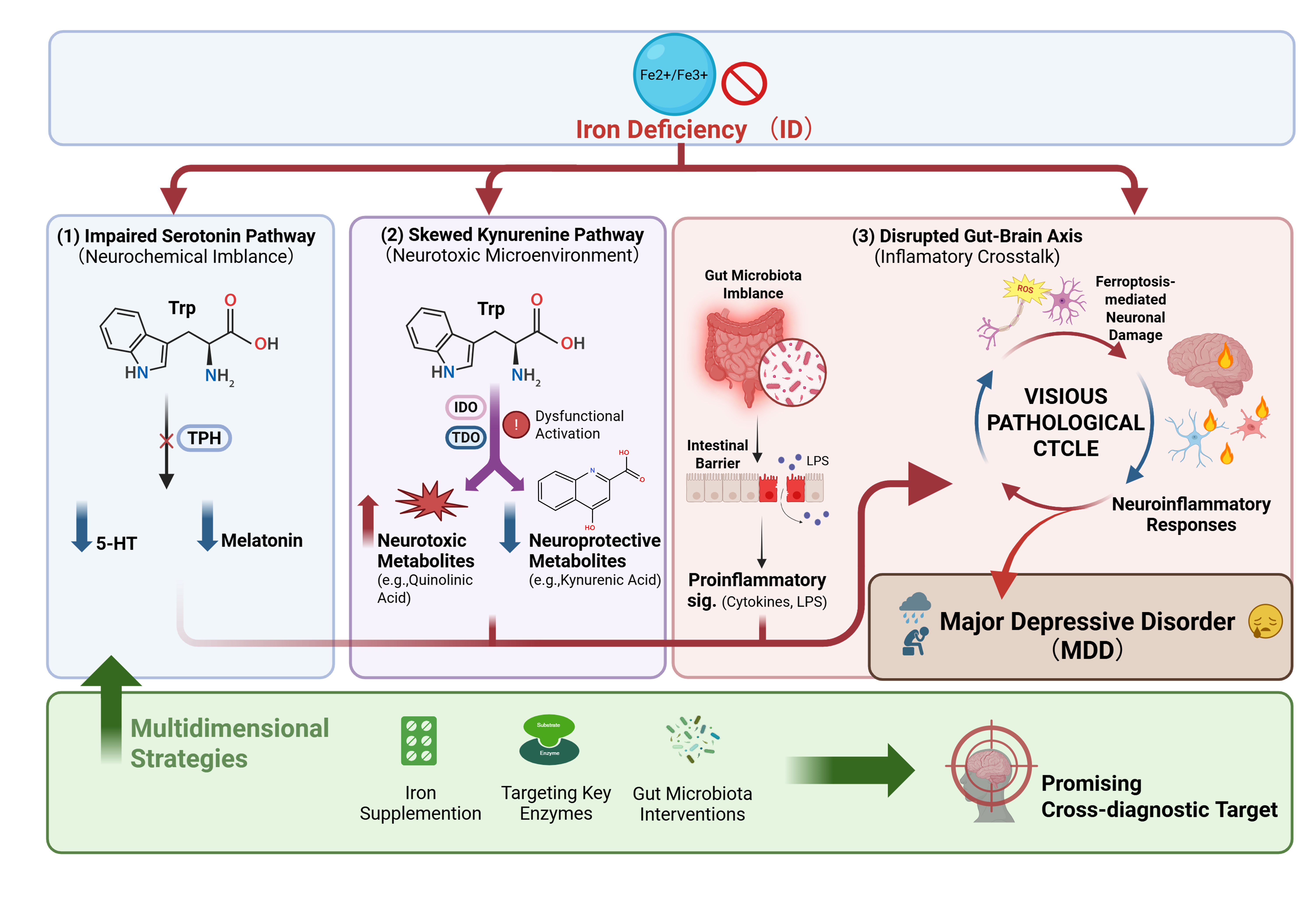
Major depressive disorder (MDD) ranks among the leading causes of disability worldwide. Current treatments often yield suboptimal outcomes, largely due to an incomplete understanding of its underlying pathology. There is, therefore, a pressing need to identify novel core pathological targets. Iron serves as an essential cofactor for several key enzymes in tryptophan (Trp) metabolism, playing a central role in its regulatory pathways. Iron deficiency (ID) can profoundly disrupt Trp metabolic homeostasis in both the peripheral and central nervous systems. This review synthesizes preclinical and clinical evidence to elucidate how ID drives the pathogenesis of MDD through the following interconnected mechanisms: (1) Impairing the activity of tryptophan hydroxylase (TPH), thereby reducing the synthesis of serotonin (5-hydroxytryptamine, 5-HT) and melatonin; (2) Skewing the kynurenine pathway (KP) flux toward neurotoxic metabolites via the “dysfunctional activation” of indoleamine 2,3-dioxygenase (IDO) and tryptophan 2,3-dioxygenase (TDO); (3) Disrupting gut microbiota-mediated indole metabolism, compromising intestinal barrier integrity, and amplifying neuroinflammatory responses. These metabolic disturbances collectively contribute to a vicious pathological cycle involving neurochemical imbalance, a neurotoxic microenvironment, peripheral-central inflammatory crosstalk, and ferroptosis-mediated neuronal damage, ultimately entrenching the depressive phenotype. Furthermore, this review outlines multidimensional therapeutic strategies targeting the iron-Trp metabolic axis for depression. In conclusion, we propose that the iron-Trp metabolic axis represents a promising cross-diagnostic target for MDD, offering new theoretical insights and practical avenues for its precision treatment.
