The rapid development of artificial intelligence (AI) is a powerful catalyst for personalized exercise monitoring and management. This narrative review examines how exercise and physiological indicators are used in health surveillance and disease management and evaluates the integration of AI in these domains. Accumulating evidence demonstrates strong associations between indicators such as gait, electromyography, and cardiorespiratory fitness with disease outcomes and overall health. AI methods are increasingly being applied to improve monitoring, diagnosis, and personalized exercise prescription, offering greater precision compared with traditional approaches. Nevertheless, challenges persist, including limited data quality, lack of multimodal integration, concerns with interpretability, and ethical issues surrounding privacy. Many studies still rely on small cohorts, limiting clinical applicability. We synthesize current findings, identify research gaps, and propose future directions to enhance the robustness and practical value of AI-driven approaches in exercise and health research.
- Open Access
- Review
Artificial Intelligence Empowers Monitoring, Assessment, and Management in Exercise
- Min Zhu 1,2,
- Yuhuai Guo 3,4,
- Mingliang Zhao 1,
- Tao Sun 1,
- Rengfei Shi 2,*,
- Xiaojiao Zheng 1,*,
- Tianlu Chen 1,*
Author Information
Received: 04 Nov 2025 | Revised: 12 Jan 2026 | Accepted: 11 Feb 2026 | Published: 27 Apr 2026
Abstract
Graphical Abstract
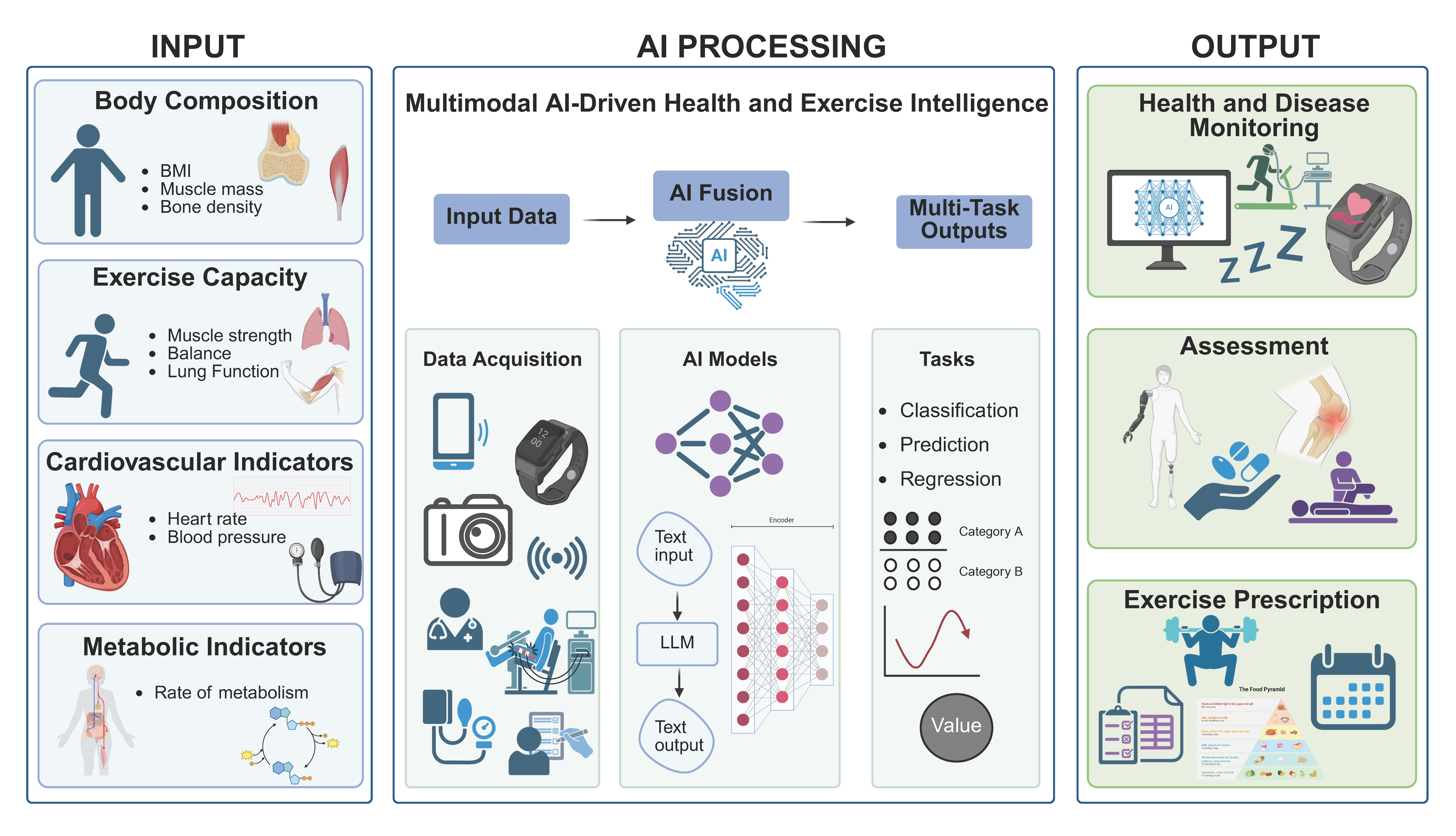
Keywords
artificial intelligence | deep learning | exercise | healthcare | sports
References
- 1.
Dimeo, F.; Pagonas, N.; Seibert, F.; Arndt, R.; Zidek, W.; Westhoff, T.H. Aerobic exercise reduces blood pressure in resistant hypertension. Hypertension 2012, 60, 653–658. https://doi.org/10.1161/HYPERTENSIONAHA.112.197780.
- 2.
Lopes, S.; Mesquita-Bastos, J.; Garcia, C.; Bertoquini, S.; Ribau, V.; Teixeira, M.; Ribeiro, I.P.; Melo, J.B.; Oliveira, J.; Figueiredo, D.; et al. Effect of Exercise Training on Ambulatory Blood Pressure Among Patients With Resistant Hypertension: A Randomized Clinical Trial. JAMA Cardiol. 2021, 6, 1317–1323. https://doi.org/10.1001/jamacardio.2021.2735.
- 3.
Jansen, A.E.; Koop, M.M.; Rosenfeldt, A.B.; Alberts, J.L. High intensity aerobic exercise improves bimanual coordination of grasping forces in Parkinson’s disease. Park. Relat. Disord. 2021, 87, 13–19. https://doi.org/10.1016/j.parkreldis.2021.04.005.
