.
- 118.
Hassanpour, P.; Sadeghsoltani, F.; Saghebasl, S.; Boroumand, S.; Khanicheragh, P.; Tafti, S.H.A.; Rahbarghazi, R.; Rahmati, M. Mitochondrial Transplantation for Cardioprotection and Induction of Angiogenesis in Ischemic Heart Disease. Stem Cell Res. Ther. 2025, 16, 54. https://doi.org/10.1186/s13287-025-04193-w.
- 119.
Zhang, X.; Yang, Y.; Wang, H.; Yan, C.; Feng, Y.; Ma, X.; Hu, M.; Li, S.; Cheng, C. Exploring Mitochondrial Health and Transplantation Strategies in DCD Heart Transplantation: A Systematic Review. J. Transl. Med. 2025, 23, 789. https://doi.org/10.1186/s12967-025-06805-8.
- 120.
Wang, Z.; Zhu, J.; Xu, M.; Ma, X.; Shen, M.; Yan, J.; Gan, G.; Zhou, X. Transplantation of Exogenous Mitochondria Mitigates Myocardial Dysfunction after Cardiac Arrest. eLife 2025, 13, RP98554. https://doi.org/10.7554/eLife.98554.3.
- 121.
Masuzawa, A.; Black, K.M.; Pacak, C.A.; Ericsson, M.; Barnett, R.J.; Drumm, C.; Seth, P.; Bloch, D.B.; Levitsky, S.; Cowan, D.B.; et al. Transplantation of Autologously Derived Mitochondria Protects the Heart from Ischemia-Reperfusion Injury. Am. J. Physiol.-Heart Circ. Physiol. 2013, 304, H966–H982. https://doi.org/10.1152/ajpheart.00883.2012.
- 122.
Zhang, A.; Liu, Y.; Pan, J.; Pontanari, F.; Chia-Hao Chang, A.; Wang, H.; Gao, S.; Wang, C.; Chang, A. Cy. Delivery of Mitochondria Confers Cardioprotection through Mitochondria Replenishment and Metabolic Compliance. Mol. Ther. 2023, 31, 1468–1479. https://doi.org/10.1016/j.ymthe.2023.02.016.
- 123.
Chouchani, E.T.; Pell, V.R.; Gaude, E.; Aksentijević, D.; Sundier, S.Y.; Robb, E.L.; Logan, A.; Nadtochiy, S.M.; Ord, E.N.J.; Smith, A.C.; et al. Ischaemic Accumulation of Succinate Controls Reperfusion Injury through Mitochondrial ROS. Nature 2014, 515, 431–435. https://doi.org/10.1038/nature13909.
- 124.
Chavda, V.; Lu, B. Reverse Electron Transport at Mitochondrial Complex I in Ischemic Stroke, Aging, and Age-Related Diseases. Antioxidants 2023, 12, 895. https://doi.org/10.3390/antiox12040895.
- 125.
Tabata Fukushima, C.; Dancil, I.-S.; Clary, H.; Shah, N.; Nadtochiy, S.M.; Brookes, P.S. Reactive Oxygen Species Generation by Reverse Electron Transfer at Mitochondrial Complex I under Simulated Early Reperfusion Conditions. Redox Biol. 2024, 70, 103047. https://doi.org/10.1016/j.redox.2024.103047.
- 126.
Alsadder, L.; Hamadah, A. Cardiac Ischaemia–Reperfusion Injury: Pathophysiology, Therapeutic Targets and Future Interventions. Biomedicines 2025, 13, 2084. https://doi.org/10.3390/biomedicines13092084.
- 127.
Morciano, G.; Pinton, P. Modulation of Mitochondrial Permeability Transition Pores in Reperfusion Injury: Mechanisms and Therapeutic Approaches. Eur. J. Clin. Investig. 2025, 55, e14331. https://doi.org/10.1111/eci.14331.
- 128.
Sorby-Adams, A.; Prime, T.A.; Miljkovic, J.L.; Prag, H.A.; Krieg, T.; Murphy, M.P. A Model of Mitochondrial Superoxide Production during Ischaemia-Reperfusion Injury for Therapeutic Development and Mechanistic Understanding. Redox Biol. 2024, 72, 103161. https://doi.org/10.1016/j.redox.2024.103161.
- 129.