- Open Access
- Review
Iron-Dependent Regulation of Tryptophan Metabolism in Depression
- Yan Zhang 1,†,
- Jinping Liu 2,†,
- Mingqian Fang 2,*,
- Ren Lai 2,*
Author Information
Received: 07 Jan 2026 | Revised: 06 Mar 2026 | Accepted: 10 Mar 2026 | Published: 24 Apr 2026
Abstract
Graphical Abstract
Keywords
References
- 1.
Irwin, C.L.; Coelho, P.S.; Kluwe-Schiavon, B.; Silva-Fernandes, A.; Goncalves, O.F.; Leite, J.; Carvalho, S. Non-pharmacological treatment-related changes of molecular biomarkers in major depressive disorder: A systematic review and meta-analysis. Int. J. Clin. Health Psychol. 2023, 23, 100367.
- 2.
Penninx, B.W.J.H.; Lamers, F.; Jansen, R.; Berk, M.; Khandaker, G.M.; De Picker, L.; Milaneschi, Y. Immuno-metabolic depression: From concept to implementation. Lancet Reg. Health Eur. 2025, 48, 101166.
- 3.
Rahman, S.; Alzarea, S. Glial mechanisms underlying major depressive disorder: Potential therapeutic opportunities. Prog. Mol. Biol. Transl. Sci. 2019, 167, 159–178.
- 4.
Komleva, P.D.; Deeb, R.; Terentieva, E.I.; Kulikov, A.V. Comparison of the Effects of Tetrahydrobiopterine, L-Tryptophan, and Iron Ions on the Thermal Stability of Wild Type and P447R Mutant Tryptophan Hydroxylase 2. Bull. Exp. Biol. Med. 2025, 178, 447–452.
- 5.
Yan, D.; Lin, Y.; Tan, X. Heme-containing enzymes and inhibitors for tryptophan metabolism. Metallomics 2017, 9, 1230–1240.
- 6.
Heimberger, A.B.; Lukas, R.V. The kynurenine pathway implicated in patient delirium: Possible indications for indoleamine 2,3 dioxygenase inhibitors. J. Clin. Investig. 2023, 133, e167845.
- 7.
Li, G.; Wu, Y.; Huang, X.; Du, M.; Tang, H. A gut-brain-gut axis orchestrates host responses counteracting microbiome-induced iron insufficiency. EMBO J. 2025, 44, 7590–7619.
- 8.
Bonilla, D.A.; Moreno, Y.; Petro, J.L.; Forero, D.A.; Vargas-Molina, S.; Odriozola-Martínez, A.; Orozco, C.A.; Stout, J.R.; Rawson, E.S.; Kreider, R.B. A Bioinformatics-Assisted Review on Iron Metabolism and Immune System to Identify Potential Biomarkers of Exercise Stress-Induced Immunosuppression. Biomedicines 2022, 10, 724.
- 9.
Behmoaras, J. The versatile biochemistry of iron in macrophage effector functions. FEBS J. 2021, 288, 6972–6989.
- 10.
Su, Q.; Gu, Y.; Yu, B.; Yu, F.; He, H.; Zhang, Q.; Meng, G.; Wu, H.; Du, H.; Liu, L.; et al. Association between Serum Ferritin Concentrations and Depressive Symptoms among Chinese Adults: A Population Study from the Tianjin Chronic Low-Grade Systemic Inflammation and Health (TCLSIHealth) Cohort Study. PLoS ONE 2016, 11, e0162682.
- 11.
Michels, N.; Clarke, G.; Olavarria-Ramirez, L.; Gomez-Martinez, S.; Diaz, L.E.; Marcos, A.; Widhalm, K.; Carvalho, L.A. Psychosocial stress and inflammation driving tryptophan breakdown in children and adolescents: A cross-sectional analysis of two cohorts. Psychoneuroendocrinology 2018, 94, 104–111.
- 12.
Haverkamp, G.L.; Loosman, W.L.; Schouten, R.W.; Franssen, C.F.; Kema, I.P.; van Diepen, M.; Dekker, F.W.; Siegert, C.E.; Honig, A. Differences in the association of inflammation and tryptophan with depressive symptoms between white and non-white chronic dialysis patients. Gen. Hosp. Psychiatry 2018, 50, 76–82.
- 13.
Specker, E.; Matthes, S.; Wesolowski, R.; Schutz, A.; Grohmann, M.; Alenina, N.; Pleimes, D.; Mallow, K.; Neuenschwander, M.; Gogolin, A.; et al. Structure-Based Design of Xanthine-Benzimidazole Derivatives as Novel and Potent Tryptophan Hydroxylase Inhibitors. J. Med. Chem. 2022, 65, 11126–11149.
- 14.
Cao, Y.; Popp, O.; Milani, N.; Qadri, F.; Kuhn, R.; Mertins, P.; Bader, M.; Alenina, N. Hyperphenylalaninemia and serotonin deficiency in Dnajc12-deficient mice. Commun. Biol. 2024, 7, 1641.
- 15.
Iyer, S.R.; Tidemand, K.D.; Babicz, J.T.; Jacobs, A.B.; Gee, L.B.; Haahr, L.T.; Yoda, Y.; Kurokuzu, M.; Kitao, S.; Saito, M.; et al. Direct coordination of pterin to Fe(II) enables neurotransmitter biosynthesis in the pterin-dependent hydroxylases. Proc. Natl. Acad. Sci. USA 2021, 118, e2022379118.
- 16.
Hasegawa, H.; Ichiyama, A. Distinctive iron requirement of tryptophan 5-monooxygenase: TPH1 requires dissociable ferrous iron. Biochem. Biophys. Res. Commun. 2005, 338, 277–284.
- 17.