- 4.
Mak, M.K.Y.; Wong-Yu, I.S.K. Six-Month Community-Based Brisk Walking and Balance Exercise Alleviates Motor Symptoms and Promotes Functions in People with Parkinson’s Disease: A Randomized Controlled Trial. J. Park. Dis. 2021, 11, 1431–1441. https://doi.org/10.3233/JPD-202503.
- 5.
Jaakkola, J.J.K.; Aalto, S.A.M.; Hernberg, S.; Kiihamaki, S.P.; Jaakkola, M.S. Regular exercise improves asthma control in adults: A randomized controlled trial. Sci. Rep. 2019, 9, 12088. https://doi.org/10.1038/s41598-019-48484-8.
- 6.
Sveaas, S.H.; Bilberg, A.; Berg, I.J.; Provan, S.A.; Rollefstad, S.; Semb, A.G.; Hagen, K.B.; Johansen, M.W.; Pedersen, E.; Dagfinrud, H. High intensity exercise for 3 months reduces disease activity in axial spondyloarthritis (axSpA): A multicentre randomised trial of 100 patients. Br. J. Sports Med. 2020, 54, 292–297. https://doi.org/10.1136/bjsports-2018-099943.
- 7.
Song, D.; Yu, D.S.F. Effects of a moderate-intensity aerobic exercise programme on the cognitive function and quality of life of community-dwelling elderly people with mild cognitive impairment: A randomised controlled trial. Int. J. Nurs. Stud. 2019, 93, 97–105. https://doi.org/10.1016/j.ijnurstu.2019.02.019.
- 8.
Kwok, J.Y.Y.; Kwan, J.C.Y.; Auyeung, M.; Mok, V.C.T.; Lau, C.K.Y.; Choi, K.C.; Chan, H.Y.L. Effects of Mindfulness Yoga vs Stretching and Resistance Training Exercises on Anxiety and Depression for People With Parkinson Disease: A Randomized Clinical Trial. JAMA Neurol. 2019, 76, 755–763. https://doi.org/10.1001/jamaneurol.2019.0534.
- 9.
Uchiyama, K.; Adachi, K.; Muraoka, K.; Nakayama, T.; Oshida, T.; Yasuda, M.; Hishikawa, A.; Minakuchi, H.; Miyashita, K.; Tokuyama, H.; et al. Home-based aerobic exercise and resistance training for severe chronic kidney disease: A randomized controlled trial. J. Cachexia Sarcopenia Muscle 2021, 12, 1789–1802. https://doi.org/10.1002/jcsm.12775.
- 10.
Hong, A.R.; Kim, S.W. Effects of Resistance Exercise on Bone Health. Endocrinol. Metab. 2018, 33, 435–444. https://doi.org/10.3803/EnM.2018.33.4.435.
- 11.
Portero, P.; Bigard, A.X.; Gamet, D.; Flageat, J.R.; Guezennec, C.Y. Effects of resistance training in humans on neck muscle performance, and electromyogram power spectrum changes. Eur. J. Appl. Physiol. 2001, 84, 540–546. https://doi.org/10.1007/s004210100399.
- 12.
Stanghelle, B.; Bentzen, H.; Giangregorio, L.; Pripp, A.H.; Skelton, D.A.; Bergland, A. Effects of a resistance and balance exercise programme on physical fitness, health-related quality of life and fear of falling in older women with osteoporosis and vertebral fracture: A randomized controlled trial. Osteoporos. Int. 2020, 31, 1069–1078. https://doi.org/10.1007/s00198-019-05256-4.
- 13.
Aitkens, S.G.; McCrory, M.A.; Kilmer, D.D.; Bernauer, E.M. Moderate resistance exercise program: Its effect in slowly progressive neuromuscular disease. Arch. Phys. Med. Rehabil. 1993, 74, 711–715.
- 14.
Lazzer, S.; Tringali, G.; Caccavale, M.; De Micheli, R.; Abbruzzese, L.; Sartorio, A. Effects of high-intensity interval training on physical capacities and substrate oxidation rate in obese adolescents. J. Endocrinol. Invest. 2017, 40, 217–226. https://doi.org/10.1007/s40618-016-0551-4.
- 15.
Wewege, M.; van den Berg, R.; Ward, R.E.; Keech, A. The effects of high-intensity interval training vs. moderate-intensity continuous training on body composition in overweight and obese adults: A systematic review and meta-analysis. Obes. Rev. 2017, 18, 635–646. https://doi.org/10.1111/obr.12532.
- 16.
Schaun, G.Z.; Alberton, C.L.; Ribeiro, D.O.; Pinto, S.S. Acute effects of high-intensity interval training and moderate-intensity continuous training sessions on cardiorespiratory parameters in healthy young men. Eur. J. Appl. Physiol. 2017, 117, 1437–1444. https://doi.org/10.1007/s00421-017-3636-7.
- 17.
Wen, D.; Utesch, T.; Wu, J.; Robertson, S.; Liu, J.; Hu, G.; Chen, H. Effects of different protocols of high intensity interval training for VO(2)max improvements in adults: A meta-analysis of randomised controlled trials. J. Sci. Med. Sport. 2019, 22, 941–947. https://doi.org/10.1016/j.jsams.2019.01.013.
- 18.
Astorino, T.A.; Allen, R.P.; Roberson, D.W.; Jurancich, M. Effect of high-intensity interval training on cardiovascular function, VO2max, and muscular force. J. Strength. Cond. Res. 2012, 26, 138–145.
- 19.
Higashi, Y.; Yoshizumi, M. Exercise and endothelial function: Role of endothelium-derived nitric oxide and oxidative stress in healthy subjects and hypertensive patients. Pharmacol. Ther. 2004, 102, 87–96. https://doi.org/10.1016/j.pharmthera.2004.02.003.
- 20.
Bender, T.; Nagy, G.; Barna, I.; Tefner, I.; Kadas, E.; Geher, P. The effect of physical therapy on beta-endorphin levels. Eur. J. Appl. Physiol. 2007, 100, 371–382. https://doi.org/10.1007/s00421-007-0469-9.
- 21.
Frosig, C.; Richter, E.A. Improved insulin sensitivity after exercise: Focus on insulin signaling. Obesity 2009, 17, S15–S20. https://doi.org/10.1038/oby.2009.383.
- 22.