Gibbs, E.T.; Lerner, C.A.; Watson, M.A.; Wong, H.-S.; Gerencser, A.A.; Brand, M.D. Site IQ in Mitochondrial Complex I Generates S1QEL-Sensitive Superoxide/Hydrogen Peroxide in Both the Reverse and Forward Reactions. Biochem. J. 2023, 480, 363–384. https://doi.org/10.1042/BCJ20220611.
- 130.
Milliken, A.S.; Nadtochiy, S.M.; Brookes, P.S. Inhibiting Succinate Release Worsens Cardiac Reperfusion Injury by Enhancing Mitochondrial Reactive Oxygen Species Generation. J. Am. Heart Assoc. 2022, 11, e026135. https://doi.org/10.1161/JAHA.122.026135.
- 131.
Borzuta, H.; Kociemba, W.; Bochenek, O.; Jarowicz, M.; Wsół, A. Advances in Pathophysiology and Novel Therapeutic Strategies for Coronary No-Reflow Phenomenon. Biomedicines 2025, 13, 1716. https://doi.org/10.3390/biomedicines13071716.
- 132.
Luo, Q.; Cheng, N.; Yang, Y.; Shao, N.; Nie, T.; Chen, J.; Huang, C.; Zhang, S.; Huang, Y.; Ieong, C.M.; et al. Multi-Stage Cooperative ROS-Responsive Hydrogel Platform for Drug Delivery in Myocardial Ischemia-Reperfusion Injury Repair. Mater. Today Bio 2025, 32, 101854. https://doi.org/10.1016/j.mtbio.2025.101854.
- 133.
Pang, M.; Wang, S.; Shi, T.; Chen, J. Overview of MitoQ on Prevention and Management of Cardiometabolic Diseases: A Scoping Review. Front. Cardiovasc. Med. 2025, 12, 1506460. https://doi.org/10.3389/fcvm.2025.1506460.
- 134.
Thorp, E.B.; Karlstaedt, A. Intersection of Immunology and Metabolism in Myocardial Disease. Circ. Res. 2024, 134, 1824–1840. https://doi.org/10.1161/CIRCRESAHA.124.323660.
- 135.
Liao, Y.; Zhu, L. At the Heart of Inflammation: Unravelling Cardiac Resident Macrophage Biology. J. Cell. Mol. Med. 2024, 28, e70050. https://doi.org/10.1111/jcmm.70050.
- 136.
Guariento, A.; Piekarski, B.L.; Doulamis, I.P.; Blitzer, D.; Ferraro, A.M.; Harrild, D.M.; Zurakowski, D.; Del Nido, P.J.; McCully, J.D.; Emani, S.M. Autologous Mitochondrial Transplantation for Cardiogenic Shock in Pediatric Patients Following Ischemia-Reperfusion Injury. J. Thorac. Cardiovasc. Surg. 2021, 162, 992–1001. https://doi.org/10.1016/j.jtcvs.2020.10.151.
- 137.
Ng, S.M.; Neubauer, S.; Rider, O.J. Myocardial Metabolism in Heart Failure. Curr. Heart Fail. Rep. 2023, 20, 63–75. https://doi.org/10.1007/s11897-023-00589-y.
- 138.
Atar, D.; Arheden, H.; Berdeaux, A.; Bonnet, J.-L.; Carlsson, M.; Clemmensen, P.; Cuvier, V.; Danchin, N.; Dubois-Rande, J.-L.; Engblom, H.; et al. Effect of Intravenous TRO40303 as an Adjunct to Primary Percutaneous Coronary Intervention for Acute ST-Elevation Myocardial Infarction: MITOCARE Study Results. Eur. Heart J. 2015, 36, 112–119. https://doi.org/10.1093/eurheartj/ehu331.
- 139.
Scandalis, L.; Kitzman, D.W.; Nicklas, B.J.; Lyles, M.; Brubaker, P.; Nelson, M.B.; Gordon, M.; Stone, J.; Bergstrom, J.; Neufer, P.D.; et al. Skeletal Muscle Mitochondrial Respiration and Exercise Intolerance in Patients with Heart Failure with Preserved Ejection Fraction. JAMA Cardiol. 2023, 8, 575. https://doi.org/10.1001/jamacardio.2023.0957.
- 140.
Shah, I.A.; Ishaq, S.; Lee, S.-D.; Wu, B.-T. Effects of Exercise Training on Cardiac Mitochondrial Functions in Diabetic Heart: A Systematic Review. Int. J. Mol. Sci. 2024, 26, 8. https://doi.org/10.3390/ijms26010008.
- 141.