Zhang, H.; He, L.; Li, S.; Zhai, M.; Ma, S.; Jin, G.; Li, M.; Zhou, F.; Tian, H.; Nuerkaman, T.; et al. Cerebral iron deficiency may induce depression through downregulation of the hippocampal glucocorticoid-glucocorticoid receptor signaling pathway. J. Affect. Disord. 2023, 332, 125–135.
- 18.
Markov, D.D.; Yatsenko, K.A.; Inozemtseva, L.S.; Grivennikov, I.A.; Myasoedov, N.F.; Dolotov, O.V. Systemic N-terminal fragments of adrenocorticotropin reduce inflammation- and stress-induced anhedonia in rats. Psychoneuroendocrinology 2017, 82, 173–186.
- 19.
Peng, Q.; Yan, H.; Wen, Y.; Lai, C.; Shi, L. Association between NR3C1 rs41423247 polymorphism and depression: A PRISMA-compliant meta-analysis. Medicine 2018, 97, e12541.
- 20.
Sun, X.; Zu, Y.; Li, X.; Zhao, S.; Sun, X.; Li, L.; Zhang, X.; Wang, W.; Liang, Y.; Wang, W.; et al.Corticosterone-induced Hippocampal 5-HT Responses were Muted in Depressive-like State. ACS Chem. Neurosci. 2021, 12, 845–856.
- 21.
Nishikura, N.; Hino, K.; Kimura, T.; Uchimura, Y.; Hino, S.; Nakao, M.; Maruo, Y.; Udagawa, J. Postweaning Iron Deficiency in Male Rats Leads to Long-Term Hyperactivity and Decreased Reelin Gene Expression in the Nucleus Accumbens. J. Nutr. 2020, 150, 212–221.
- 22.
Aboagye, B.; Weber, T.; Merdian, H.L.; Bartsch, D.; Lesch, K.P.; Waider, J. Serotonin deficiency induced after brain maturation rescues consequences of early life adversity. Sci. Rep. 2021, 11, 5368.
- 23.
Hu, Y.W.; Liu, J.; Qiu, Z.H.; Li, X.Y.; Li, J.; Chen, L.; Wang, T.; Wang, X.F.; Feng, Z.J.; Bai, W.T.; et al. Effects of astrocytes in the dorsal hippocampus on anxiety-like and depressive-like behaviors in hemiparkinsonian rats. Behav. Brain Res. 2025, 486, 115553.
- 24.
Dolsak, A.; Gobec, S.; Sova, M. Indoleamine and tryptophan 2,3-dioxygenases as important future therapeutic targets. Pharmacol. Ther. 2021, 221, 107746.
- 25.
Kennedy, P.J.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Kynurenine pathway metabolism and the microbiota-gut-brain axis. Neuropharmacology 2017, 112, 399–412.
- 26.
Kudo, Y.; Koh, I.; Sugimoto, J. Localization of Indoleamine 2,3-Dioxygenase-1 and Indoleamine 2,3-Dioxygenase-2 at the Human Maternal-Fetal Interface. Int. J. Tryptophan Res. 2020, 13, 1178646920984163.
- 27.
Nelp, M.T.; Zheng, V.; Davis, K.M.; Stiefel, W.K.; Groves, J.T. Potent Activation of Indoleamine 2,3-Dioxygenase by Polysulfides. J. Am. Chem. Soc. 2019, 141, 15288–15300.
- 28.
Sin, R.; Sotogaku, N.; Ohnishi, Y.N.; Shuto, T.; Kuroiwa, M.; Kawahara, Y.; Sugiyama, K.; Murakami, Y.; Kanai, M.; Funakoshi, H.; et al. Inhibition of STAT-mediated cytokine responses to chemically-induced colitis prevents inflammation-associated neurobehavioral impairments. Brain Behav. Immun. 2023, 114, 173–186.
- 29.
Okamoto, N.; Hoshikawa, T.; Honma, Y.; Chibaatar, E.; Ikenouchi, A.; Harada, M.; Yoshimura, R. Effect modification of tumor necrosis factor-α on the kynurenine and serotonin pathways in major depressive disorder on type 2 diabetes mellitus. Eur. Arch. Psychiatry Clin. Neurosci. 2024, 274, 1697–1707.
- 30.
Erhardt, S.; Schwieler, L.; Imbeault, S.; Engberg, G. The kynurenine pathway in schizophrenia and bipolar disorder. Neuropharmacology 2017, 112, 297–306.
- 31.
Lanser, L.; Kink, P.; Egger, E.M.; Willenbacher, W.; Fuchs, D.; Weiss, G.; Kurz, K. Inflammation-Induced Tryptophan Breakdown is Related With Anemia, Fatigue, and Depression in Cancer. Front. Immunol. 2020, 11, 249.
- 32.
Öztürk, M.; Yalın Sapmaz, Ş.; Kandemir, H.; Taneli, F.; Aydemir, O. The role of the kynurenine pathway and quinolinic acid in adolescent major depressive disorder. Int. J. Clin. Pract. 2021, 75, e13739.
- 33.
Brown, S.J.; Huang, X.; Newell, K.A. The kynurenine pathway in major depression: What we know and where to next. Neurosci. Biobehav. Rev. 2021, 127, 917–927.
- 34.
Ahmad, M.H.; Rizvi, M.A.; Fatima, M.; Mondal, A.C. Pathophysiological implications of neuroinflammation mediated HPA axis dysregulation in the prognosis of cancer and depression. Mol. Cell. Endocrinol. 2021, 520, 111093.
- 35.
Liu, X.C.; Erhardt, S.; Goiny, M.; Engberg, G.; Mathé, A.A. Decreased levels of kynurenic acid in prefrontal cortex in a genetic animal model of depression. Acta Neuropsychiatr. 2017, 29, 54–58.