Luan, X.; Tian, X.; Zhang, H.; Huang, R.; Li, N.; Chen, P.; Wang, R. Exercise as a prescription for patients with various diseases. J. Sport. Health Sci. 2019, 8, 422–441. https://doi.org/10.1016/j.jshs.2019.04.002.
- 23.
Mendes, R.; Sousa, N.; Almeida, A.; Subtil, P.; Guedes-Marques, F.; Reis, V.M.; Themudo-Barata, J.L. Exercise prescription for patients with type 2 diabetes-a synthesis of international recommendations: Narrative review. Br. J. Sports Med. 2016, 50, 1379–1381. https://doi.org/10.1136/bjsports-2015-094895.
- 24.
Farrahi, V.; Muhammad, U.; Rostami, M.; Oussalah, M. AccNet24: A deep learning framework for classifying 24-hour activity behaviours from wrist-worn accelerometer data under free-living environments. Int. J. Med. Inform. 2023, 172, 105004. https://doi.org/10.1016/j.ijmedinf.2023.105004.
- 25.
Qiu, Y.; Wang, J.; Jin, Z.; Chen, H.; Zhang, M.; Guo, L. Pose-guided matching based on deep learning for assessing quality of action on rehabilitation training. Biomed. Signal Process. Control 2022, 72, 103323. https://doi.org/10.1016/j.bspc.2021.103323.
- 26.
Kaur, R.; Levy, J.; Motl, R.W.; Sowers, R.; Hernandez, M.E. Deep Learning for Multiple Sclerosis Differentiation Using Multi-Stride Dynamics in Gait. IEEE Trans. Biomed. Eng. 2023, 70, 2181–2192. https://doi.org/10.1109/TBME.2023.3238680.
- 27.
Meyer, B.M.; Tulipani, L.J.; Gurchiek, R.D.; Allen, D.A.; Adamowicz, L.; Larie, D.; Solomon, A.J.; Cheney, N.; McGinnis, R.S. Wearables and Deep Learning Classify Fall Risk From Gait in Multiple Sclerosis. IEEE J. Biomed. Health Inform. 2021, 25, 1824–1831. https://doi.org/10.1109/JBHI.2020.3025049.
- 28.
Xia, Y.; Yao, Z.; Ye, Q.; Cheng, N. A Dual-Modal Attention-Enhanced Deep Learning Network for Quantification of Parkinson’s Disease Characteristics. IEEE Trans. Neural Syst. Rehabil. Eng. 2020, 28, 42–51. https://doi.org/10.1109/TNSRE.2019.2946194.
- 29.
McArdle, W.D.; Katch, F.I.; Katch, V.L. Essentials of Exercise Physiology; Lippincott Williams & Wilkins: Ambler, PA, USA, 2006.
- 30.
McGinnis, P.M. Biomechanics of Sport and Exercise; Human Kinetics: Champaign, IL, USA, 2013.
- 31.
Woolf, S.H.; Jonas, S.; Kaplan-Liss, E. Health Promotion and Disease Prevention in Clinical Practice; Lippincott Williams & Wilkins: Ambler, PA, USA, 2008.
- 32.
Tirosh, A.; Shai, I.; Afek, A.; Dubnov-Raz, G.; Ayalon, N.; Gordon, B.; Derazne, E.; Tzur, D.; Shamis, A.; Vinker, S. Adolescent BMI trajectory and risk of diabetes versus coronary disease. N. Engl. J. Med. 2011, 364, 1315–1325.
- 33.
Brown, C.D.; Higgins, M.; Donato, K.A.; Rohde, F.C.; Garrison, R.; Obarzanek, E.; Ernst, N.D.; Horan, M. Body mass index and the prevalence of hypertension and dyslipidemia. Obes. Res. 2000, 8, 605–619. https://doi.org/10.1038/oby.2000.79.
- 34.
Volpato, S.; Bianchi, L.; Lauretani, F.; Lauretani, F.; Bandinelli, S.; Guralnik, J.M.; Zuliani, G.; Ferrucci, L. Role of muscle mass and muscle quality in the association between diabetes and gait speed. Diabetes Care 2012, 35, 1672–1679. https://doi.org/10.2337/dc11-2202.
- 35.
John, S.G.; Sigrist, M.K.; Taal, M.W.; McIntyre, C.W. Natural history of skeletal muscle mass changes in chronic kidney disease stage 4 and 5 patients: An observational study. PLoS ONE 2013, 8, e65372. https://doi.org/10.1371/journal.pone.0065372.
- 36.
Gan, D.; Wang, L.; Jia, M.; Ru, Y.; Ma, Y.; Zheng, W.; Zhao, X.; Yang, F.; Wang, T.; Mu, Y.; et al. Low muscle mass and low muscle strength associate with nonalcoholic fatty liver disease. Clin. Nutr. 2020, 39, 1124–1130. https://doi.org/10.1016/j.clnu.2019.04.023.
- 37.
Kim, G.; Lee, S.E.; Lee, Y.B.; Jun, J.E.; Ahn, J.; Bae, J.C.; Jin, S.M.; Hur, K.Y.; Jee, J.H.; Lee, M.K.; et al. Relationship Between Relative Skeletal Muscle Mass and Nonalcoholic Fatty Liver Disease: A 7-Year Longitudinal Study. Hepatology 2018, 68, 1755–1768. https://doi.org/10.1002/hep.30049.
- 38.
Tyrovolas, S.; Panagiotakos, D.; Georgousopoulou, E.; Chrysohoou, C.; Tousoulis, D.; Haro, J.M.; Pitsavos, C. Skeletal muscle mass in relation to 10 year cardiovascular disease incidence among middle aged and older adults: The ATTICA study. J. Epidemiol. Community Health 2020, 74, 26–31. https://doi.org/10.1136/jech-2019-212268.
- 39.
Bryant, R.V.; Ooi, S.; Schultz, C.G.; Goess, C.; Grafton, R.; Hughes, J.; Lim, A.; Bartholomeusz, F.D.; Andrews, J.M. Low muscle mass and sarcopenia: Common and predictive of osteopenia in inflammatory bowel disease. Aliment. Pharmacol. Ther. 2015, 41, 895–906. https://doi.org/10.1111/apt.13156.
- 40.