Mounsey, L.A.; Guo, M.; Lau, E.S.; Ho, J.E. Exercise Training in Heart Failure: Clinical Benefits and Mechanisms. Circ. Res. 2025, 137, 273–289. https://doi.org/10.1161/CIRCRESAHA.124.325533.
- 142.
Gibson, C.M.; Giugliano, R.P.; Kloner, R.A.; Bode, C.; Tendera, M.; Jánosi, A.; Merkely, B.; Godlewski, J.; Halaby, R.; Korjian, S.; et al. EMBRACE STEMI Study: A Phase 2a Trial to Evaluate the Safety, Tolerability, and Efficacy of Intravenous MTP-131 on Reperfusion Injury in Patients Undergoing Primary Percutaneous Coronary Intervention. Eur. Heart J. 2016, 37, 1296–1303. https://doi.org/10.1093/eurheartj/ehv597.
- 143.
Butler, J.; Khan, M.S.; Anker, S.D.; Fonarow, G.C.; Kim, R.J.; Nodari, S.; O’Connor, C.M.; Pieske, B.; Pieske-Kraigher, E.; Sabbah, H.N.; et al. Effects of Elamipretide on Left Ventricular Function in Patients with Heart Failure with Reduced Ejection Fraction: The PROGRESS-HF Phase 2 Trial. J. Card. Fail. 2020, 26, 429–437. https://doi.org/10.1016/j.cardfail.2020.02.001.
- 144.
Packer, M.; Anker, S.D.; Butler, J.; Filippatos, G.; Pocock, S.J.; Carson, P.; Januzzi, J.; Verma, S.; Tsutsui, H.; Brueckmann, M.; et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N. Engl. J. Med. 2020, 383, 1413–1424. https://doi.org/10.1056/NEJMoa2022190.
- 145.
Yao, H.; He, Q.; Wei, S.; Xiang, L.; Luo, Y.; Huang, C.; Liu, W.; Zheng, C.; Li, X.; Gao, Y. Targeted Inhibition of Macrophage STING Signaling Alleviates Inflammatory Injury and Ventricular Remodeling in Acute Myocardial Infarction. Acta Pharm. Sin. B 2025, 15, 4030–4046. https://doi.org/10.1016/j.apsb.2025.06.014.
- 146.
Pan, X.; Hao, E.; Zhang, F.; Wei, W.; Du, Z.; Yan, G.; Wang, X.; Deng, J.; Hou, X. Diabetes Cardiomyopathy: Targeted Regulation of Mitochondrial Dysfunction and Therapeutic Potential of Plant Secondary Metabolites. Front. Pharmacol. 2024, 15, 1401961. https://doi.org/10.3389/fphar.2024.1401961.
- 147.
Chen, Y.; Liu, X.; Liu, Y.; Li, Y.; Li, D.; Mei, Z.; Deng, Y. Mitochondrial Quality Control in Diabetes Mellitus and Complications: Molecular Mechanisms and Therapeutic Strategies. Cell Death Dis. 2025, 16, 652. https://doi.org/10.1038/s41419-025-07936-y.
- 148.
Xiong, Z.; Liao, Y.; Zhang, Z.; Wan, Z.; Liang, S.; Guo, J. Molecular Insights into Oxidative-Stress-Mediated Cardiomyopathy and Potential Therapeutic Strategies. Biomolecules 2025, 15, 670. https://doi.org/10.3390/biom15050670.
- 149.
Shang, D.; Zhang, X.; Liu, H.; Tu, Z. Suppressing Endothelial Senescence: A Comprehensive Analysis of Metformin’s Mechanisms and Implications. Life Sci. 2025, 376, 123730. https://doi.org/10.1016/j.lfs.2025.123730.
- 150.
Mauriello, A.; Correra, A.; Molinari, R.; Del Vecchio, G.E.; Tessitore, V.; D’Andrea, A.; Russo, V. Mitochondrial Dysfunction in Atrial Fibrillation: The Need for a Strong Pharmacological Approach. Biomedicines 2024, 12, 2720. https://doi.org/10.3390/biomedicines12122720.
- 151.
Pfenniger, A.; Yoo, S.; Arora, R. Oxidative Stress and Atrial Fibrillation. J. Mol. Cell. Cardiol. 2024, 196, 141–151. https://doi.org/10.1016/j.yjmcc.2024.09.011.
- 152.