- 36.
Wu, Y.; Zhong, X.; Mai, N.; Wen, Y.; Shang, D.; Hu, L.; Chen, B.; Zhang, M.; Ning, Y. Kynurenine pathway changes in late-life depression. J. Affect. Disord. 2018, 235, 76–81.
- 37.
Wang, A.; Guan, C.; Wang, T.; Mu, G.; Tuo, Y. Indole-3-Lactic Acid, a Tryptophan Metabolite of Lactiplantibacillus plantarum DPUL-S164, Improved Intestinal Barrier Damage by Activating AhR and Nrf2 Signaling Pathways. J. Agric. Food Chem. 2023, 71, 18792–18801.
- 38.
Kou, R.W.; Li, Z.Q.; Wang, J.L.; Jiang, S.Q.; Zhang, R.J.; He, Y.Q.; Xia, B.; Gao, J.M. Ganoderic Acid A Mitigates Inflammatory Bowel Disease through Modulation of AhR Activity by Microbial Tryptophan Metabolism. J. Agric. Food Chem. 2024, 72, 17912–17923.
- 39.
Wang, X.; Huang, S.; Zhang, M.; Su, Y.; Pan, Z.; Liang, J.; Xie, X.; Wang, Q.; Chen, J.; Zhou, L.; et al. Gegen Qinlian decoction activates AhR/IL-22 to repair intestinal barrier by modulating gut microbiota-related tryptophan metabolism in ulcerative colitis mice. J. Ethnopharmacol. 2023, 302, 115919.
- 40.
Peng, M.; Fu, J.; Song, Y.; Lu, M.; Zhang, B. Diosmetin Alleviates Cerebral Ischemia/Reperfusion Injury by Modulating Tryptophan Metabolism Through the Microbiota-Gut-Brain Axis. J. Agric. Food Chem. 2025, 73, 22448–22465.
- 41.
Xiao, L.; Tang, R.; Wang, J.; Wan, D.; Yin, Y.; Xie, L. Gut Microbiota Bridges the Iron Homeostasis and Host Health. Sci. China Life Sci. 2023, 66, 1952–1975.
- 42.
Bardhan, P.; Mei, X.; Lai, N.K.; Mell, B.; Tummala, R.; Aryal, S.; Manandhar, I.; Hwang, H.; Jhuma, T.A.; Atluri, R.R.; et al. Salt-Responsive Gut Microbiota Induces Sex-Specific Blood Pressure Changes. Circ. Res. 2024, 135, 1122–1137.
- 43.
Mayneris-Perxachs, J.; Moreno-Navarrete, J.M.; Fernandez-Real, J.M. The Role of Iron in Host-Microbiota Crosstalk and Its Effects on Systemic Glucose Metabolism. Nat. Rev. Endocrinol. 2022, 18, 683–698.
- 44.
Ranhotra, H.S. Discrete Interplay of Gut Microbiota L-Tryptophan Metabolites in Host Biology and Disease. Mol. Cell. Biochem. 2024, 479, 2273–2290.
- 45.
Quarta, E.; Bourqqia-Ramzi, M.; Munoz-Rodriguez, D.; Garcia-Esteban, M.T.; Murciano-Cespedosa, A.; Mateos Gonzalez, A.; Conejero-Meca, F.J.; Lombardo-Hernandez, J.; Mansilla-Guardiola, J.; Baroni, S.; et al. Impact of Iron Deficiency on the Growth and Bioelectrical Profile of Different Gut Bacteria. Microbiologyopen 2025, 14, e70015.
- 46.
Hassamal, S. Chronic Stress, Neuroinflammation, and Depression: An Overview of Pathophysiological Mechanisms and Emerging Anti-Inflammatories. Front. Psychiatry 2023, 14, 1130989.
- 47.
Tremblay, M.E.; Madore, C.; Bordeleau, M.; Tian, L.; Verkhratsky, A. Neuropathobiology of COVID-19: The Role for Glia. Front. Cell. Neurosci. 2020, 14, 592214.
- 48.
Liu, J.R.; Miao, H.; Deng, D.Q.; Vaziri, N.D.; Li, P.; Zhao, Y.Y. Gut Microbiota-Derived Tryptophan Metabolism Mediates Renal Fibrosis by Aryl Hydrocarbon Receptor Signaling Activation. Cell. Mol. Life Sci. 2021, 78, 909–922.
- 49.
Roager, H.M.; Licht, T.R. Microbial tryptophan catabolites in health and disease. Nat. Commun. 2018, 9, 3294.
- 50.
Wu, L.; Hu, Z.; Lv, Y.; Ge, C.; Luo, X.; Zhan, S.; Huang, W.; Shen, X.; Yu, D.; Liu, B. Hericium erinaceus Polysaccharides Ameliorate Nonalcoholic Fatty Liver Disease via Gut Microbiota and Tryptophan Metabolism Regulation in an Aged Laying Hen Model. Int. J. Biol. Macromol. 2024, 273, 132735.
- 51.
Wang, L.; Zhang, Y.; Ran, Y.; Li, L.; Mei, L.; Ye, F.; Sun, Y.; Wang, T.; Quan, X.; Shi, H.; et al. Association Between AHR in EGCs and IBS-D Patients: The Indole Pathway of Tryptophan Metabolism. Front. Nutr. 2025, 12, 1566595.
- 52.
Bader, M. Inhibition of Serotonin Synthesis: A Novel Therapeutic Paradigm. Pharmacol. Ther. 2020, 205, 107423.
- 53.