Lodder, M.C.; de Jong, Z.; Kostense, P.J.; Molenaar, E.T.; Staal, K.; Voskuyl, A.E.; Hazes, J.M.; Dijkmans, B.A.; Lems, W.F. Bone mineral density in patients with rheumatoid arthritis: Relation between disease severity and low bone mineral density. Ann. Rheum. Dis. 2004, 63, 1576–1580. https://doi.org/10.1136/ard.2003.016253.
- 41.
Klingberg, E.; Lorentzon, M.; Göthlin, J.; Mellström, D.; Geijer, M.; Ohlsson, C.; Atkinson, E.J.; Khosla, S.; Carlsten, H.; Forsblad-d’Elia, H. Bone microarchitecture in ankylosing spondylitis and the association with bone mineral density, fractures, and syndesmophytes. Arthritis Res. Ther. 2013, 15, 1–11.
- 42.
Kjensli, A.; Mowinckel, P.; Ryg, M.S.; Falch, J.A. Low bone mineral density is related to severity of chronic obstructive pulmonary disease. Bone 2007, 40, 493–497. https://doi.org/10.1016/j.bone.2006.09.005.
- 43.
Groothoff, J.W.; Offringa, M.; Van Eck-Smit, B.L.; Gruppen, M.P.; Van De Kar, N.J.; Wolff, E.D.; Lilien, M.R.; Davin, J.C.; Heymans, H.S.; Dekker, F.W. Severe bone disease and low bone mineral density after juvenile renal failure. Kidney Int. 2003, 63, 266–275. https://doi.org/10.1046/j.1523-1755.2003.00727.x.
- 44.
Gonzalez, D.; Mazure, R.; Mautalen, C.; Vazquez, H.; Bai, J. Body composition and bone mineral density in untreated and treated patients with celiac disease. Bone 1995, 16, 231–234.
- 45.
Kamanli, A.; Ardicoglu, O.; Ozgocmen, S.; Yoldas, T.K. Bone mineral density in patients with Parkinson’s disease. Aging Clin. Exp. Res. 2008, 20, 277–279.
- 46.
Inkster, L.M.; Eng, J.J.; MacIntyre, D.L.; Stoessl, A.J. Leg muscle strength is reduced in Parkinson’s disease and relates to the ability to rise from a chair. Mov. Disord. 2003, 18, 157–162. https://doi.org/10.1002/mds.10299.
- 47.
Allen, N.E.; Canning, C.G.; Sherrington, C.; Fung, V.S. Bradykinesia, muscle weakness and reduced muscle power in Parkinson’s disease. Mov. Disord. 2009, 24, 1344–1351. https://doi.org/10.1002/mds.22609.
- 48.
Liu, M.; He, P.; Ye, Z.; Zhang, Y.; Zhou, C.; Yang, S.; Zhang, Y.; Qin, X. Association of handgrip strength and walking pace with incident Parkinson’s disease. J. Cachexia Sarcopenia Muscle 2024, 15, 198–207. https://doi.org/10.1002/jcsm.13366.
- 49.
Kuo, K.; Zhang, Y.R.; Chen, S.D.; He, X.Y.; Huang, S.Y.; Wu, B.S.; Deng, Y.T.; Yang, L.; Ou, Y.N.; Guo, Y.; et al. Associations of grip strength, walking pace, and the risk of incident dementia: A prospective cohort study of 340212 participants. Alzheimers Dement 2023, 19, 1415–1427. https://doi.org/10.1002/alz.12793.
- 50.
Yahia, A.; Ghroubi, S.; Mhiri, C.; Elleuch, M.H. Relationship between muscular strength, gait and postural parameters in multiple sclerosis. Ann. Phys. Rehabil. Med. 2011, 54, 144–155. https://doi.org/10.1016/j.rehab.2011.02.004.
- 51.
Malmstroem, S.; Grove-Laugesen, D.; Riis, A.L.; Bruun, B.J.; Ebbehoj, E.; Hansen, K.W.; Watt, T.; Rejnmark, L. Muscle Performance and Postural Stability Are Reduced in Patients with Newly Diagnosed Graves’ Disease. Thyroid. 2019, 29, 783–789. https://doi.org/10.1089/thy.2018.0318.
- 52.
Dziubek, W.; Bulinska, K.; Stefanska, M.; Wozniewski, M.; Kropielnicka, K.; Jasinski, T.; Jasinski, R.; Pilch, U.; Dabrowska, G.; Skorkowska-Telichowska, K.; et al. Peripheral arterial disease decreases muscle torque and functional walking capacity in elderly. Maturitas 2015, 81, 480–486. https://doi.org/10.1016/j.maturitas.2015.06.001.
- 53.
Farrow, M.; Biglands, J.; Tanner, S.; Hensor, E.M.A.; Buch, M.H.; Emery, P.; Tan, A.L. Muscle deterioration due to rheumatoid arthritis: Assessment by quantitative MRI and strength testing. Rheumatology 2021, 60, 1216–1225. https://doi.org/10.1093/rheumatology/keaa364.
- 54.
Ekdahl, C.; Broman, G. Muscle strength, endurance, and aerobic capacity in rheumatoid arthritis: A comparative study with healthy subjects. Ann. Rheum. Dis. 1992, 51, 35–40.
- 55.
Culvenor, A.G.; Felson, D.T.; Niu, J.; Wirth, W.; Sattler, M.; Dannhauer, T.; Eckstein, F. Thigh Muscle Specific-Strength and the Risk of Incident Knee Osteoarthritis: The Influence of Sex and Greater Body Mass Index. Arthritis Care Res 2017, 69, 1266–1270. https://doi.org/10.1002/acr.23182.
- 56.
Orlando, G.; Balducci, S.; Bazzucchi, I.; Pugliese, G.; Sacchetti, M. Muscle fatigability in type 2 diabetes. Diabetes Metab. Res. Rev. 2017, 33, e2821. https://doi.org/10.1002/dmrr.2821.
- 57.
Lichter, D.G.; Benedict, R.H.B.; Hershey, L.A. Importance of Balance-Gait Disorder as a Risk Factor for Cognitive Impairment, Dementia and Related Non-Motor Symptoms in Parkinson’s Disease. J. Park. Dis. 2018, 8, 539–552. https://doi.org/10.3233/JPD-181375.
- 58.
Shum, S.B.; Pang, M.Y. Children with attention deficit hyperactivity disorder have impaired balance function: Involvement of somatosensory, visual, and vestibular systems. J. Pediatr. 2009, 155, 245–249.
- 59.