Chang, Y.; Zou, Q. Mitochondrial Calcium Homeostasis and Atrial Fibrillation: Mechanisms and Therapeutic Strategies Review. Curr. Probl. Cardiol. 2025, 50, 102988. https://doi.org/10.1016/j.cpcardiol.2025.102988.
- 153.
Rocca, C.; Soda, T.; De Francesco, E.M.; Fiorillo, M.; Moccia, F.; Viglietto, G.; Angelone, T.; Amodio, N. Mitochondrial Dysfunction at the Crossroad of Cardiovascular Diseases and Cancer. J. Transl. Med. 2023, 21, 635. https://doi.org/10.1186/s12967-023-04498-5.
- 154.
Liu, G.Z.; Xu, W.; Zang, Y.X.; Lou, Q.; Hang, P.Z.; Gao, Q.; Shi, H.; Liu, Q.Y.; Wang, H.; Sun, X.; et al. Honokiol Inhibits Atrial Metabolic Remodeling in Atrial Fibrillation Through Sirt3 Pathway. Front. Pharmacol. 2022, 13, 813272. https://doi.org/10.3389/fphar.2022.813272.
- 155.
Oropeza-Almazán, Y.; Blatter, L.A. Role of Mitochondrial ROS for Calcium Alternans in Atrial Myocytes. Biomolecules 2024, 14, 144. https://doi.org/10.3390/biom14020144.
- 156.
Qi, X.; Xiong, F.; Xiao, J.; Muthukumarasamy, K.M.; Altuntas, Y.; Zhong, Y.; Abu-Taha, I.; Bruns, F.; Tekook, M.; Kamler, M.; et al. Time-Dependent Mitochondrial Remodeling in Experimental Atrial Fibrillation and Potential Therapeutic Relevance. Pathology February 1, 2025. https://doi.org/10.1101/2025.01.29.635508.
- 157.
Zhang, Q.; Siyuan, Z.; Xing, C.; Ruxiu, L. SIRT3 Regulates Mitochondrial Function: A Promising Star Target for Cardiovascular Disease Therapy. Biomed. Pharmacother. 2024, 170, 116004. https://doi.org/10.1016/j.biopha.2023.116004.
- 158.
Peng, H.; Yuan, J.; Wang, Z.; Mo, B.; Wang, Y.; Wang, Y.; Wang, Q. NR4A3 Prevents Diabetes Induced Atrial Cardiomyopathy by Maintaining Mitochondrial Energy Metabolism and Reducing Oxidative Stress. eBioMedicine 2024, 106, 105268. https://doi.org/10.1016/j.ebiom.2024.105268.
- 159.
Cao, Y.; Cui, L.; Tuo, S.; Liu, H.; Cui, S. Resveratrol Mediates Mitochondrial Function through the Sirtuin 3 Pathway to Improve Abnormal Metabolic Remodeling in Atrial Fibrillation. Eur. J. Histochem. 2024, 68, 4004. https://doi.org/10.4081/ejh.2024.4004.
- 160.
Qin, A.; Wen, Z.; Xiong, S. Myocardial Mitochondrial DNA Drives Macrophage Inflammatory Response through STING Signaling in Coxsackievirus B3-Induced Viral Myocarditis. Cells 2023, 12, 2555. https://doi.org/10.3390/cells12212555.
- 161.
Qi, Y.; Yin, J.; Xia, W.; Yang, S. Exploring the Role of Mitochondrial Antiviral Signaling Protein in Cardiac Diseases. Front. Immunol. 2025, 16, 1540774. https://doi.org/10.3389/fimmu.2025.1540774.
- 162.
An, C.; Li, Z.; Chen, Y.; Huang, S.; Yang, F.; Hu, Y.; Xu, T.; Zhang, C.; Ge, S. The cGAS-STING Pathway in Cardiovascular Diseases: From Basic Research to Clinical Perspectives. Cell Biosci. 2024, 14, 58. https://doi.org/10.1186/s13578-024-01242-4.
- 163.
Mou, J.; Chen, Y.; Zhu, X.; Xu, B.; Wang, M.; Xie, J.; Lin, T.; Gu, Q.; Wu, Q.; Che, Z.; et al. Emerging Role of the cGAS-STING Pathway in Cardiovascular Diseases: Biologic Function, Mechanisms and Targeted Therapy. Mol. Med. 2025, 31, 218. https://doi.org/10.1186/s10020-025-01273-8.

This work is licensed under a Creative Commons Attribution 4.0 International License.