Shah, H.E.; Bhawnani, N.; Ethirajulu, A.; Alkasabera, A.; Onyali, C.B.; Anim-Koranteng, C.; Mostafa, J.A. Iron Deficiency-Induced Changes in the Hippocampus, Corpus Striatum, and Monoamines Levels That Lead to Anxiety, Depression, Sleep Disorders, and Psychotic Disorders. Cureus 2021, 13, e18138.
- 54.
Sen, A. Deficient Synthesis of Melatonin in COVID-19 Can Impair the Resistance of Coronavirus Patients to Mucormycosis. Med. Hypotheses 2021, 158, 110722.
- 55.
Wu, Q.; Ren, Q.; Wang, X.; Bai, H.; Tian, D.; Gao, G.; Wang, F.; Yu, P.; Chang, Y.Z. Cellular Iron Depletion Enhances Behavioral Rhythm by Limiting Brain Per1 Expression in Mice. CNS Neurosci. Ther. 2024, 30, e14592.
- 56.
Leung, W.; Singh, I.; McWilliams, S.; Stockler, S.; Ipsiroglu, O.S. Iron Deficiency and Sleep—A Scoping Review Sleep. Med. Rev. 2020, 51, 101274.
- 57.
Jiao, H.; Yan, Z.; Ma, Q.; Li, X.; Jiang, Y.; Liu, Y.; Chen, J. Influence of Xiaoyaosan on Depressive-Like Behaviors in Chronic Stress-Depressed Rats Through Regulating Tryptophan Metabolism in Hippocampus. Neuropsychiatr. Dis. Treat. 2019, 15, 21–31.
- 58.
De la Fuente Munoz, M.; Roman-Carmena, M.; Amor, S.; Garcia-Villalon, A.L.; Espinel, A.E.; Gonzalez-Hedstrom, D.; Granado Garcia, M. Effects of Supplementation with the Standardized Extract of Saffron (affron®) on the Kynurenine Pathway and Melatonin Synthesis in Rats. Antioxidants 2023, 12, 1619.
- 59.
Lieberman, H.R.; Agarwal, S.; Fulgoni, V.L. Tryptophan Intake in the US Adult Population Is Not Related to Liver or Kidney Function but Is Associated with Depression and Sleep Outcomes. J. Nutr. 2016, 146, 2609S–2615S.
- 60.
Phillips, R.S.; Iradukunda, E.C.; Hughes, T.; Bowen, J.P. Modulation of Enzyme Activity in the Kynurenine Pathway by Kynurenine Monooxygenase Inhibition. Front. Mol. Biosci. 2019, 6, 3.
- 61.
Zheng, H.; Teague, T.K.; Yeh, F.C.; Burrows, K.; Figueroa-Hall, L.K.; Aupperle, R.L.; Khalsa, S.S.; Paulus, M.P.; Savitz, J. C-Reactive Protein and the Kynurenic Acid to Quinolinic Acid Ratio Are Independently Associated with White Matter Integrity in Major Depressive Disorder. Brain Behav. Immun. 2022, 105, 180–189.
- 62.
Wenninger, J.; Meinitzer, A.; Holasek, S.; Schnedl, W.J.; Zelzer, S.; Mangge, H.; Herrmann, M.; Enko, D. Associations between Tryptophan and Iron Metabolism Observed in Individuals with and without Iron Deficiency. Sci. Rep. 2019, 9, 14548.
- 63.
Busse, M.; Hettler, V.; Fischer, V.; Mawrin, C.; Hartig, R.; Dobrowolny, H.; Bogerts, B.; Frodl, T.; Busse, S. Increased Quinolinic Acid in Peripheral Mononuclear Cells in Alzheimer's Dementia. Eur. Arch. Psychiatry Clin. Neurosci. 2018, 268, 493–500.
- 64.
Fukuwatari, T. Possibility of Amino Acid Treatment to Prevent the Psychiatric Disorders via Modulation of the Production of Tryptophan Metabolite Kynurenic Acid. Nutrients 2020, 12, 1403.
- 65.
Kim, Y.; Won, E. The Influence of Stress on Neuroinflammation and Alterations in Brain Structure and Function in Major Depressive Disorder. Behav. Brain Res. 2017, 329, 6–11.
- 66.
Dey, S.; Banerjee Dixit, A.; Tripathi, M.; Doddamani, R.S.; Sharma, M.C.; Lalwani, S.; Chandra, P.S.; Banerjee, J. Altered Hippocampal Kynurenine Pathway Metabolism Contributes to Hyperexcitability in Human Mesial Temporal Lobe Epilepsy-Hippocampal Sclerosis. Br. J. Pharmacol. 2021, 178, 3959–3976.
- 67.
Brown, S.J.; Brown, A.M.; Purves-Tyson, T.D.; Huang, X.F.; Shannon Weickert, C.; Newell, K.A. Alterations in the Kynurenine Pathway and Excitatory Amino Acid Transporter-2 in Depression with and without Psychosis: Evidence of a Potential Astrocyte Pathology. J. Psychiatr. Res. 2022, 147, 203–211.
- 68.
Bansal, Y.; Singh, R.; Parhar, I.; Kuhad, A.; Soga, T. Quinolinic Acid and Nuclear Factor Erythroid 2-Related Factor 2 in Depression: Role in Neuroprogression. Front. Pharmacol. 2019, 10, 452.
- 69.
Kezai, A.M.; Badiane, P.Y.; Hennart, B.; Allorge, D.; Marion, S.; Hebert, S.S. MicroRNA-132 Regulates Quinolinic Acid Production in the Brain during LPS-Induced Neuroinflammation. Front. Immunol. 2025, 16, 1644783.
- 70.