Beauchamp, M.K.; Sibley, K.M.; Lakhani, B.; Romano, J.; Mathur, S.; Goldstein, R.S.; Brooks, D. Impairments in systems underlying control of balance in COPD. Chest 2012, 141, 1496–1503. https://doi.org/10.1378/chest.11-1708.
- 60.
Bui, D.S.; Burgess, J.A.; Lowe, A.J.; Perret, J.L.; Lodge, C.J.; Bui, M.; Morrison, S.; Thompson, B.R.; Thomas, P.S.; Giles, G.G.; et al. Childhood Lung Function Predicts Adult Chronic Obstructive Pulmonary Disease and Asthma-Chronic Obstructive Pulmonary Disease Overlap Syndrome. Am. J. Respir. Crit. Care Med. 2017, 196, 39–46. https://doi.org/10.1164/rccm.201606-1272OC.
- 61.
Marott, J.L.; Ingebrigtsen, T.S.; Colak, Y.; Vestbo, J.; Lange, P. Lung Function Trajectories Leading to Chronic Obstructive Pulmonary Disease as Predictors of Exacerbations and Mortality. Am. J. Respir. Crit. Care Med. 2020, 202, 210–218. https://doi.org/10.1164/rccm.201911-2115OC.
- 62.
Iversen, K.K.; Kjaergaard, J.; Akkan, D.; Kober, L.; Torp-Pedersen, C.; Hassager, C.; Vestbo, J.; Kjoller, E.; Group, E.L.F.S. The prognostic importance of lung function in patients admitted with heart failure. Eur. J. Heart Fail. 2010, 12, 685–691. https://doi.org/10.1093/eurjhf/hfq050.
- 63.
Tolmunen, T.; Laukkanen, J.A.; Hintikka, J.; Kurl, S.; Viinamaki, H.; Salonen, R.; Kauhanen, J.; Kaplan, G.A.; Salonen, J.T. Low maximal oxygen uptake is associated with elevated depressive symptoms in middle-aged men. Eur. J. Epidemiol. 2006, 21, 701–706. https://doi.org/10.1007/s10654-006-9038-5.
- 64.
Cooney, M.T.; Vartiainen, E.; Laatikainen, T.; Juolevi, A.; Dudina, A.; Graham, I.M. Elevated resting heart rate is an independent risk factor for cardiovascular disease in healthy men and women. Am. Heart J. 2010, 159, 612–619.e3. https://doi.org/10.1016/j.ahj.2009.12.029.
- 65.
Kotecha, D.; New, G.; Flather, M.D.; Eccleston, D.; Pepper, J.; Krum, H. Five-minute heart rate variability can predict obstructive angiographic coronary disease. Heart 2012, 98, 395–401. https://doi.org/10.1136/heartjnl-2011-300033.
- 66.
Jensen, M.T.; Marott, J.L.; Lange, P.; Vestbo, J.; Schnohr, P.; Nielsen, O.W.; Jensen, J.S.; Jensen, G.B. Resting heart rate is a predictor of mortality in COPD. Eur. Respir. J. 2013, 42, 341–349. https://doi.org/10.1183/09031936.00072212.
- 67.
Klag, M.J.; Whelton, P.K.; Randall, B.L.; Neaton, J.D.; Brancati, F.L.; Ford, C.E.; Shulman, N.B.; Stamler, J. Blood pressure and end-stage renal disease in men. N. Engl. J. Med. 1996, 334, 13–18.
- 68.
Rogers, A. Blood pressure and risk of stroke in patients with cerebrovascular disease. Brit Med. J. 1996, 313, 1470.
- 69.
Knecht, S.; Wersching, H.; Lohmann, H.; Bruchmann, M.; Duning, T.; Dziewas, R.; Berger, K.; Ringelstein, E.B. High-normal blood pressure is associated with poor cognitive performance. Hypertension 2008, 51, 663–668.
- 70.
Buscemi, S.; Verga, S.; Caimi, G.; Cerasola, G. A low resting metabolic rate is associated with metabolic syndrome. Clin. Nutr. 2007, 26, 806–809. https://doi.org/10.1016/j.clnu.2007.08.010.
- 71.
Zhou, J.; Ye, Z.; Wei, P.; Yi, F.; Ouyang, M.; Xiong, S.; Liu, Y.; Li, J.; Liu, M.; Xi, H.; et al. Effect of basal metabolic rate on osteoporosis: A Mendelian randomization study. Front. Public. Health 2023, 11, 1096519. https://doi.org/10.3389/fpubh.2023.1096519.
- 72.
Capecci, M.; Petrelli, M.; Emanuelli, B.; Millevolte, M.; Nicolai, A.; Provinciali, L.; Ceravolo, M.G. Rest energy expenditure in Parkinson’s disease: Role of disease progression and dopaminergic therapy. Park. Relat. Disord. 2013, 19, 238–241. https://doi.org/10.1016/j.parkreldis.2012.10.016.
- 73.
Steyn, F.J.; Ioannides, Z.A.; van Eijk, R.P.A.; Heggie, S.; Thorpe, K.A.; Ceslis, A.; Heshmat, S.; Henders, A.K.; Wray, N.R.; van den Berg, L.H.; et al. Hypermetabolism in ALS is associated with greater functional decline and shorter survival. J. Neurol. Neurosurg. Psychiatry 2018, 89, 1016–1023. https://doi.org/10.1136/jnnp-2017-317887.
- 74.
Bray, G.A. Commentary on classics in obesity. 1. Quetelet: Quantitative medicine. Obes. Res. 1994, 2, 68–71. https://doi.org/10.1002/j.1550-8528.1994.tb00046.x.
- 75.
Wang, L.; Zhou, B.; Zhao, Z.; Yang, L.; Zhang, M.; Jiang, Y.; Li, Y.; Zhou, M.; Wang, L.; Huang, Z. Body-mass index and obesity in urban and rural China: Findings from consecutive nationally representative surveys during 2004–18. Lancet 2021, 398, 53–63.
- 76.
Ortega, F.B.; Lavie, C.J.; Blair, S.N. Obesity and Cardiovascular Disease. Circ. Res. 2016, 118, 1752–1770. https://doi.org/10.1161/CIRCRESAHA.115.306883.
- 77.
Biolo, G.; Cederholm, T.; Muscaritoli, M. Muscle contractile and metabolic dysfunction is a common feature of sarcopenia of aging and chronic diseases: From sarcopenic obesity to cachexia. Clin. Nutr. 2014, 33, 737–748.
- 78.
Haseltine, K.N.; Chukir, T.; Smith, P.J.; Jacob, J.T.; Bilezikian, J.P.; Farooki, A. Bone mineral density: Clinical relevance and quantitative assessment. J. Nucl. Med. 2021, 62, 446–454.
- 79.
Runolfsdottir, H.L.; Sigurdsson, G.; Franzson, L.; Indridason, O.S. Gender comparison of factors associated with age-related differences in bone mineral density. Arch. Osteoporos. 2015, 10, 214. https://doi.org/10.1007/s11657-015-0214-7.
- 80.
McClung, M.R. The relationship between bone mineral density and fracture risk. Curr. Osteoporos. Rep. 2005, 3, 57–63.
- 81.
Goodpaster, B.H.; Park, S.W.; Harris, T.B.; Kritchevsky, S.B.; Nevitt, M.; Schwartz, A.V.; Simonsick, E.M.; Tylavsky, F.A.; Visser, M.; Newman, A.B. The loss of skeletal muscle strength, mass, and quality in older adults: The health, aging and body composition study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 1059–1064.
- 82.
Frontera, W.R.; Hughes, V.A.; Lutz, K.J.; Evans, W.J. A cross-sectional study of muscle strength and mass in 45-to 78-yr-old men and women. J. Appl. Physiol. 1991, 71, 644–650.
- 83.
Agusti, A.; Faner, R. Lung function trajectories in health and disease. Lancet Respir. Med. 2019, 7, 358–364. https://doi.org/10.1016/S2213-2600(18)30529-0.
- 84.
Fox, K.; Borer, J.S.; Camm, A.J.; Danchin, N.; Ferrari, R.; Lopez Sendon, J.L.; Steg, P.G.; Tardif, J.C.; Tavazzi, L.; Tendera, M.; et al. Resting heart rate in cardiovascular disease. J. Am. Coll. Cardiol. 2007, 50, 823–830. https://doi.org/10.1016/j.jacc.2007.04.079.
- 85.
Conen, D.; Ridker, P.M.; Mora, S.; Buring, J.E.; Glynn, R.J. Blood pressure and risk of developing type 2 diabetes mellitus: The Women’s Health Study. Eur. Heart J. 2007, 28, 2937–2943. https://doi.org/10.1093/eurheartj/ehm400.
- 86.
Ruggiero, C.; Metter, E.J.; Melenovsky, V.; Cherubini, A.; Najjar, S.S.; Ble, A.; Senin, U.; Longo, D.L.; Ferrucci, L. High basal metabolic rate is a risk factor for mortality: The Baltimore Longitudinal Study of Aging. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2008, 63, 698–706.
- 87.
Han, H.; Chen, S.; Wang, X.; Jin, J.; Li, X.; Li, Z. Association between muscle strength and mass and bone mineral density in the US general population: Data from NHANES 1999–2002. J. Orthop. Surg. Res. 2023, 18, 397. https://doi.org/10.1186/s13018-023-03877-4.
- 88.
Snow-Harter, C.; Bouxsein, M.; Lewis, B.; Charette, S.; Weinstein, P.; Marcus, R. Muscle strength as a predictor of bone mineral density in young women. J. Bone Miner. Res. 1990, 5, 589–595.
- 89.
Taaffe, D.R.; Cauley, J.A.; Danielson, M.; Nevitt, M.C.; Lang, T.F.; Bauer, D.C.; Harris, T.B. Race and sex effects on the association between muscle strength, soft tissue, and bone mineral density in healthy elders: The Health, Aging, and Body Composition Study. J. Bone Miner. Res. 2001, 16, 1343–1352.
- 90.
Wang, T.; Li, T. Deep Learning-Based Football Player Detection in Videos. Comput. Intell. Neurosci. 2022, 2022, 3540642. https://doi.org/10.1155/2022/3540642.
- 91.
Kong, L.; Huang, D.; Wang, Y. Long-Term Action Dependence-Based Hierarchical Deep Association for Multi-Athlete Tracking in Sports Videos. IEEE Trans. Image Process. 2020, 29, 7957–7969. https://doi.org/10.1109/tip.2020.3009034.
- 92.
Cornier, M.A.; Despres, J.P.; Davis, N.; Grossniklaus, D.A.; Klein, S.; Lamarche, B.; Lopez-Jimenez, F.; Rao, G.; St-Onge, M.P.; Towfighi, A.; et al. Assessing adiposity: A scientific statement from the American Heart Association. Circulation 2011, 124, 1996–2019. https://doi.org/10.1161/CIR.0b013e318233bc6a.
- 93.
Guh, D.P.; Zhang, W.; Bansback, N.; Amarsi, Z.; Birmingham, C.L.; Anis, A.H. The incidence of co-morbidities related to obesity and overweight: A systematic review and meta-analysis. BMC Public. Health 2009, 9, 88. https://doi.org/10.1186/1471-2458-9-88.
- 94.
Wang, Z.; Nakayama, T. Inflammation, a link between obesity and cardiovascular disease. Mediat. Inflamm. 2010, 2010, 535918. https://doi.org/10.1155/2010/535918.
- 95.
Alexopoulos, N.; Katritsis, D.; Raggi, P. Visceral adipose tissue as a source of inflammation and promoter of atherosclerosis. Atherosclerosis 2014, 233, 104–112. https://doi.org/10.1016/j.atherosclerosis.2013.12.023.
- 96.
Srikanthan, P.; Horwich, T.B.; Tseng, C.H. Relation of Muscle Mass and Fat Mass to Cardiovascular Disease Mortality. Am. J. Cardiol. 2016, 117, 1355–1360. https://doi.org/10.1016/j.amjcard.2016.01.033.
- 97.
Veronese, N.; Stubbs, B.; Crepaldi, G.; Solmi, M.; Cooper, C.; Harvey, N.C.; Reginster, J.Y.; Rizzoli, R.; Civitelli, R.; Schofield, P.; et al. Relationship Between Low Bone Mineral Density and Fractures With Incident Cardiovascular Disease: A Systematic Review and Meta-Analysis. J. Bone Miner. Res. 2017, 32, 1126–1135. https://doi.org/10.1002/jbmr.3089.
- 98.
Wiklund, P.; Nordstrom, A.; Jansson, J.H.; Weinehall, L.; Nordstrom, P. Low bone mineral density is associated with increased risk for myocardial infarction in men and women. Osteoporos. Int. 2012, 23, 963–970. https://doi.org/10.1007/s00198-011-1631-0.