Rodrigues, F.T.S.; de Souza, M.R.M.; Lima, C.N.C.; da Silva, F.E.R.; Costa, D.V.D.S.; Dos Santos, C.C.; Miyajima, F.; de Sousa, F.C.F.; Vasconcelos, S.M.M.; Barichello, T.; et al. Major Depression Model Induced by Repeated and Intermittent Lipopolysaccharide Administration: Long-Lasting Behavioral, Neuroimmune and Neuroprogressive Alterations. J. Psychiatr. Res. 2018, 107, 57–67.
- 71.
Ahmed, A.; Misrani, A.; Tabassum, S.; Yang, L.; Long, C. Minocycline inhibits sleep deprivation-induced aberrant microglial activation and Keap1-Nrf2 expression in mouse hippocampus. Brain Res. Bull. 2021, 174, 41–52.
- 72.
Scuderi, S.A.; Ardizzone, A.; Calcaterra, E.; Palermo, N.; De Luca, F.; Catalfamo, A.; Esposito, E.; Capra, A.P. The Gut-Brain Axis in Brain Tumors: Insights into Tumor Development, Progression, and Therapy. Biomedicines 2025, 13, 2172.
- 73.
Murray, T.E.; Richards, C.M.; Robert-Gostlin, V.N.; Bernath, A.K.; Lindhout, I.A.; Klegeris, A. Potential neurotoxic activity of diverse molecules released by astrocytes. Brain Res. Bull. 2022, 189, 80–101.
- 74.
Chen, Y.; Schlotterer, A.; Kurowski, L.; Li, L.; Dannehl, M.; Hammes, H.P.; Lin, J. miRNA-124 Prevents Rat Diabetic Retinopathy by Inhibiting the Microglial Inflammatory Response. Int. J. Mol. Sci. 2023, 24, 2291.
- 75.
Dos Santos Souza, C.; Grangeiro, M.S.; Lima Pereira, E.P.; Dos Santos, C.C.; da Silva, A.B.; Sampaio, G.P.; Ribeiro Figueiredo, D.D.; David, J.M.; David, J.P.; da Silva, V.D.A.; et al. Agathisflavone, a flavonoid derived from Poincianella pyramidalis (Tul.), enhances neuronal population and protects against glutamate excitotoxicity. Neurotoxicology 2018, 65, 85–97.
- 76.
Efremova, L.; Chovancova, P.; Adam, M.; Gutbier, S.; Schildknecht, S.; Leist, M. Switching from astrocytic neuroprotection to neurodegeneration by cytokine stimulation. Arch. Toxicol. 2017, 91, 231–246.
- 77.
Rodriguez-Zas, S.L.; Wu, C.; Southey, B.R.; O'Connor, J.C.; Nixon, S.E.; Garcia, R.; Zavala, C.; Lawson, M.; McCusker, R.H.; Romanova, E.V.; et al. Disruption of microglia histone acetylation and protein pathways in mice exhibiting inflammation-associated depression-like symptoms. Psychoneuroendocrinology 2018, 97, 47–58.
- 78.
Savonije, K.; Meek, A.; Weaver, D.F. Indoleamine 2,3-Dioxygenase as a Therapeutic Target for Alzheimer’s Disease and Geriatric Depression. Brain Sci. 2023, 13, 852.
- 79.
Bansal, Y.; Singh, R.; Sodhi, R.K.; Khare, P.; Dhingra, R.; Dhingra, N.; Bishnoi, M.; Kondepudi, K.K.; Kuhad, A. Kynurenine monooxygenase inhibition and associated reduced quinolinic acid reverses depression-like behaviour by upregulating Nrf2/ARE pathway in mouse model of depression: In-vivo and In-silico studies. Neuropharmacology 2022, 215, 109169.
- 80.
Liu, X.; Luo, Q.; Zhao, Y.; Ren, P.; Jin, Y.; Zhou, J. The Ferroptosis-Mitochondrial Axis in Depression: Unraveling the Feedforward Loop of Oxidative Stress, Metabolic Homeostasis Dysregulation, and Neuroinflammation. Antioxidants 2025, 14, 613.
- 81.
Liu, D.; Liang, C.H.; Huang, B.; Zhuang, X.; Cui, W.; Yang, L.; Yang, Y.; Zhang, Y.; Fu, X.; Zhang, X.; et al. Tryptophan Metabolism Acts as a New Anti-Ferroptotic Pathway to Mediate Tumor Growth. Adv. Sci. 2023, 10, e2204006.
- 82.
Wen, J.; Li, L.; Ou, D.; Li, J.; Yang, Y.; Duan, L.; Zhang, X.; Zhu, Y.; Hao, J.; Tong, Y. Higenamine protects against doxorubicin-induced heart failure by attenuating ferroptosis via modulating the Nrf2/GPX4 signaling pathway. Phytomedicine 2025, 141, 156670.
- 83.
Zhou, A.; Feng, H.Y.; Fan, C.N.; Wang, J.; Yuan, Z.Y.; Xu, G.H.; Li, C.F.; Huang, W.F.; Yi, L.T. Asiaticoside Attenuates Chronic Restraint Stress-Induced Hippocampal CA1 Neuronal Ferroptosis via Activating BDNF/Nrf2/GPX4 Signaling Pathway. Drug Des. Devel. Ther. 2025, 19, 793–810.
- 84.
Ma, T.; Du, J.; Zhang, Y.; Wang, Y.; Wang, B.; Zhang, T. GPX4-independent ferroptosis—A new strategy in disease’s therapy. Cell Death Discov. 2022, 8, 434.
- 85.
Ye, L.; Wen, X.; Qin, J.; Zhang, X.; Wang, Y.; Wang, Z.; Zhou, T.; Di, Y.; He, W. Metabolism-regulated ferroptosis in cancer progression and therapy. Cell Death Discov. 2024, 15, 196.