- 99.
Frankenfield, D.C.; Rowe, W.A.; Cooney, R.N.; Smith, J.S.; Becker, D. Limits of body mass index to detect obesity and predict body composition. Nutrition 2001, 17, 26–30.
- 100.
Voulodimos, A.; Doulamis, N.; Doulamis, A.; Protopapadakis, E. Deep Learning for Computer Vision: A Brief Review. Comput. Intell. Neurosci. 2018, 2018, 1–13. https://doi.org/10.1155/2018/7068349.
- 101.
Kaur, R.; Motl, R.W.; Sowers, R.; Hernandez, M.E. A Vision-Based Framework for Predicting Multiple Sclerosis and Parkinson’s Disease Gait Dysfunctions-A Deep Learning Approach. IEEE J. Biomed. Health Inform. 2023, 27, 190–201. https://doi.org/10.1109/JBHI.2022.3208077.
- 102.
Gao, Q.; Yao, S.; Tian, Y.; Zhang, C.; Zhao, T.; Wu, D.; Yu, G.; Lu, H. Automating General Movements Assessment with quantitative deep learning to facilitate early screening of cerebral palsy. Nat. Commun. 2023, 14, 8294. https://doi.org/10.1038/s41467-023-44141-x.
- 103.
Chen, J.; Soangra, R.; Grant-Beuttler, M.; Nanehkaran, Y.A.; Wen, Y. Dense & Attention Convolutional Neural Networks for Toe Walking Recognition. IEEE Trans. Neural Syst. Rehabil. Eng. 2023, 31, 2235–2245. https://doi.org/10.1109/TNSRE.2023.3272362.
- 104.
Zhao, A.; Li, J.; Dong, J.; Qi, L.; Zhang, Q.; Li, N.; Wang, X.; Zhou, H. Multimodal Gait Recognition for Neurodegenerative Diseases. IEEE Trans. Cybern. 2022, 52, 9439–9453. https://doi.org/10.1109/TCYB.2021.3056104.
- 105.
Hou, J.; Tian, Z. Application of recurrent neural network in predicting athletes’ sports achievement. J. Supercomput. 2021, 78, 5507–5525. https://doi.org/10.1007/s11227-021-04082-y.
- 106.
Tsai, Y.-H.; Wu, S.-K.; Yu, S.-S.; Tsai, M.-H. A Novel Hybrid Deep Neural Network for Predicting Athlete Performance Using Dynamic Brain Waves. Mathematics 2023, 11, 903. https://doi.org/10.3390/math11040903.
- 107.
Lee, K.; Kim, J.H.; Hong, H.; Jeong, Y.; Ryu, H.; Kim, H.; Lee, S.U. Deep learning model for classifying shoulder pain rehabilitation exercises using IMU sensor. J. Neuroeng. Rehabil. 2024, 21, 42. https://doi.org/10.1186/s12984-024-01343-8.
- 108.
Panwar, M.; Biswas, D.; Bajaj, H.; Jobges, M.; Turk, R.; Maharatna, K.; Acharyya, A. Rehab-Net: Deep Learning Framework for Arm Movement Classification Using Wearable Sensors for Stroke Rehabilitation. IEEE Trans. Biomed. Eng. 2019, 66, 3026–3037. https://doi.org/10.1109/TBME.2019.2899927.
- 109.
Wang, Y.; Wu, Q.; Dey, N.; Fong, S.; Ashour, A.S. Deep back propagation–long short-term memory network based upper-limb sEMG signal classification for automated rehabilitation. Biocybern. Biomed. Eng. 2020, 40, 987–1001. https://doi.org/10.1016/j.bbe.2020.05.003.
- 110.
Faruk, T.; Shum, L.C.; Iaboni, A.; Khan, S.S. Walking path images from real-time location data predict degree of cognitive impairment. Artif. Intell. Med. 2023, 144, 102657. https://doi.org/10.1016/j.artmed.2023.102657.
- 111.
Bae, J.H.; Seo, J.W.; Kim, D.Y. Deep-learning model for predicting physical fitness in possible sarcopenia: Analysis of the Korean physical fitness award from 2010 to 2023. Front Public Health 2023, 11, 1241388. https://doi.org/10.3389/fpubh.2023.1241388.
- 112.
Wang, Y.; Pei, Z.; Wang, C.; Tang, Z. Depth-aware pose estimation using deep learning for exoskeleton gait analysis. Sci. Rep. 2023, 13, 22681. https://doi.org/10.1038/s41598-023-50207-z.
- 113.
Washif, J.A.; Pagaduan, J.; James, C.; Dergaa, I.; Beaven, C.M. Artificial intelligence in sport: Exploring the potential of using ChatGPT in resistance training prescription. Biol. Sport. 2024, 41, 209–220. https://doi.org/10.5114/biolsport.2024.132987.
- 114.
Netz, Y.; Argov, E.; Yekutieli, Z.; Ayalon, M.; Tchelet, K.; Ben-Sira, D.; Amar, Y.; Jacobs, J.M. Personalized multicomponent exercise programs using smartphone technology among older people: Protocol for a randomized controlled trial. BMC Geriatr. 2021, 21, 605. https://doi.org/10.1186/s12877-021-02559-2.
- 115.
Tan, M.; Xiao, Y.; Jing, F.; Xie, Y.; Lu, S.; Xiang, M.; Ren, H. Evaluating machine learning-enabled and multimodal data-driven exercise prescriptions for mental health: A randomized controlled trial protocol. Front. Psychiatry 2024, 15, 1352420. https://doi.org/10.3389/fpsyt.2024.1352420.
- 116.
Liu, X.; Gao, B.; Suleiman, B.; You, H.; Ma, Z.; Liu, Y.; Anaissi, A. Privacy-preserving personalized fitness recommender system P3FitRec: A multi-level deep learning approach. ACM Trans. Knowl. Discov. Data 2023, 17, 1–24.
- 117.
Annegarn, J.; Spruit, M.A.; Savelberg, H.H.; Willems, P.J.; van de Bool, C.; Schols, A.M.; Wouters, E.F.; Meijer, K. Differences in walking pattern during 6-min walk test between patients with COPD and healthy subjects. PLoS ONE 2012, 7, e37329. https://doi.org/10.1371/journal.pone.0037329.
- 118.