- 86.
Wang, L.; Xu, R.; Huang, C.; Yi, G.; Li, Z.; Zhang, H.; Ye, R.; Qi, S.; Huang, G.; Qu, S. Targeting the ferroptosis crosstalk: Novel alternative strategies for the treatment of major depressive disorder. Gen. Psychiatry 2023, 36, e101072.
- 87.
Dang, R.; Wang, M.; Li, X.; Wang, H.; Liu, L.; Wu, Q.; Zhao, J.; Ji, P.; Zhong, L.; Licinio, J.; et al. Edaravone ameliorates depressive and anxiety-like behaviors via Sirt1/Nrf2/HO-1/Gpx4 pathway. J. Neuroinflammation 2022, 19, 41.
- 88.
Xu, K.; Liu, G.; Fu, C. The Tryptophan Pathway Targeting Antioxidant Capacity in the Placenta. Oxid. Med. Cell. Longev. 2018, 2018, 1054797.
- 89.
Cheng, Y.; Song, Y.; Chen, H.; Li, Q.; Gao, Y.; Lu, G.; Luo, C. Ferroptosis Mediated by Lipid Reactive Oxygen Species: A Possible Causal Link of Neuroinflammation to Neurological Disorders. Oxid. Med. Cell. Longev. 2021, 2021, 5005136.
- 90.
Wang, M.; Tang, G.; Zhou, C.; Guo, H.; Hu, Z.; Hu, Q.; Li, G. Revisiting the intersection of microglial activation and neuroinflammation in Alzheimer’s disease from the perspective of ferroptosis. Chem. Biol. Interact. 2023, 375, 110387.
- 91.
Xue, C.; He, Z.; Zeng, M.; Wang, Z.; Chen, Q.; Qin, F.; Chen, M.; Ye, H.; Chen, J. The Protective Effects of Polygala tenuifolia and Tenuifolin on Corticosterone-Evoked Ferroptosis, Oxidative Stress, and Neuroinflammation: Insights from Molecular Dynamics Simulations and In Vitro Experiments. Foods 2024, 13, 3358.
- 92.
Li, T.; Wu, Q.; Chen, Y.; Zou, J.; Zhang, E. Efficacy and Safety of Shengxuening Combined with Conventional Iron Supplementation in the Treatment of Anemia during Pregnancy: A Systematic Review and Meta-analysis. Evid. Based Complement. Alternat. Med. 2022, 2022, 1773616.
- 93.
Komleva, P.D.; Terentieva, E.I.; Izyurov, A.E.; Kulikov, A.V. Iron Ions Increase the Thermal Stability In Vitro and Activity In Vivo of the 447R Mutant Form of Mouse Tryptophan Hydroxylase 2. Int. J. Mol. Sci. 2025, 26, 8188.
- 94.
Fang, X.; Wu, Y.; Dai, Y.; Xiao, H.; Li, S.; Chen, X.; Yuan, M.; Guo, Y.; Ma, L.; Lin, D.; et al. In Situ Recovery of Serotonin Synthesis by a Tryptophan Hydroxylase-Like Nanozyme for the Treatment of Depression. J. Am. Chem. Soc. 2025, 147, 9111–9121.
- 95.
Lee, H.S.; Chao, H.H.; Huang, W.T.; Chen, S.C.; Yang, H.Y. Psychiatric disorders risk in patients with iron deficiency anemia and association with iron supplementation medications: A nationwide database analysis. BMC Psychiatry 2020, 20, 216.
- 96.
Kassir, A. Iron deficiency: A diagnostic and therapeutic perspective in psychiatry. Encephale 2017, 43, 85–89.
- 97.
Subedi, B.P.; Fitzpatrick, P.F. Mutagenesis of an Active-Site Loop in Tryptophan Hydroxylase Dramatically Slows the Formation of an Early Intermediate in Catalysis. J. Am. Chem. Soc. 2018, 140, 5185–5192.
- 98.
Scherer, T.; Allegri, G.; Sarkissian, C.N.; Ying, M.; Grisch-Chan, H.M.; Rassi, A.; Winn, S.R.; Harding, C.O.; Martinez, A.; Thony, B. Tetrahydrobiopterin treatment reduces brain L-Phe but only partially improves serotonin in hyperphenylalaninemic ENU1/2 mice. J. Inherit. Metab. Dis. 2018, 41, 709–718.
- 99.
Cronin, S.J.F.; Seehus, C.; Weidinger, A.; Talbot, S.; Reissig, S.; Seifert, M.; Pierson, Y.; McNeill, E.; Longhi, M.S.; Turnes, B.L.; et al. The metabolite BH4 controls T cell proliferation in autoimmunity and cancer. Nature 2018, 563, 564–568.
- 100.
Hu, Q.; Wei, W.; Wu, D.; Huang, F.; Li, M.; Li, W.; Yin, J.; Peng, Y.; Lu, Y.; Zhao, Q.; et al. Blockade of GCH1/BH4 Axis Activates Ferritinophagy to Mitigate the Resistance of Colorectal Cancer to Erastin-Induced Ferroptosis. Front. Cell Dev. Biol. 2022, 10, 810327.
- 101.
Matsuda, A.; Hata, A.; Tanaka, A.; Matsuda, H. Canine mast cell tumour cells regulate tryptophan catabolism via the expression of indoleamine 2,3-dioxygenase. Res. Vet. Sci. 2021, 137, 159–162.
- 102.