Dalton, A.; Khalil, H.; Busse, M.; Rosser, A.; van Deursen, R.; Olaighin, G. Analysis of gait and balance through a single triaxial accelerometer in presymptomatic and symptomatic Huntington’s disease. Gait Posture 2013, 37, 49–54. https://doi.org/10.1016/j.gaitpost.2012.05.028.
- 119.
Kautz, T.; Groh, B.H.; Hannink, J.; Jensen, U.; Strubberg, H.; Eskofier, B.M. Activity recognition in beach volleyball using a Deep Convolutional Neural Network: Leveraging the potential of Deep Learning in sports. Data Min. Knowl. Discov. 2017, 31, 1678–1705.
- 120.
Zhang, W.; Su, C.; He, C. Rehabilitation Exercise Recognition and Evaluation Based on Smart Sensors With Deep Learning Framework. IEEE Access 2020, 8, 77561–77571. https://doi.org/10.1109/access.2020.2989128.
- 121.
Johnson, W.R.; Mian, A.; Lloyd, D.G.; Alderson, J.A. On-field player workload exposure and knee injury risk monitoring via deep learning. J. Biomech. 2019, 93, 185–193. https://doi.org/10.1016/j.jbiomech.2019.07.002.
- 122.
Jacob, S.; Menon, V.G.; Al-Turjman, F.; Vinoj, P.G.; Mostarda, L. Artificial Muscle Intelligence System With Deep Learning for Post-Stroke Assistance and Rehabilitation. IEEE Access 2019, 7, 133463–133473. https://doi.org/10.1109/access.2019.2941491.
- 123.
Liao, Y.; Vakanski, A.; Xian, M. A Deep Learning Framework for Assessing Physical Rehabilitation Exercises. IEEE Trans. Neural Syst. Rehabil. Eng. 2020, 28, 468–477. https://doi.org/10.1109/TNSRE.2020.2966249.
- 124.
Ruiz, J.R.; Ramirez-Lechuga, J.; Ortega, F.B.; Castro-Pinero, J.; Benitez, J.M.; Arauzo-Azofra, A.; Sanchez, C.; Sjostrom, M.; Castillo, M.J.; Gutierrez, A.; et al. Artificial neural network-based equation for estimating VO2max from the 20 m shuttle run test in adolescents. Artif. Intell. Med. 2008, 44, 233–245. https://doi.org/10.1016/j.artmed.2008.06.004.
- 125.
Hillen, B.; Lopez, D.A.; Schomer, E.; Nagele, M.; Simon, P. Towards Exercise Radiomics: Deep Neural Network-Based Automatic Analysis of Thermal Images Captured During Exercise. IEEE J. Biomed. Health Inform. 2022, 26, 4530–4540. https://doi.org/10.1109/JBHI.2022.3186530.
- 126.
Zhang, K.; Wang, J.; de Silva, C.W.; Fu, C. Unsupervised Cross-Subject Adaptation for Predicting Human Locomotion Intent. IEEE Trans. Neural Syst. Rehabil. Eng. 2020, 28, 646–657. https://doi.org/10.1109/TNSRE.2020.2966749.
- 127.
Del Din, S.; Godfrey, A.; Rochester, L. Validation of an Accelerometer to Quantify a Comprehensive Battery of Gait Characteristics in Healthy Older Adults and Parkinson’s Disease: Toward Clinical and at Home Use. IEEE J. Biomed. Health Inform. 2016, 20, 838–847. https://doi.org/10.1109/JBHI.2015.2419317.
- 128.
Stenum, J.; Rossi, C.; Roemmich, R.T. Two-dimensional video-based analysis of human gait using pose estimation. PLoS Comput. Biol. 2021, 17, e1008935. https://doi.org/10.1371/journal.pcbi.1008935.
- 129.
Simoni, L.; Scarton, A.; Macchi, C.; Gori, F.; Pasquini, G.; Pogliaghi, S. Quantitative and Qualitative Running Gait Analysis through an Innovative Video-Based Approach. Sensors 2021, 21, 2977. https://doi.org/10.3390/s21092977.
- 130.
Mennella, C.; Maniscalco, U.; Pietro, G.; Esposito, M. A deep learning system to monitor and assess rehabilitation exercises in home-based remote and unsupervised conditions. Comput. Biol. Med. 2023, 166, 107485. https://doi.org/10.1016/j.compbiomed.2023.107485.
- 131.
Przybyło, J. A deep learning approach for remote heart rate estimation. Biomed. Signal Process. Control 2022, 74, 103457. https://doi.org/10.1016/j.bspc.2021.103457.
- 132.
Bobić, V.; Ðurić-Jovičić, M.; Dragaševic-Mišković, N.; Kostić, V.S.; Kvaščev, G. Comparison of Two Deep Learning Models for the Recognition of Parkinson’s Disease Gait Patterns. In Proceedings of the 2023 10th International Conference on Electrical, Electronic and Computing Engineering (IcETRAN), East Sarajevo, Bosnia and Herzegovina, 5–8 June 2023; pp. 1–5.
- 133.
Vasudevan, P.; Faerie Mattins, R.; Srivarshan, S.; Narayanan, A.; Wadhwani, G.; Parvathi, R.; Maheswari, R. Gait Image Classification Using Deep Learning Models for Medical Diagnosis. Comput. Mater. Contin. 2023, 74, 6039–6063. https://doi.org/10.32604/cmc.2023.032331.
- 134.
Ordonez, F.J.; Roggen, D. Deep Convolutional and LSTM Recurrent Neural Networks for Multimodal Wearable Activity Recognition. Sensors 2016, 16, 115. https://doi.org/10.3390/s16010115.
- 135.
Yuan, H.; Chan, S.; Creagh, A.P.; Tong, C.; Acquah, A.; Clifton, D.A.; Doherty, A. Self-supervised learning for human activity recognition using 700,000 person-days of wearable data. npj Digit. Med. 2024, 7, 91.
- 136.
Liu, Y.; Qin, X.; Gao, Y.; Li, X.; Feng, C. SETransformer: A hybrid attention-based architecture for robust human activity recognition. arXiv 2025, arXiv:2505.19369.
- 137.
Zhang, H.; Zhuang, Z.; Wang, X.; Yang, X.; Zhang, Y. MoPFormer: Motion-Primitive Transformer for Wearable-Sensor Activity Recognition. arXiv 2025, arXiv:2505.20744.

This work is licensed under a Creative Commons Attribution 4.0 International License.