Qin, Y.; Hu, X.; Zhao, H.L.; Kurban, N.; Chen, X.; Yi, J.K.; Zhang, Y.; Cui, S.Y.; Zhang, Y.H. Inhibition of Indoleamine 2,3-Dioxygenase Exerts Antidepressant-like Effects through Distinct Pathways in Prelimbic and Infralimbic Cortices in Rats under Intracerebroventricular Injection with Streptozotocin. Int. J. Mol. Sci. 2024, 25, 7496.
- 103.
Laugeray, A.; Launay, J.M.; Callebert, J.; Mutlu, O.; Guillemin, G.J.; Belzung, C.; Barone, P.R. Chronic Treatment with the IDO1 Inhibitor 1-Methyl-D-Tryptophan Minimizes the Behavioural and Biochemical Abnormalities Induced by Unpredictable Chronic Mild Stress in Mice—Comparison with Fluoxetine. PLoS ONE 2016, 11, e0164337.
- 104.
Amin, S.A.; Val, J.P.G.A.; Martins, J.P.A.; Piotto, S. First 4D-QSAR Study of Human Kynurenine 3 Monooxygenase (hKMO) Inhibitors: Integrating Chemical Space Networks and an Explainable Artificial Intelligence Platform for Neurodegenerative Disease Drug Discovery. ACS Omega 2025, 10, 39751–39762.
- 105.
Zhang, S.; Collier, M.E.W.; Heyes, D.J.; Giorgini, F.; Scrutton, N.S. Advantages of brain penetrating inhibitors of kynurenine-3-monooxygenase for treatment of neurodegenerative diseases. Arch. Biochem. Biophys. 2021, 697, 108702.
- 106.
Wang, A.; Guan, C.; Wang, T.; Mu, G.; Tuo, Y. Lactobacillus-derived indole derivatives ameliorate intestinal barrier damage in rat pups with complementary food administration. Food Funct. 2024, 15, 8775–8787.
- 107.
Geng, X.; Liu, J.; Feng, H.; Xu, J.; Gu, J.; Ding, X.; Zhou, G.; Zhang, T.; Liu, Y.; Wang, M.; et al. Glehnia littoralis polysaccharides alleviate lung and intestinal mucosal barrier damage in rats with ulcerative colitis via aryl hydrocarbon receptor activation. Int. J. Biol. Macromol. 2025, 327, 147318.
- 108.
He, Z.; Guo, J.; Zhang, H.; Yu, J.; Zhou, Y.; Wang, Y.; Li, T.; Yan, M.; Li, B.; Chen, Y.; et al. Atractylodes macrocephala Koidz polysaccharide improves glycolipid metabolism disorders through activation of aryl hydrocarbon receptor by gut flora-produced tryptophan metabolites. Int. J. Biol. Macromol. 2023, 253, 126987.
- 109.
Burton, T.C.; Lv, N.; Tsai, P.; Penalver Bernabe, B.; Tussing-Humphreys, L.; Xiao, L.; Pandey, G.N.; Wu, Y.; Ajilore, O.A.; Ma, J. Associations between fecal short-chain fatty acids, plasma inflammatory cytokines, and dietary markers with depression and anxiety: Post hoc analysis of the ENGAGE-2 pilot trial. Am. J. Clin. Nutr. 2023, 117, 717–730.
- 110.
Qian, X.; Li, Q.; Zhu, H.; Chen, Y.; Lin, G.; Zhang, H.; Chen, W.; Wang, G.; Tian, P. Bifidobacteria with indole-3-lactic acid-producing capacity exhibit psychobiotic potential via reducing neuroinflammation. Cell Rep. Med. 2024, 5, 101798.
- 111.
Wang, H.; Liang, Q.; Wen, Z.; Ma, W.; Ji, S.; Zhang, H.; Zhang, X. Enriched environment alleviates NLRP3 inflammasome mediated neuroinflammation in diabetes complicated with depression rats. Sci. Rep. 2025, 15, 14214.
- 112.
Gupta, S.K.; Vyavahare, S.; Duchesne Blanes, I.L.; Berger, F.; Isales, C.; Fulzele, S. Microbiota-derived tryptophan metabolism: Impacts on health, aging, and disease. Exp. Gerontol. 2023, 183, 112319.
- 113.
Chew, C.; Matsuyama, M.; Davies, P.S.W.; Hill, R.J.; Morrison, M.; Martin, R.; Codoñer, F.M.; Knol, J.; Roeselers, G. A young child formula supplemented with a synbiotic mixture of scGOS/lcFOS and Bifidobacterium breve M-16V improves the gut microbiota and iron status in healthy toddlers. Front. Pediatr. 2024, 12, 1193027.
- 114.
Dinan, T.G.; Cryan, J.F. Melancholic microbes: A link between gut microbiota and depression? Neurogastroenterol. Motil. 2013, 25, 713–719.
- 115.
Slykerman, R.F.; Hood, F.; Wickens, K.; Thompson, J.M.D.; Barthow, C.; Murphy, R.; Kang, J.; Rowden, J.; Stone, P.; Crane, J.; et al.; Probiotic in Pregnancy Study Group. Effect of Lactobacillus rhamnosus HN001 in Pregnancy on Postpartum Symptoms of Depression and Anxiety: A Randomised Double-blind Placebo-controlled Trial. EBioMedicine 2017, 24, 159–165.
- 116.
Majeed, M.; Nagabhushanam, K.; Arumugam, S.; Majeed, S.; Ali, F. Bacillus coagulans MTCC 5856 for the management of major depression with irritable bowel syndrome: A randomised, double-blind, placebo controlled, multi-centre, pilot clinical study. Food Nutr. Res. 2018, 62, 1218.

This work is licensed under a Creative Commons Attribution 4.0 International License.


